Acids Bases Acids Bases Acids often behave like

Acids & Bases

Acids & Bases

Acids often behave like acids only when dissolved in water Therefore, acids are often written with subscript (aq) = aquatic = water The chemical formula of an acid usually starts with Hydrogen (H+) Acid: compound that forms H⁺ ions when dissolved in water Two categories for acids: 1) Binary acids: consist of hydrogen and non-metal (e. g. , HCl) 2) Oxyacids: consist of hydrogen, oxygen, and another element (e. g. , H 2 CO 3)
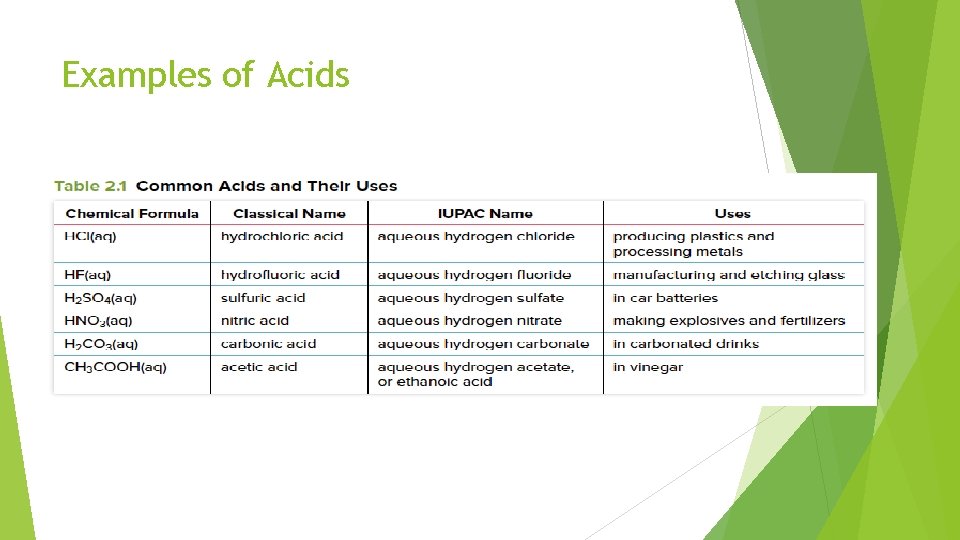
Examples of Acids

Bases Bases, like acids, often behave like bases only when dissolved in water Therefore, bases are often written with subscript (aq) = aquatic = water Also referred to as alkaline since the alkaline earth metals, when reacted with water, produce basic solutions The chemical formula of a base usually ends with hydroxide (-OH) Base: a compound that forms OH⁻ ions when dissolved in water
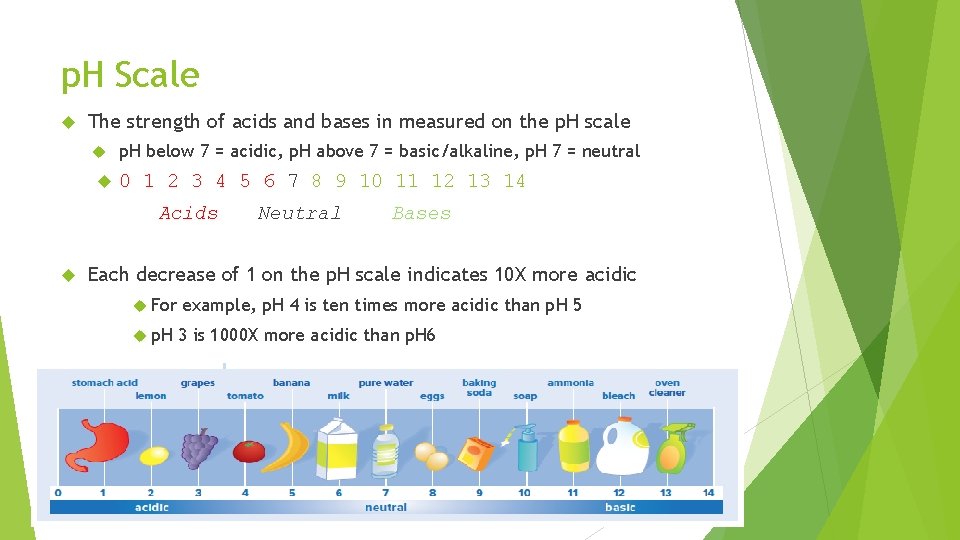
p. H Scale The strength of acids and bases in measured on the p. H scale p. H below 7 = acidic, p. H above 7 = basic/alkaline, p. H 7 = neutral 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Acids Neutral Bases Each decrease of 1 on the p. H scale indicates 10 X more acidic For example, p. H 4 is ten times more acidic than p. H 5 p. H 3 is 1000 X more acidic than p. H 6

