Acids Bases Acids vacids are sour tasting v

Acids & Bases Acids: vacids are sour tasting v. Arrhenius acid: acid Any substance that, when dissolved in water, increases the concentration of hydronium ion (H 3 O+) v. Bronsted-Lowry acid: acid A proton donor v. Lewis acid: acid An electron acceptor Bases: vbases are bitter tasting and slippery v. Arrhenius base: base Any substance that, when dissolved in water, increases the concentration of hydroxide ion (OH-) v. Bronsted-Lowery base: base A proton acceptor v. Lewis acid: acid An electron donor
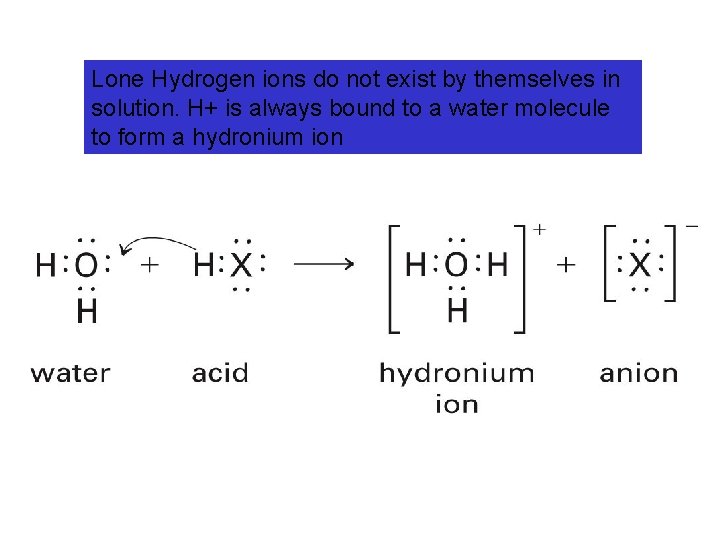
Lone Hydrogen ions do not exist by themselves in solution. H+ is always bound to a water molecule to form a hydronium ion
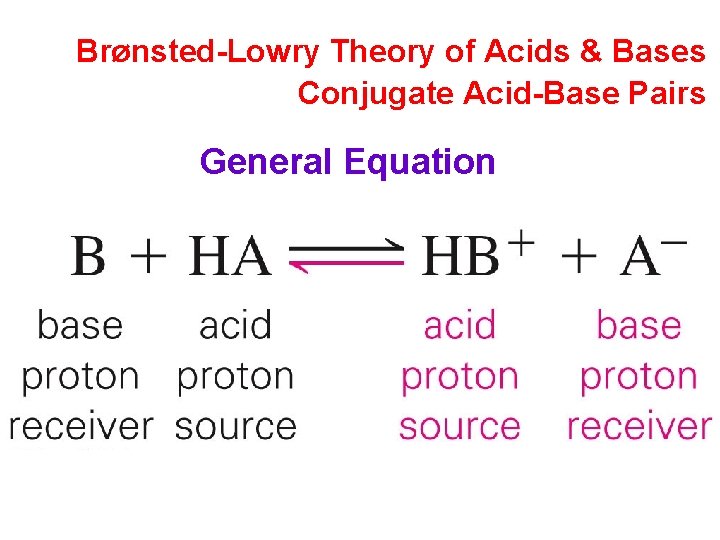
Brønsted-Lowry Theory of Acids & Bases Conjugate Acid-Base Pairs General Equation
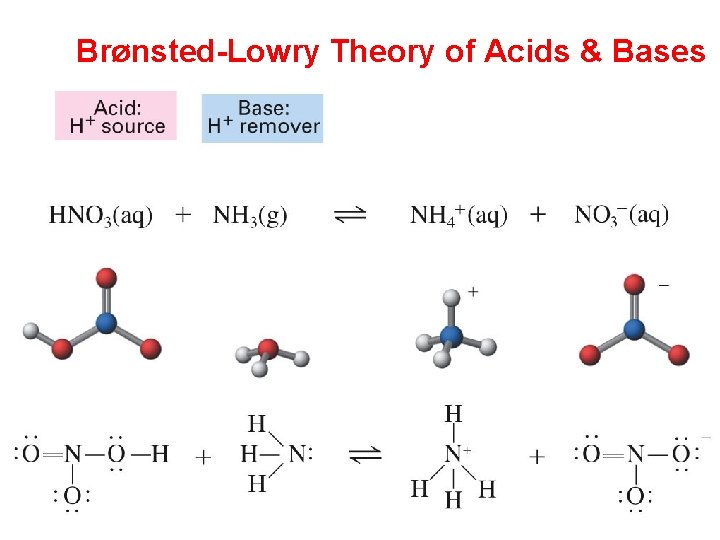
Brønsted-Lowry Theory of Acids & Bases
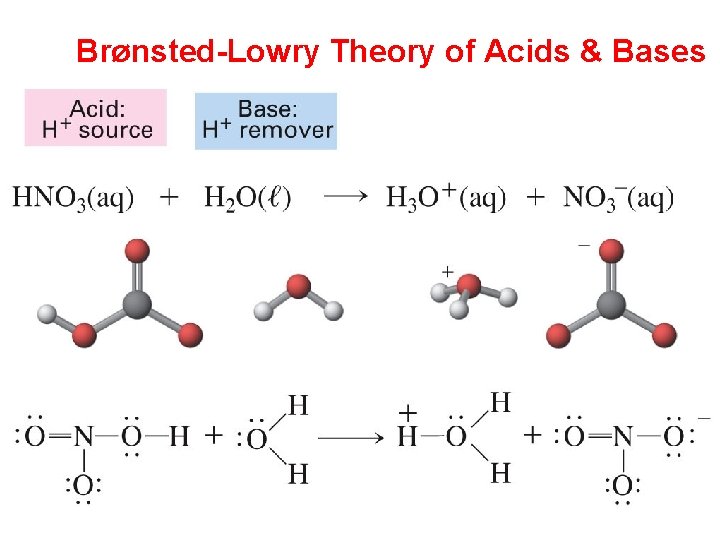
Brønsted-Lowry Theory of Acids & Bases
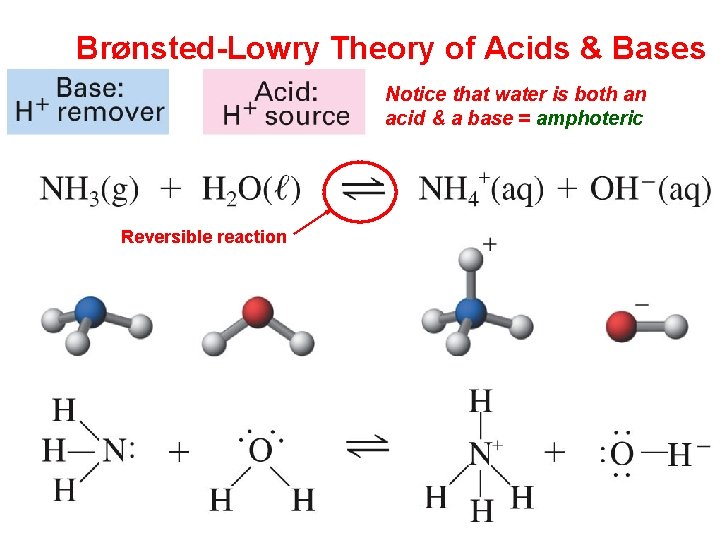
Brønsted-Lowry Theory of Acids & Bases Notice that water is both an acid & a base = amphoteric Reversible reaction

