ACIDS BASES ACIDS Properties of Acids Sour Taste

ACIDS & BASES

ACIDS Properties of Acids: § Sour Taste § Electrolytes § Change indicator colors § React with metals to form hydrogen § React with carbonates to form carbon dioxide § React with bases to form salt & water § Produces H + in water Naming Acids § Look at ending § -ate polyatomic ions -ic acids § example: § -ite polyatomic ions -ous acids § example: § -ide monatomic ions hydro –ic acid § example:

Bases Properties of bases: § Bitter § Feel Slippery § Electrolytes § Change indicator colors § React with acids to produce salt & water § Produce OH - in water Naming Bases § name the metal and hydroxide

Water Self ionization of water: H 2 O + H 2 O H 3 O + + OH § Hydronium ion H 3 O + § Hydroxide ion OH - Only 1 X 10 – 14 M of water molecules do this so [H 3 O + ][OH - ] = 1 X 10 – 14 M § If [H 3 O + ]=[OH - ] neutral solution § If [H 3 O + ]>[OH - ] acidic solution § If [H 3 O + ]<[OH - ] basic or alkaline solution If the hydroxide concentration is 1 X 10 – 4 M, what is the hydronium ion concentration?
![Formulas [H 3 O + ][OH - ] = 1 X 10 – 1 Formulas [H 3 O + ][OH - ] = 1 X 10 – 1](http://slidetodoc.com/presentation_image_h2/9ff67d4098901b2f8642c2c9c185b165/image-5.jpg)
Formulas [H 3 O + ][OH - ] = 1 X 10 – 1 4 M Use to find one [] given the other [] § Find the hydroxide ion concentration if the hydronium ion concentration is 1 X 10 -5 M. Is this acidic basic or neutral? p. H=-log [H 3 O + ] § Use to find p. H given [H + ] § Find the p. H if the hydronium ion concentration is 2. 1 X 10 -5 M. p. OH= -log [OH - ] § Use to find p. OH given [OH - ] § Find the p. OH if the hydroxide ion concentration is 4. 1 X 10 5 M.

p. H § p. OH 1 -6 Acid 14 -8 7 neutral 7 p. H +p. OH=14 § Use to find one given the other. § Example: Find the p. OH if the p. H is 8. 2. [H + ]= 10 - p H § Use to find [H 3 O + ] given to p. H § Example: Find the [H + ] given a p. H of 11. 5. [OH - ]= 10 - p O H § Use to find [OH - ] given to p. OH § Example: Find the [OH - ] given a p. OH of 7. 5. 8 -14 base 6 -1

Measuring p. H To measure p. H: § Use acid/base indicators: change color based on [hydrogen ion ], many types pg 590 § p. H paper- have indicators on them § p. H meter

3 Types of Acids & Bases Arrhenius acids & bases Bronsted-Lowery acids & bases Lewis acids & bases

3 TYPES OF ACIDS & BASES Arrhenius acid: Produce H + ion Arrhenius base: Produce OH - ion Bronsted-Lowery Acid: H + donor Bronsted Lowery Base: H + acceptor Lewis acid- can accept pair of e- to form a covalent bond (H + ) Lewis base- can donate pair of e- to form a covalent bond (OH - )
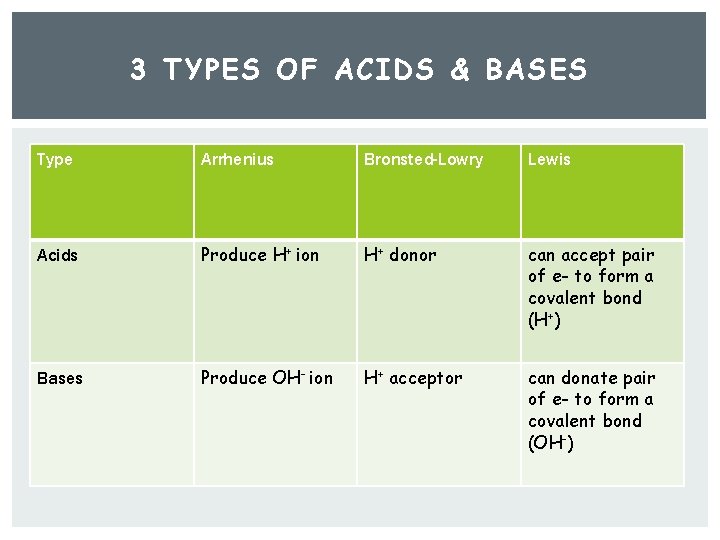
3 TYPES OF ACIDS & BASES Type Arrhenius Bronsted-Lowry Lewis Acids Produce H+ ion H+ donor can accept pair of e- to form a covalent bond (H+) Bases Produce OH- ion H+ acceptor can donate pair of e- to form a covalent bond (OH-)

Conjugate Pairs Be able to identify conjugate acid base pair: § Conjugate base – what is left of acid after hydrogen ion is donated § Conjugate acid – base plus the hydrogen ion that was donated/accepted Amphoteric- can be acid or base Identify the conjugate acid-base pairs in the following reactions. H 2 SO 4 + H 2 O HSO 4 - + H 3 O + HI(aq) + H 2 O(l) H 3 O + (aq) + I - (aq)

ionization Ionization Strong acid- completely ionizes becomes weak CB Weak acid- incompletely ionizes becomes strong CB Can write ionization equation: Can write acid dissociation constant (K a ) K a = [Products]/[Reactants] § Don’t include water § Smaller #=weaker acid § Polyprotic acids- take 1 H at a time Also applies to bases called K b

Example Questions Write the steps in the complete ionization of H 2 S. Write the ionization equation and acid ionization constant expression for the following acids. § a. § b. § c. HCl HNO 2 H 3 PO 4 Write ionization equations and base ionization constant expressions for the following bases. § a. propylamine (C 3 H 7 NH 2 ) Given: 0. 1 M solution of ethanoic acid is partially ionized. The [H + ] is 1. 34 X 10 - 3 M. What is the K a of ethanoic acid?

Neutralization Neutralization- Double displacement reaction § § Acid + Base salt + water Nitric acid & calcium hydroxide Sulfuric acid & aluminum hydroxide Can find acid & base that form salt § Na. Cl § Na. HCO 3

Titration § Titration- Use known to find unknown § Need mol ratio for H + : OH§ Steps in titration: § Measure volume of acid soln. Of [unknown] in flask § Add indicator § Measured volume of base [known] ‘standard’ are added until slight color change (endpoint) § Use MV=MV § A 25 m. L soln of hydrochloric acid neutralized by 18 m. L of 1 M Na. OH. What is the [hydrochloric acid]?

Salts § Made from an acid & base Strong acid + strong base neutral salt solution. Weak acid + strong base basic salt solution. Strong acid + weak base Acidic salt soution. Be able to predict the acid & base that make the salt Buffers- solution where the p. H remains constant when an acid/base are added Can absorb or release H +
- Slides: 16