Acids Bases and Buffers The BrnstedLowry definitions of

Acids, Bases and Buffers The BrØnsted-Lowry definitions of an acid and a base are: Acid: species that donates a proton Base: species that can accept a proton When an acid is added to water, the acid dissociates releasing H+ ions into solution. Depending on the formula and bonding, different acids can release different numbers of protons: HCl H+ + Cl- - this is monoprotic as it releases one proton H 2 SO 4 2 H+ + SO 42 - - this is diprotic as it releases two protons H 3 PO 4 3 H+ + PO 43 - - this is triprotic as is releases three protons Writing ionic equations 1. Write out the balanced chemical equation 2. Split up each compound into its ions – be careful with solids – they don’t dissociate, only aqueous solutions dissociate! 3. Cancel species that appear on both sides of the equation Example: Aqueous sodium carbonate and hydrochloric acid Na 2 CO 3 + 2 HCl 2 Na. Cl + CO 2 + H 2 O 2 Na+ + CO 32 - + 2 H+ + 2 Cl- 2 Na+ + 2 Cl- + CO 2 + H 2 O CO 32 - + 2 H+ CO 2 + H 2 O
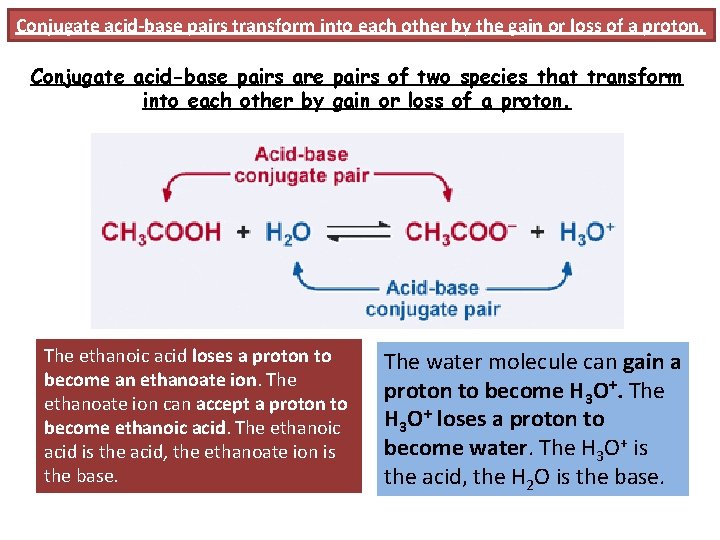
Conjugate acid-base pairs transform into each other by the gain or loss of a proton. Conjugate acid-base pairs are pairs of two species that transform into each other by gain or loss of a proton. The ethanoic acid loses a proton to become an ethanoate ion. The ethanoate ion can accept a proton to become ethanoic acid. The ethanoic acid is the acid, the ethanoate ion is the base. The water molecule can gain a proton to become H 3 O+. The H 3 O+ loses a proton to become water. The H 3 O+ is the acid, the H 2 O is the base.