ELECTROLYTES Electrolytes are species which conducts electricity when dissolved in water. Acids, Bases, and Salts are all electrolytes. Salts and strong Acids or Bases form Strong Electrolytes. Salt and strong acids (and bases) are fully dissociated therefore all of the ions present are available to conduct electricity. HCl(s) + H 2 O H 3 O+ + Cl. Weak Acids and Weak Bases for Weak Electrolytes. Weaks electrolytes are partially dissociated therefore not all species in solution are ions, some of the molecular form is present. Weak electrolytes have less ions avalible to conduct electricity. NH 3 + H 2 O NH 4+ + OH-
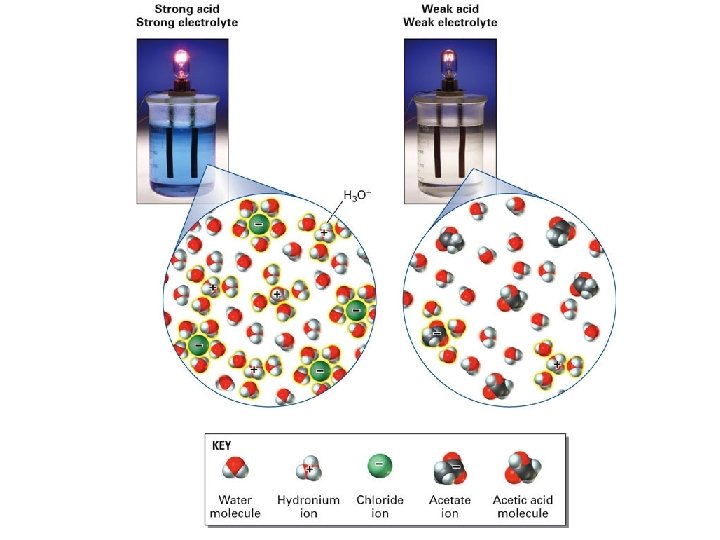
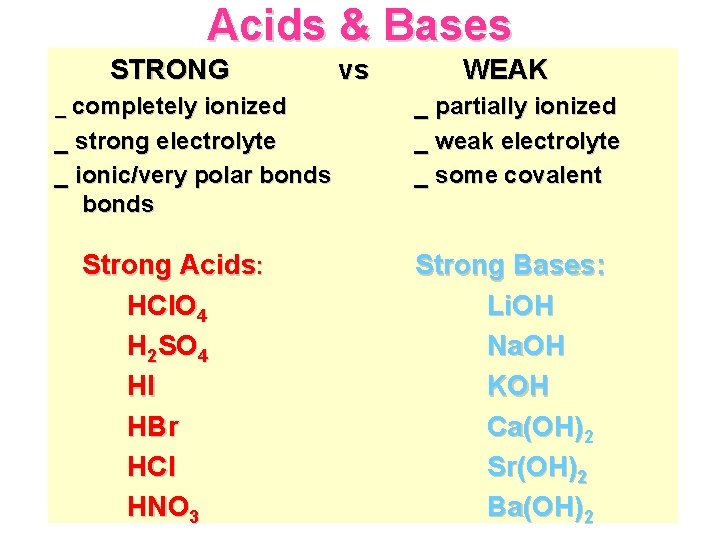
