Acids Bases and Salts Notes 13 Properties of

Acids, Bases and Salts Notes #13

Properties of acids 1. Sour 2. React with active metals to produce Hydrogen gas 3. Electrolytes 4. React with bases to produce salt and water

Properties of Bases 1. Bitter 2. Slippery 3. React with acids to produce salt and water 4. Electrolytes

Arrhenius Definitions Acids: increase hydrogen ion concentrations in solution. (Have ionizable hydrogen) Examples: HCl, H 2 SO 4, HNO 3 Bases: increase hydroxide ion concentrations in solution. Examples: Na. OH, Mg(OH)2, Fe(OH)3 Salt: the cation of a base plus the anion of an acid. Examples Na. Cl, Mg. SO 4, Fe(NO 3)3
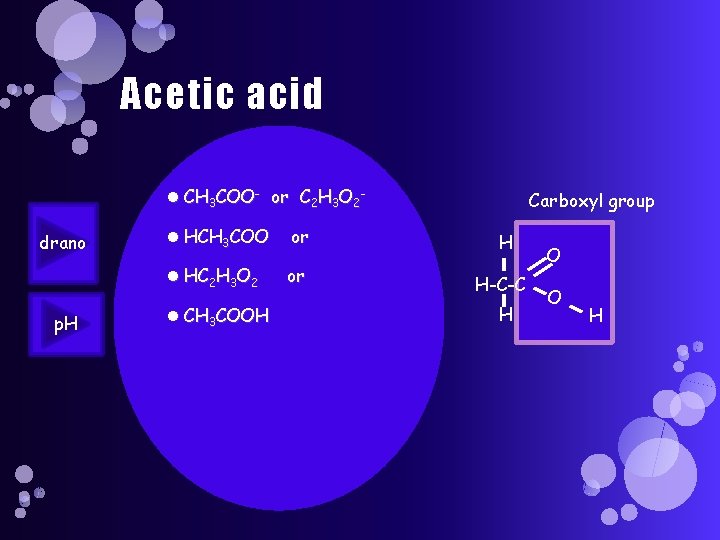
Acetic acid CH 3 COO- or C 2 H 3 O 2 drano p. H HCH 3 COO or HC 2 H 3 O 2 or CH 3 COOH Carboxyl group H H-C-C H O O H

Neutralization reaction HCl + Na. OH → Na. Cl + H 2 O H 2 SO 4 + Mg(OH)2 → Mg. SO 4 + H 2 O
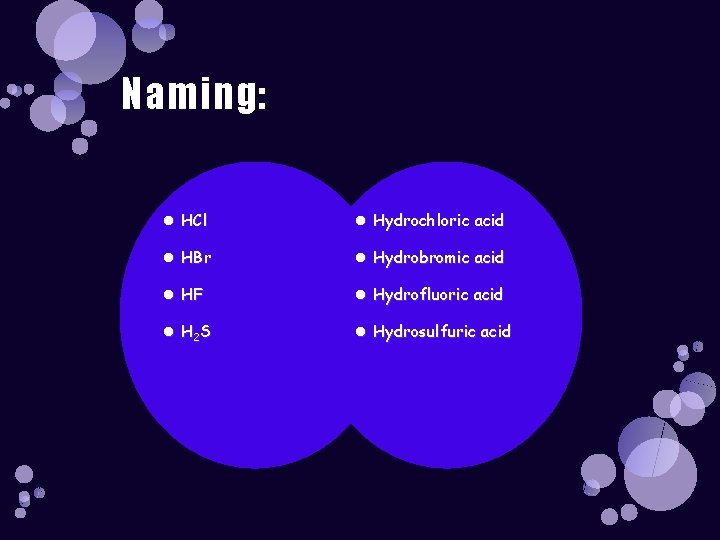
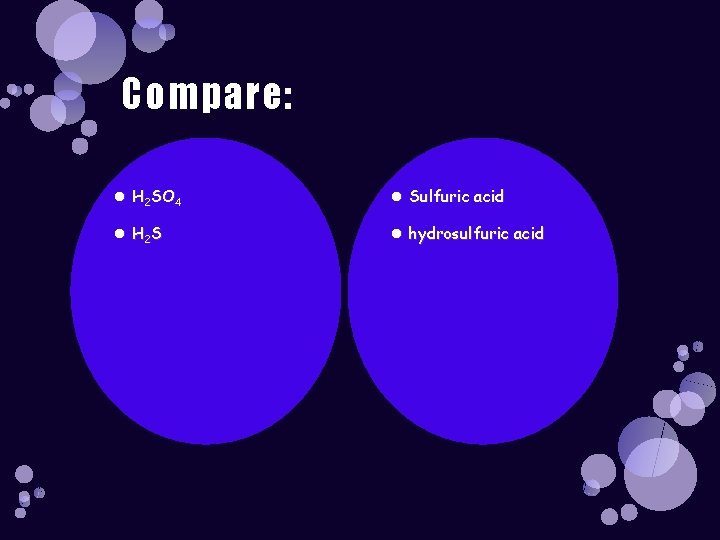
Naming: HCl Hydrochloric acid HBr Hydrobromic acid HF Hydrofluoric acid H 2 S Hydrosulfuric acid