p. H Indicators The p. H of acids and bases cannot be determined by sight LITMUS PAPER Two colours: Red VS Blue litmus paper turns red in acids Red litmus paper turns blue in bases UNIVERSAL INDICATOR Contains many indicators that turn different colours at different p. H values (can be in liquid form, or on paper strips like litmus)

p. H Indicators p. H METER uses electrical probes to measure how solutions conduct electricity OTHER INDICATORS Bromothymol blue for p. H 6 - 7. 6 phenolphthalein for p. H 8. 2 - 10 Many natural sources, such as beets and cabbage, are also indicators Most common indicators are in the data booklet

Production of Ions Acids and bases can conduct electricity because they release ions in solution. Acids release hydrogen ions, H+(aq) Bases release hydroxide ions OH–(aq) The p. H of a solution refers to the concentration of ions it has. Square brackets are used to signify concentration, [H+(aq)], [OH–(aq)] High [H+(aq)] = low p. H, very acidic High [OH–(aq)] = high p. H, very basic A solution cannot have BOTH high [H+(aq)] and [OH–(aq)]; they cancel each other out and form water. This process is called neutraliztion.

Neutralization Reaction A chemical reaction in which an acid reacts with a base to form salt and water It is a type of double replacement reaction. e. g. , HCl(aq) + Na. OH(aq) H 2 O(ℓ) + Na. Cl(s)
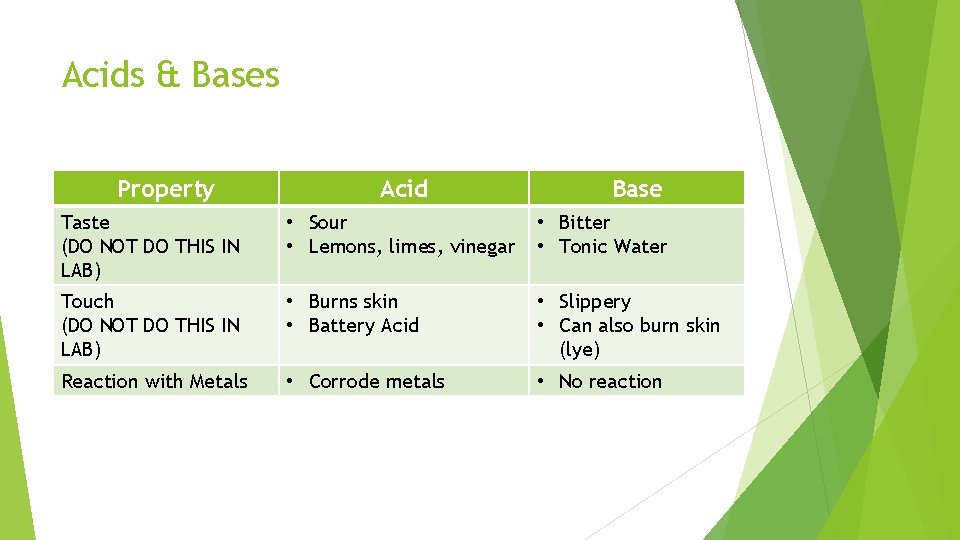
Acids & Bases Property Acid Base Taste (DO NOT DO THIS IN LAB) • Sour • Lemons, limes, vinegar • Bitter • Tonic Water Touch (DO NOT DO THIS IN LAB) • Burns skin • Battery Acid • Slippery • Can also burn skin (lye) Reaction with Metals • Corrode metals • No reaction

Acids & Bases Property Acid Base p. H • Less than 7 • Greater than 7 Conductivity • Conductive Production of Ions • Forms H+ ions in solution • Firms OH- ions in solution
- Slides: 12