Acids & Bases STRONG _ completely ionized _ strong electrolyte _ ionic/very polar bonds Strong Acids: HCl. O 4 H 2 SO 4 HI HBr HCl HNO 3 vs WEAK _ partially ionized _ weak electrolyte _ some covalent Strong Bases: Li. OH Na. OH KOH Ca(OH)2 Sr(OH)2 Ba(OH)2
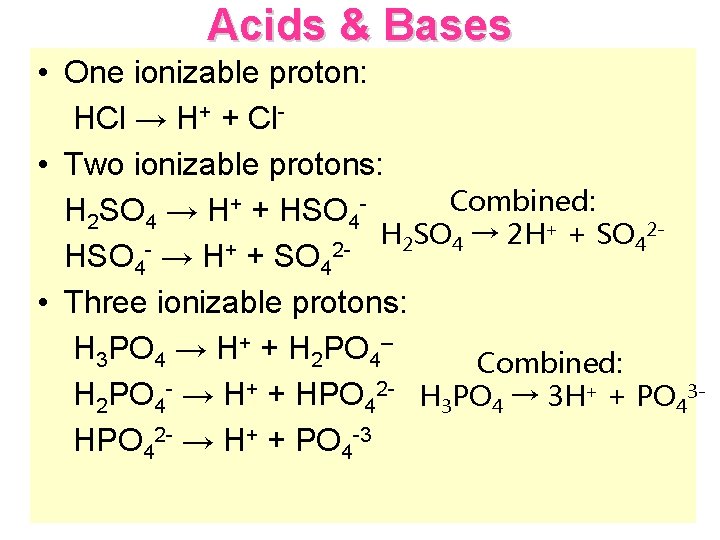
Acids & Bases • One ionizable proton: HCl → H+ + Cl • Two ionizable protons: Combined: H 2 SO 4 → H+ + HSO 4+ + SO 2 H SO → 2 H 2 4 4 + 2 HSO 4 → H + SO 4 • Three ionizable protons: H 3 PO 4 → H+ + H 2 PO 4– Combined: H 2 PO 4 - → H+ + HPO 42 - H 3 PO 4 → 3 H+ + PO 43 HPO 42 - → H+ + PO 4 -3
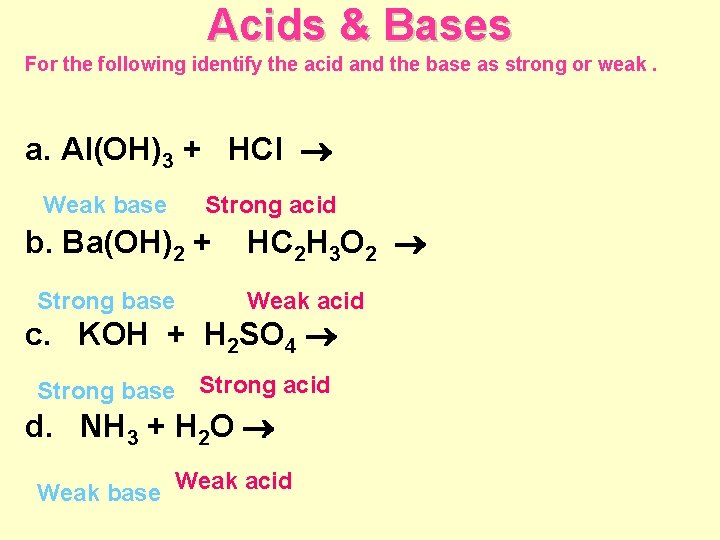
