Acids and Bases your name Properties Acids Sour

Acids and Bases your name

Properties • Acids: Sour to taste React with some metals to form H 2 gas Turn litmus red Phenolphthalein stays colorless Electrolytes (conduct) Form H+ (H 3 O+): hydronium ions when attached to water molecule your name

• Bases: Bitter to taste Slippery to touch Turn litmus blue Phenolphthalein turns MAGENTA!!!!! Electrolytes Many form OH- (hydroxide ions) in water your name

• Acidic solutions: more H 3 O+ • Basic solutions: more OH • Neutral solutions: [H 3 O+] = [OH-] your name

Definitions of Acids and Bases I. Arrhenius (traditional): Acids: produce H 3 O+ in water Bases: produce OH- in water Ex: HCl(g) H+(aq) + Cl-(aq) Na. OH(s) Na+(aq) + OH-(aq) **** most common definition used in high school Chemistry!!!!! your name

II. Brǿnsted-Lowry: Acids: H+ donor (proton donor) Bases: H+ acceptor (proton acceptor) your name
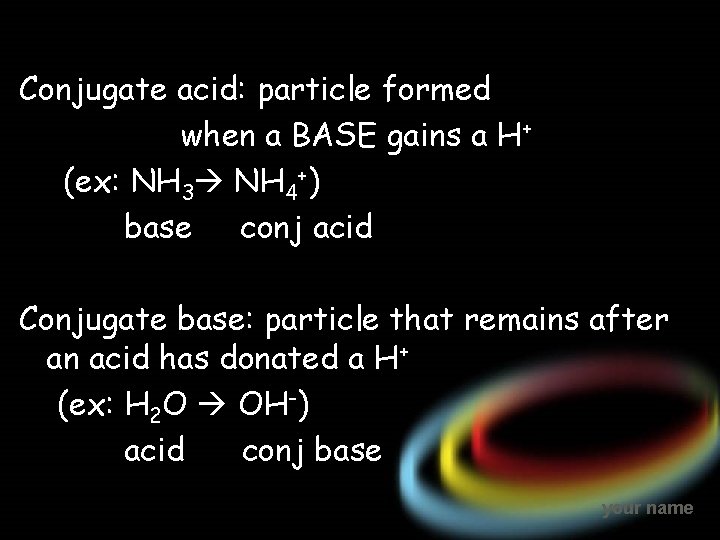
Conjugate acid: particle formed when a BASE gains a H+ (ex: NH 3 NH 4+) base conj acid Conjugate base: particle that remains after an acid has donated a H+ (ex: H 2 O OH-) acid conj base your name

• Conjugate acid-base pair: two substances related by the loss or gain of a single H+……. Always paired with acid and base…… • Ex: NH 3 + H 2 O NH 4+ + OHwhich is which? • Ex: HCl + H 2 O H 3 O+ + Clhow about here? ? ? your name

Types of Acids and Bases I. Strong acids and bases: Strong electrolytes because they ionize 100% in water!!!!!!! • Ex: strong acids: HCl, HNO 3, H 2 SO 4, HCl. O 4, HI, HBr strong bases: (called ionic hydroxides) Na. OH, KOH, Ca(OH)2, Mg(OH)2…. Do you notice anything about the metals involved? ? ? your name

II. Weak acids and bases: weak electrolytes because they only SLIGHTLY ionize in water!!!!!! ex: weak acids: low [H+] H 3 PO 4, HC 2 H 3 O 2, H 2 CO 3 weak bases: low [OH-] NH 3 (NH 4 OH in water) your name

Don’t confuse molarity and strength for acids or bases!!! Molarity: concentration Strength: degree of dissociation Ex: strong acid with low concentration: 0. 5 M HCl weak acid with high concentration: 12. 0 M HC 2 H 3 O 2 your name

• Water is AMPHOTERIC (a switch hitter? ? ? ) can act as either an acid or base depending on what it is coupled with • Monoprotic acids: donate only one H+ (ex: HCl. O 4, HCl, HBr, HNO 3) • Polyprotic acids: donate more than one H+ (ex: H 2 SO 4, H 2 CO 3) **these actually ionize in steps, with each H+ peeling away your name

Naming Acids • Binary acids – H and one other element • Hydro _____ ic acid • HCl • HF • HI hydrochloric acid hydrofluoric acid hydroiodic acid your name

Naming Ternary Acids • Most common (-ate becomes –ic) – H 2 SO 4 – HNO 3 – HCl. O 3 sulfate ion = sulfuric acid nitrate ion = nitric acid chlorate ion= chloric acid • One less oxygen (-ite becomes –ous) – H 2 SO 3 – HNO 2 – HCl. O 2 sulfite ion= sulfurous acid nitrite ion= nitrous acid chlorite ion=chlorous acid • Even less oxygen (add prefix hypo-) – HCl. O hypochlorous acid • More oxygen than most common (add prefix per-) – HCl. O 4 perchloric acid your name

Naming Bases • Name the cation first, followed by hydroxide – Na. OH – Ca(OH)2 sodium hydroxide calcium hydroxide – NH 3 ammonia (this is an exception but you need to know it!) your name
- Slides: 15