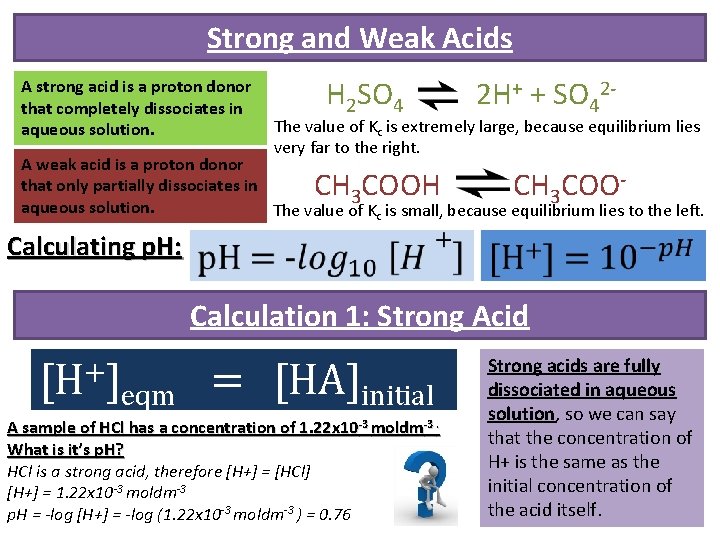
Strong and Weak Acids A strong acid is a proton donor that completely dissociates in aqueous solution. H 2 SO 4 2 H+ + SO 42 - The value of Kc is extremely large, because equilibrium lies very far to the right. A weak acid is a proton donor that only partially dissociates in 3 3 aqueous solution. The value of Kc is small, because equilibrium lies to the left. CH COOH CH COO Calculating p. H: Calculation 1: Strong Acid [H+] eqm = [HA]initial A sample of HCl has a concentration of 1. 22 x 10 -3 moldm-3. What is it’s p. H? HCl is a strong acid, therefore [H+] = [HCl] [H+] = 1. 22 x 10 -3 moldm-3 p. H = -log [H+] = -log (1. 22 x 10 -3 moldm-3 ) = 0. 76 Strong acids are fully dissociated in aqueous solution, so we can say that the concentration of H+ is the same as the initial concentration of the acid itself.

Calculation 2: Strong base using Kw Kw is known as the ionic product of water. Kw has a value of 1 x 10 -14 at 25°. The definition of Kw is represented by the following equation: A strong base fully dissociates in aqueous solution. Therefore, we can say the concentration of OH- is the same as the initial A solution of KOH has a concentration of the base. of 0. 050 moldm-3. What is its p. H? Kw = + [H ][OH ] We can use the Kw equation in rearranged form to find the [H+] and thereby find the p. H. = 2 x 10 -13 p. H = -log [H+] = -log (2 x 10 -3 ) = 12. 70 Ka : The Acid Dissociation Constant The acid Ka values can be made more dissociation manageable if expressed in logarithmic constant, Ka, shows form, called p. Ka : the extent of the dissociation of an Strong Acids have high Ka values and low acid. It is given by p. Ka values – this indicates a large extent of dissociation. the expression:

Calculation 3: Weak Acid using Ka The concentration of a sample of nitrous acid HNO 2 is 0. 055 moldm-3. Ka = 4. 7 x 10 -4. Calculate the p. H. = 2. 29
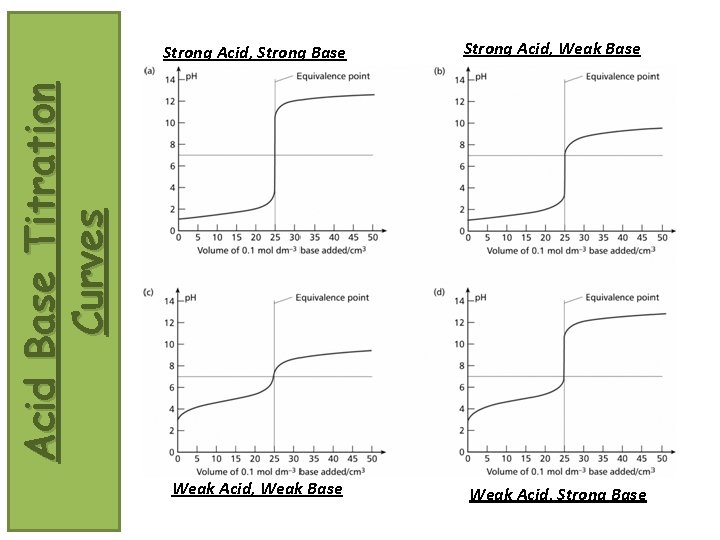
Strong Acid, Weak Base Acid Base Titration Curves Strong Acid, Strong Base Weak Acid, Weak Base Weak Acid, Strong Base
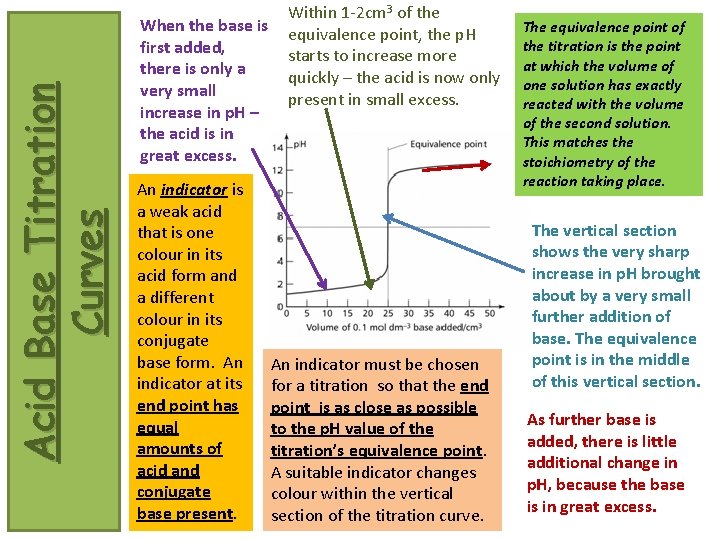