Acids & Bases For the following identify the acid and the base as strong or weak. a. Al(OH)3 + HCl Weak base Strong acid b. Ba(OH)2 + Strong base HC 2 H 3 O 2 Weak acid c. KOH + H 2 SO 4 Strong base Strong acid d. NH 3 + H 2 O Weak base Weak acid
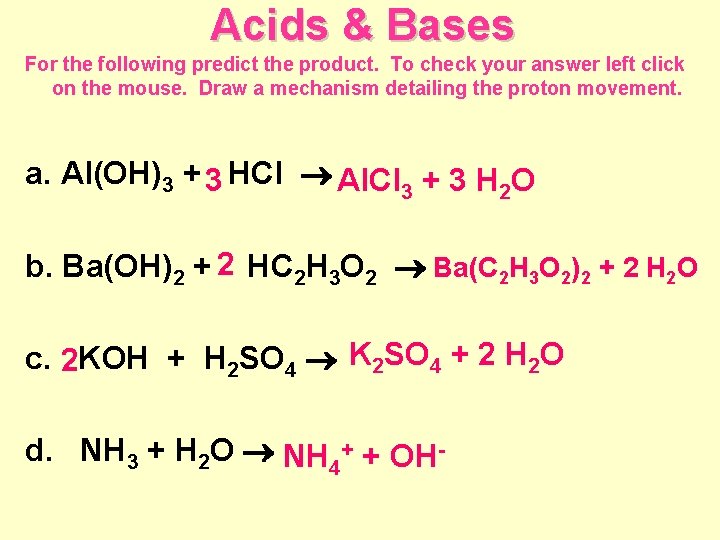
Acids & Bases For the following predict the product. To check your answer left click on the mouse. Draw a mechanism detailing the proton movement. a. Al(OH)3 + 3 HCl Al. Cl 3 + 3 H 2 O b. Ba(OH)2 + 2 HC 2 H 3 O 2 Ba(C 2 H 3 O 2)2 + 2 H 2 O c. 2 KOH + H 2 SO 4 K 2 SO 4 + 2 H 2 O d. NH 3 + H 2 O NH 4+ + OH-
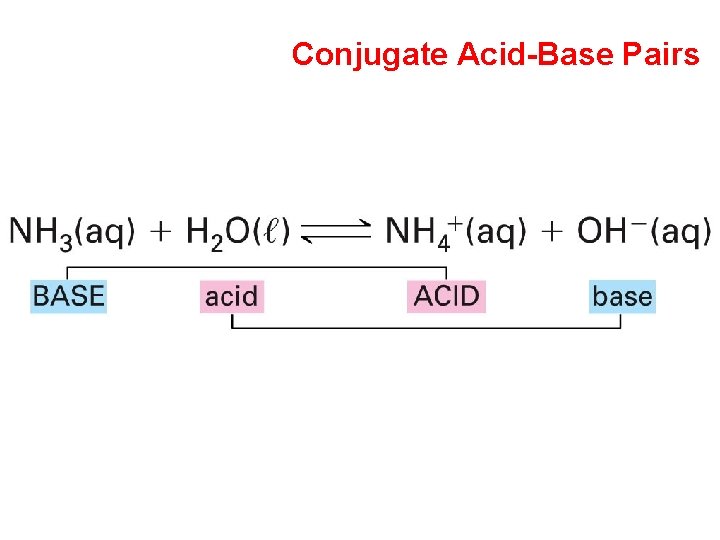
Conjugate Acid-Base Pairs
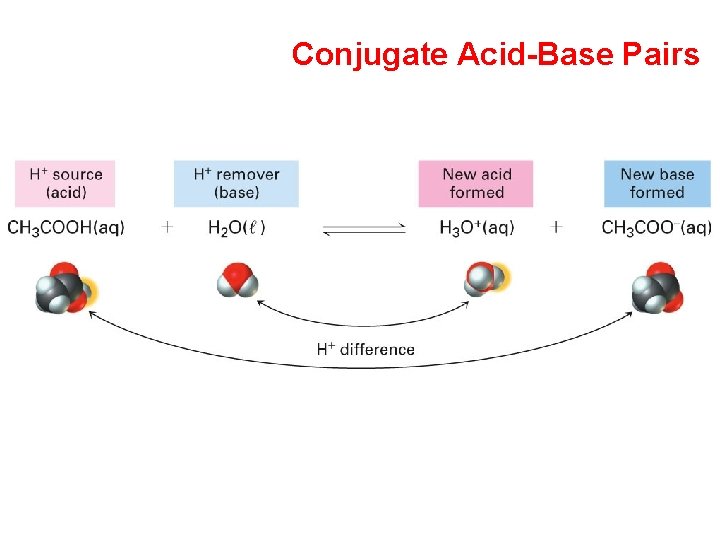
Conjugate Acid-Base Pairs

