Acids Bases and Buffers OH MY Acids Bases

Acids, Bases and Buffers OH MY!!
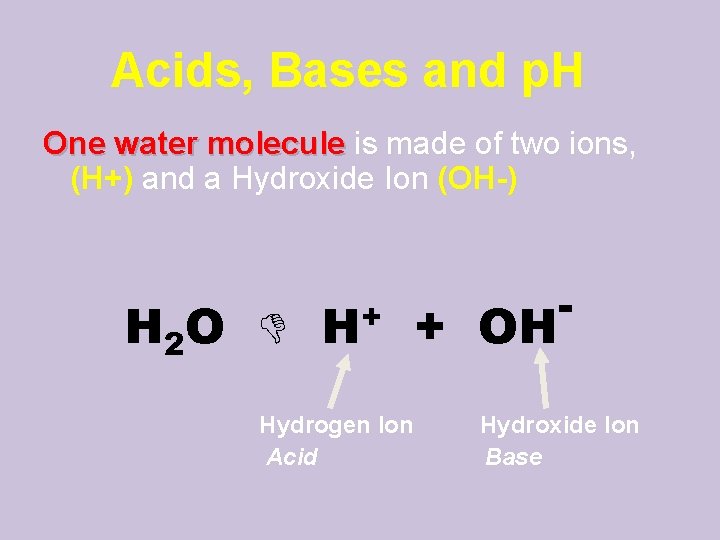
Acids, Bases and p. H One water molecule is made of two ions, (H+) and a Hydroxide Ion (OH-) H 2 O H+ + OH Hydrogen Ion Acid - Hydroxide Ion Base

Acids and Bases Acid: A solution with lots of H+ ions • p. H 0 up to 7 is acid (acidic) Base: A solution with lots of OH- ions • p. H above 7 – 14 is basic (alkaline) b

The p. H Scale…acids • Indicates the percent of H+ ions • Ranges from 0 – 14 • p. H of 7 is neutral • Each p. H unit represents a factor of 10 X change in concentration – p. H 3 is 10 x 10 (1000 x) stronger than a p. H of 6
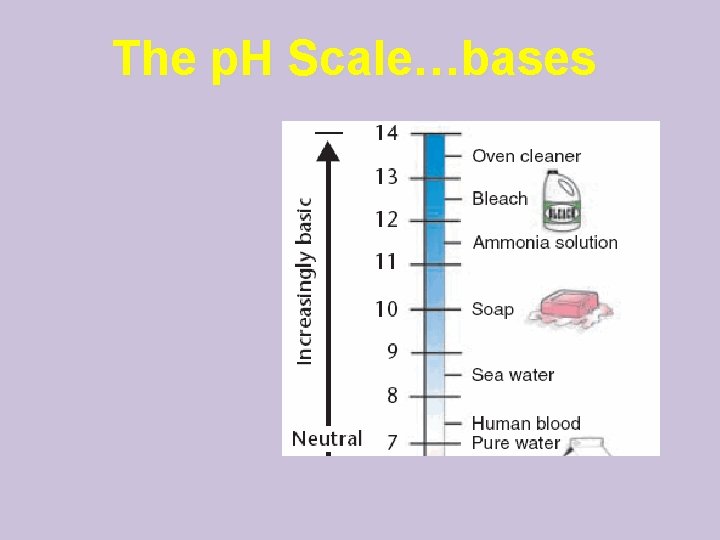
The p. H Scale…bases

Buffers • Weak acids or bases that react with strong acids or bases to prevent sharp, sudden changes in p. H (neutralization). • Produced naturally by the body to maintain homeostasis Weak Acid Weak Base

Procedure: Wear Goggles at all times! 1. Add 50 m. L of cabbage juice indicator 2. Half teaspoon baking soda to one beaker— label “Buffer” 3. Label other beaker “Control. ” 4. Half teaspoon of Sprite to “Buffer” beaker. Stir. 5. Continue adding Sprite until “Buffer” matches other beaker color. 5. Add 2 drops of vinegar to “Control” beaker-swirl. 6. Repeat with “Buffer. ” 7. Alternate adding vinegar. 8. Observe color reactions and record amounts of vinegar.

After Clean Up! • What is alkalosis? • Answer • What could lead questions in #10. to alkalosis in the body? • Add the • What is acidosis? following after reading the info • What could lead to acidosis in the on the reverse body? side:

Ticket out the Door 1. Draw a water molecule with labeled atoms and charges. 2. What is the property called that describes water sticking to itself? 3. What makes a solution acidic? 4. What has a higher p. H, bleach or lemon juice?
- Slides: 9