Acid Base Titration Curves When the base is first added, there is only a very small increase in p. H – the acid is in great excess. An indicator is a weak acid that is one colour in its acid form and a different colour in its conjugate base form. An indicator at its end point has equal amounts of acid and conjugate base present. Within 1 -2 cm 3 of the equivalence point, the p. H starts to increase more quickly – the acid is now only present in small excess. An indicator must be chosen for a titration so that the end point is as close as possible to the p. H value of the titration’s equivalence point. A suitable indicator changes colour within the vertical section of the titration curve. The equivalence point of the titration is the point at which the volume of one solution has exactly reacted with the volume of the second solution. This matches the stoichiometry of the reaction taking place. The vertical section shows the very sharp increase in p. H brought about by a very small further addition of base. The equivalence point is in the middle of this vertical section. As further base is added, there is little additional change in p. H, because the base is in great excess.

Buffer Solutions A buffer solution is a system that minimises the p. H change on addition of small amounts of acid or base. A buffer system is made from a weak acid and a salt of the weak acid – for example, methanoic acid and sodium methanoate. Alternatively, the weak acid could be partially neutralised by a an aqueous alkali such as Na. OH, to give a solution containing a mixture of the salt and an excess of the weak acid. How does a buffer system work? In the ethanoic acid/sodium ethanoate buffer system: The weak acid CH 3 COOH dissociates partially: CH 3 COOH H+ + CH 3 COO- The salt (conjugate base), CH 3 COO-Na+ dissociates completely: CH 3 COO-Na+ CH 3 COO- + Na+ The equilibrium mixture formed contains a high concentration of the weak acid and its conjugate base. The resulting buffer solution contains large reservoirs of the weak acid and its conjugate base , both of which control p. H. The overall principle of the buffer solution is that the weak acid removes added alkali and the conjugate base removes added acid. On addition of acid: On addition of alkali: § [H+] increased § [OH-] increased § Conjugate base CH 3 COO- reacts with H+ § Added OH- reacts with H+ to form H 2 O § Equilibrium shifts left removing most of added H+ ions. § CH 3 COOH dissociates to form CH 3 COO- + H+. Equilibrium shifts right restoring most of H+ ions that have reacted.