Acids & Bases For the following Identify the conjugate acid and the conjugate base. The conjugate refers to the acid or base produced in an acid/base reaction. The acid reactant produces its conjugate base (CB). CB a. Al(OH)3 + 3 HCl Al. Cl 3 + 3 H 2 O CB CA b. Ba(OH)2 + 2 HC 2 H 3 O 2 Ba(C 2 H 3 O 2)2 + 2 H 2 O CB c. 2 KOH + H 2 SO 4 K 2 SO 4 + 2 H 2 O CB d. NH 3 + H 2 O NH 4+ + OHCA CB CA CA

TITRATION Titration of a strong acid with a strong base ENDPOINT = POINT OF NEUTRALIZATION = EQUIVALENCE POINT At the end point for the titration of a strong acid with a strong base, the moles of acid (H+) equals the moles of base (OH-) to produce the neutral species water (H 2 O). If the mole ratio in the balanced chemical equation is 1: 1 then the following equation can be used. MOLES OF ACID = MOLES OF BASE nacid = nbase Since M=n/V M AV A = M B V B

TITRATION M AV A = M B V B 1. Suppose 75. 00 m. L of hydrochloric acid was required to neutralize 22. 50 m. Lof 0. 52 M Na. OH. What is the molarity of the acid? HCl + Na. OH H 2 O + Na. Cl Ma Va = Mb Vb rearranges to Ma = Mb Vb / Va so Ma = (0. 52 M) (22. 50 m. L) / (75. 00 m. L) = 0. 16 M Now you try: 2. If 37. 12 m. L of 0. 843 M HNO 3 neutralized 40. 50 m. L of KOH, what is the molarity of the base? Mb = 0. 773 mol/L

