Acids and bases vocabulary ACID ACID A solution

Acids and bases vocabulary • ACID: ACID A solution with an excess of H+ ions; Low p. H • BASE: A solution with an excess of OH- ions; High p. H. Another word for base is ALKALI. • NEUTRAL: A solution which has a p. H of 7. It is neither acidic nor basic.

Acids and Bases Vocabulary • STRONG ACID: An acid which has a very low p. H (0 -3). All of the acid becomes ions (completely ionizes) in water. • WEAK ACID: only partially ionizes in an aqueous solution--not every molecule breaks apart. p. H close to 7 (4 -6) • STRONG BASE: A base which has a very high p. H (11 -14). All of the base becomes ions. (completely ionizes) in water. • WEAK BASE: A base that only partially ionizes in water-not every molecule breaks apart. p. H close to 7 (8 -10).

Acids and Bases Vocabulary • Electrolyte: A solution with dissolved ions, Like H+ and OH- conducts electricity • Non-electrolyte: A solution with NO dissolved ions, does not conduct electricity • AQUEOUS: A solution thats solvent is water. • Ionize- to break apart into + and – ions H 2 O breaks apart into H+ and OH-. Most ionic compounds ionize.
Acids and bases • Acids are compounds which break into hydrogen (H+) ions and another compound when placed in an aqueous solution. • Bases are compounds which break up into Hydroxide (OH-) ions and another compound when placed in an aqueous solution. • If you have an IONIC compound and you put it in water it will break apart into two ions. If one of those ions is H+. . . The solution is acidic. If one of the ions is OH-. . . The solution is basic.

p. H scale • • • the "p. H" scale measures how acidic or basic a liquid is The scale goes from "0" to "14“ Acids are found between "0" and "7" Distilled water is 7, Which is neutral Bases are from "7" to "14"

p. H of common substances at right Bases • feel slippery • change litmus blue • neutralizes when mixed with acids • Bitter tasting • high p. H

p. H of common substances at right Acids - taste sour - are corrosive - change litmus red - neutralize when mixed with bases - Low p. H

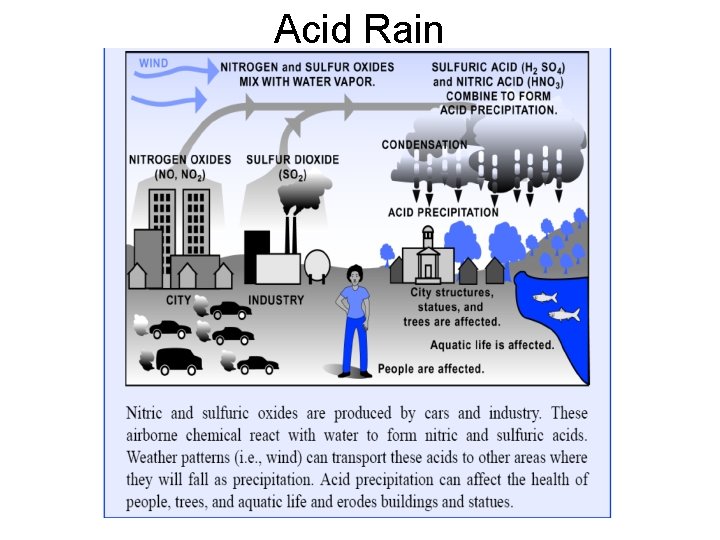
Acid Rain • Rain water is normally slightly acidic, (p. H 5. 6) because it mixes with CO 2 in the atmosphere to form Carbonic acid • However, Acid rain occurs when the p. H dips below 5. 6 • There are four main gases which cause this to happen sulfur dioxide (SO 2), sulfur trioxide (SO 3), nitrogen oxide (NO), and nitrogen dioxide (NO 2)

Acid Rain These gases are produced by: • the burning of coal and oil that contain sulfur • industrial processes such as metal purification. • Nitrogen oxide is a leading by-product of fuel combustion from traffic and power plants.
Acid Rain

Dangers of Acid Rain • Most plants and animals function best when p. H is nearly neutral. • At the extreme ends of the p. H scale, where you find strong acids and strong bases, many life processes will not occur. • For example, fish have trouble reproducing when the p. H of their watery environment is too acidic. • Also, plants have trouble growing if soil is acidified below 5. 1

Dangers of Acid Rain • Also, since acids are very corrosive they tend to damage many kinds of rock, especially limestone and marble

Can you think of any ways that we could prevent acid rain
- Slides: 13