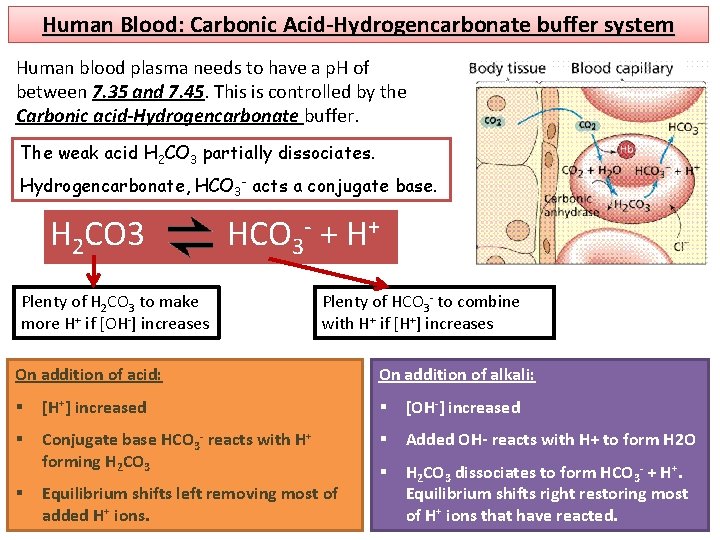
Human Blood: Carbonic Acid-Hydrogencarbonate buffer system Human blood plasma needs to have a p. H of between 7. 35 and 7. 45. This is controlled by the Carbonic acid-Hydrogencarbonate buffer. The weak acid H 2 CO 3 partially dissociates. Hydrogencarbonate, HCO 3 - acts a conjugate base. H 2 CO 3 HCO 3 - + H+ Plenty of H 2 CO 3 to make more H+ if [OH-] increases Plenty of HCO 3 - to combine with H+ if [H+] increases On addition of acid: On addition of alkali: § [H+] increased § [OH-] increased § Conjugate base HCO 3 - reacts with H+ forming H 2 CO 3 § Added OH- reacts with H+ to form H 2 O § H 2 CO 3 dissociates to form HCO 3 - + H+. Equilibrium shifts right restoring most of H+ ions that have reacted. § Equilibrium shifts left removing most of added H+ ions.
Buffer Solutions The p. H of a buffer depends on the amount of dissociation, the temperature and the concentration ratio of the weak acid and its conjugate base. The expression used for p. H of a buffer solution is: Calculation 4: p. H of a buffer solution Calculate the p. H of the following buffer solution: 50 cm 3 of 1. 0 moldm-3 methanoic acid, Ka = 1. 78 x 10 -4 moldm-3; 20 cm 3 of 1. 0 mol dm-3 sodium methanoate. Number moles acid = 1 x 50 x 10 -3 = 0. 05 Number moles sodium methanoate = 1 x 20 x 10 -3 = 0. 02

Calculation 5: p. H change of a buffer solution How would we calculate the p. H change when 10 cm 3 of 0. 1 moldm-3 Na. OH is added to the buffer solution in the previous example? When Na. OH is added, a reaction occurs between What we already know about the acid and the Na. OH base: buffer solution: HCOOH + OH- HCOO- + H 2 O Initial moles of acid = 0. 05 moles Initial moles of Na. COOH = 0. 02 moles So, for every one mole of Na. OH added, we get one Initial p. H of buffer = 3. 35 mole more of the conjugate base, Na. COOH and -4 -3 Ka = 1. 78 x 10 moldm one mole less of acid. Moles Na. OH added: 0. 1 x 10 -3 = 0. 001 Therefore, the amount of acid present will go down by this amount and the amount of conjugate base will go up by this amount. We can now use a Ka Initial Moles Change Final Moles HCOOH 0. 05 -0. 001 0. 049 Na. COOH 0. 02 +0. 001 0. 021 calculation with the new amounts present in the buffer solution to calculate the p. H: p. H= -log(4. 15 x 10 -4) = 3. 38 therefore p. H change = 3. 38 – 3. 35 = 0. 03

Calculation 6: Neutralisation 1. Calculate moles of H+ before reaction. 2. Calculate moles of OHbefore reaction 3. Calculate excess amount of H+ or OH- remaining after the reaction 4. Calculate excess concentration of OH- or H+ 5. Find p. H suing appropriate calculation. Calculate the p. H when 62 cm 3 of 1. 05 moldm-3 Na. OH is added to 23 cm 3 of 0. 91 moldm-3 HCl. Calculate the p. H when 35 cm 3 of 1. 25 moldm-3 Na. OH is added to 73 cm 3 of 0. 92 moldm-3 HCl.
- Slides: 12