Molarity and Titration

TITRATION Titration of a strong acid with a strong base ENDPOINT = POINT OF NEUTRALIZATION = EQUIVALENCE POINT At the end point for the titration of a strong acid with a strong base, the moles of acid (H+) equals the moles of base (OH-) to produce the neutral species water (H 2 O). If the mole ratio in the balanced chemical equation is NOT 1: 1 then you must rely on the mole relationship and handle the problem like any other stoichiometry problem. MOLES OF ACID = MOLES OF BASE nacid = nbase

TITRATION 1. If 37. 12 m. L of 0. 543 M Li. OH neutralized 40. 50 m. L of H 2 SO 4, what is the molarity of the acid? 2 Li. OH + H 2 SO 4 Li 2 SO 4 + 2 H 2 O First calculate the moles of base: 0. 03712 L Li. OH (0. 543 mol/1 L) = 0. 0202 mol Li. OH Next calculate the moles of acid: 0. 0202 mol Li. OH (1 mol H 2 SO 4 / 2 mol Li. OH)= 0. 0101 mol H 2 SO 4 Last calculate the Molarity: Ma = n/V = 0. 010 mol H 2 SO 4 / 0. 4050 L = 0. 248 M 2. If 20. 42 m. L of Ba(OH)2 solution was used to titrate 29. 26 m. L of 0. 430 M HCl, what is the molarity of the barium hydroxide solution? Mb = 0. 308 mol/L

Molarity and Titration • A student finds that 23. 54 m. L of a 0. 122 M Na. OH solution is required to titrate a 30. 00 -m. L sample of hydr acid solution. What is the molarity of the acid? • A student finds that 37. 80 m. L of a 0. 4052 M Na. HCO 3 solution is required to titrate a 20. 00 m. L sample of sulfuric acid solution. What is the molarity of the acid? • The reaction equation is: H 2 SO 4 + 2 Na. HCO 3 → Na 2 SO 4 + 2 H 2 O + 2 CO 2
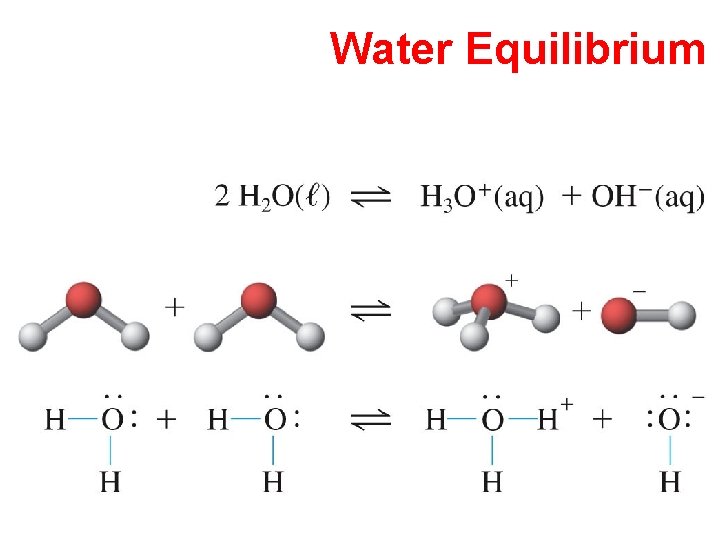
Water Equilibrium
![Water Equilibrium Kw = [H+] [OH-] = 1. 0 x 10 -14 Equilibrium constant Water Equilibrium Kw = [H+] [OH-] = 1. 0 x 10 -14 Equilibrium constant](http://slidetodoc.com/presentation_image/88f8df126ee5080c56d1998e8d8c4b06/image-23.jpg)
Water Equilibrium Kw = [H+] [OH-] = 1. 0 x 10 -14 Equilibrium constant for water q Water or water solutions in which [H+] = [OH-] = 10 -7 M are neutral solutions. q A solution in which [H+] > [OH-] is acidic q A solution in which [H+] < [OH-] is basic