Naming: Binary Acids HCl Hydrochloric acid HBr Hydrobromic acid HF Hydrofluoric acid H 2 S Hydrosulfuric acid
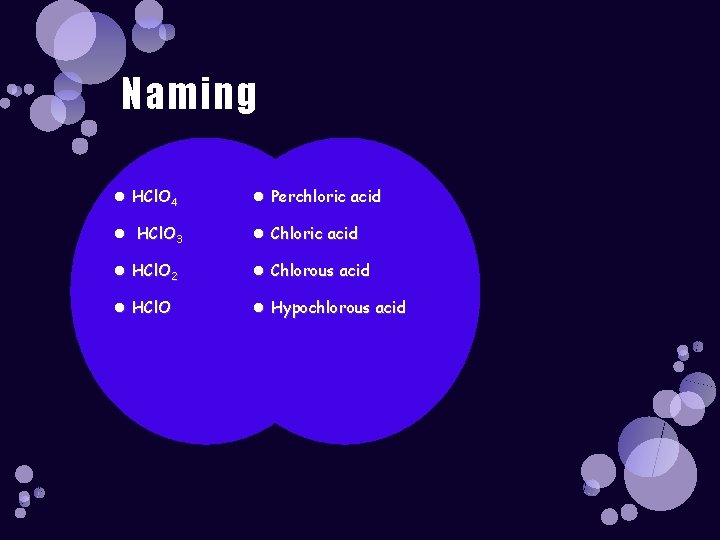
Naming HCl. O 4 Perchloric acid HCl. O 3 Chloric acid HCl. O 2 Chlorous acid HCl. O Hypochlorous acid
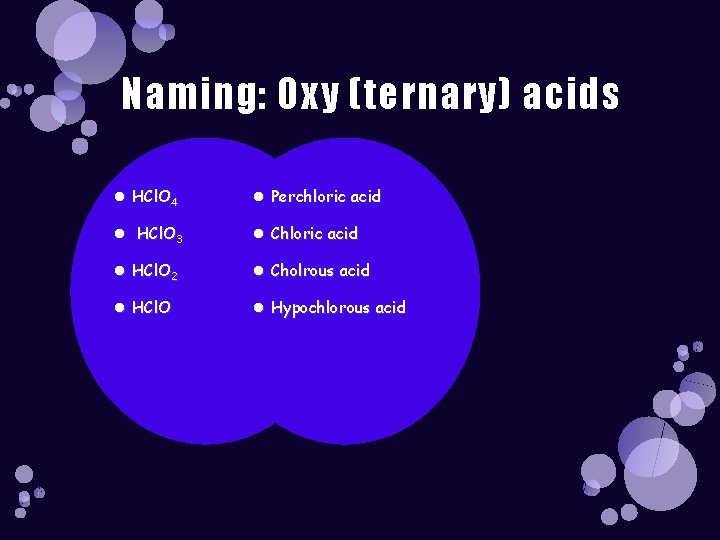
Naming: Oxy (ternary) acids HCl. O 4 Perchloric acid HCl. O 3 Chloric acid HCl. O 2 Cholrous acid HCl. O Hypochlorous acid

Now write the formulas and names of the sulfate family of acids H 2 SO 5 H 2 SO 4 H 2 SO 3 H 2 SO 2 Persulfuric acid Sulfuric acid sulfurous acid Hyposulfurous acid
Compare: H 2 SO 4 Sulfuric acid H 2 S hydrosulfuric acid
- Slides: 12