p. H A measure of the hydronium ion • The scale for measuring the hydronium ion concentration [H 3 O+] in any solution must be able to cover a large range. A logarithmic scale covers factors of 10. The “p” in p. H stands for log. • A solution with a p. H of 1 has [H 3 O+] of 0. 1 mol/L or 10 -1 • A solution with a p. H of 3 has [H 3 O+] of 0. 001 mol/L or 10 -3 • A solution with a p. H of 7 has [H 3 O+] of 0. 0000001 mol/L or 10 -7 p. H = - log [H 3 O+]

The p. H scale ranges from 1 to 10 -14 mol/L or from 1 to 14. p. H = - log [H 3 O+] 1 2 3 4 5 6 7 8 9 10 11 12 13 14 acid neutral base
![Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+] Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+]](http://slidetodoc.com/presentation_image/88f8df126ee5080c56d1998e8d8c4b06/image-26.jpg)
Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+] allows for: [H 3 O+] = 10 -p. H If p. H is a measure of the hydronium ion concentration the same equations could be used to describe the hydroxide (base) concentration. [OH-] = 10 -p. OH = - log [OH-] thus: p. H + p. OH = 14 ; the entire p. H range!

PRACTICE PROBLEM #25 1. How many milliliters of 1. 25 M Li. OH must be added to neutralize 34. 7 m. L of 0. 389 M HNO 3? 10. 8 m. L 2. What mass of Sr(OH)2 will be required to neutralize 19. 54 m. L of 0. 00850 M HBr solution? 0. 0101 g 3. How many m. L of 0. 998 M H 2 SO 4 must be added to neutralize 47. 9 m. L of 1. 233 M KOH? 29. 6 m. L 4. What is the molar concentration of hydronium ion in a solution of p. H 8. 25? -9 5. 623 x 10 M 5. What is the p. H of a solution that has a molar concentration of hydronium ion of 9. 15 x 10 -5? p. H = 4. 0 6. What is the p. OH of a solution that has a molar concentration of hydronium ion of 8. 55 x 10 -10? p. OH = 4. 9

GROUP STUDY PROBLEM #25 ______1. How many milliliters of 0. 75 M KOH must be added to neutralize 50. 0 m. L of 2. 50 M HCl? ______2. What mass of Ca(OH)2 will be required to neutralize 100 m. L of 0. 170 M HCl solution? ______3. How many m. L of 0. 554 M H 2 SO 4 must be added to neutralize 25. 0 m. L of 0. 9855 M Na. OH? ______ 4. What is the molar concentration of hydronium ion in a solution of p. H 2. 45? ______ 5. What is the p. H of a solution that has a molar concentration of hydronium ion of 3. 75 x 10 -9? ______ 6. What is the p. OH of a solution that has a molar concentration of hydronium ion of 4. 99 x 10 -4?
- Slides: 28