Acids and Bases Types of Ions H ions

Acids and Bases


Types of Ions • H+ ions – Hydrogen Ions • OH- ions – Hydroxide Ions
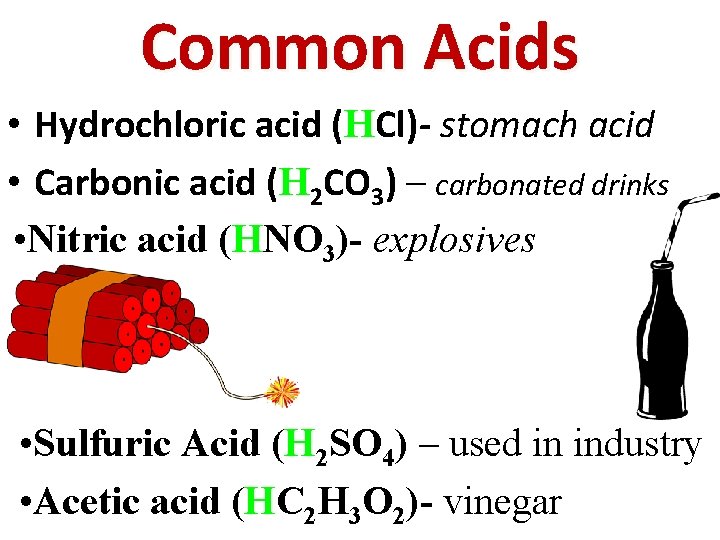
Common Acids • Hydrochloric acid (HCl)- stomach acid • Carbonic acid (H 2 CO 3) – carbonated drinks • Nitric acid (HNO 3)- explosives • Sulfuric Acid (H 2 SO 4) – used in industry • Acetic acid (HC 2 H 3 O 2)- vinegar

Properties of Acids • Taste sour • Produces gas when placed on a metal or metal carbonate • Turns blue litmus paper red • Have more H+ hydrogen than OHhydroxide

Naming Acids all start with an H!!! • Ending of name as an ionic compound ends in: -ide -ite -ate • New name becomes – Hydro-root-ic acid – Root-ous acid – Root-ic acid

Common Bases • Sodium Hydroxide (Na. OH) – soap • Magnesium Hydroxide Mg(OH)2 – antacid • Group 1 A or 2 A cations w/ OH • Ammonia (NH 3)- household cleaners

Properties of Bases • Feels slippery • Tastes bitter • Corrosive • Burns skin • Turns red litmus paper blue • Higher concentration of OHhydroxide than hydrogen H+

Naming Bases • Name the metal followed by hydroxide regardless of how many hydroxides are there.

Practice Naming/Writing Formulas • Mg(OH)2 • H 2 SO 4 • Hydrosulfuric acid • Calcium hydroxide • Aluminum hydroxide • HBr • Li. OH • Phosphoric acid • Sulfurous acid

Strong Acids: • HCl • HBr • HI • H 2 SO 4 • HNO 3 • HCl. O 4 • HCl. O 3 Strong Bases: • Lithium hydroxide • Sodium hydroxide • Potassium hydroxide • Rubidium hydroxide • Cesium hydroxide • Calcium hydroxide • Strontium hydroxide • Barium hydroxide

Strong vs. Weak Acids and Bases • Strong Acid/Base – An acid/base that breaks apart completely when it is placed in water. Are excellent conductors of electricity • Weak Acid/Base – partially ionizes and therefore are poor conductors

Arrhenius Theory • Acids contain hydrogen • Bases contain hydroxide • Neutralization: • Acid + Base Water + Salt • Hacid + Base. OH HOH + Base. Acid • Hydrogen from the acid bonds with hydroxide from base combine to form water. The other 2 ions bond to form a salt (ionic bonds) HNO 3 + Na. OH H 2 O + Na. NO 3

According to Arrhenius Identify Acid or Base • H 2 S • Na. OH • Al(OH)3 • H 2 CO 3

The Amphoterism of Water • Amphoteric: The ability to act as an ACID or BASE depending on what it is around. • While other substances are able to do this as well, WATER is the most common

Self Ionization • When two water molecules combine to form one OH- and one Hydronium atom H 3 O+

Bronsted Lowry Theory Sheep Theory • An acid is a proton donor • A base is a proton acceptor • Conjugate Acid – Base Pairs – A conjugate acid of the base → Base + H+ – A conjugate base of the acid → Acid − H+

According to Bronsted Lowry Determine Acid, Base, Conjugate Acid and Conjugate Base

Kw: Water Constant • Value of the equilibrium constant expression for the self ionization of water • Kw = [H+] x [OH-] • If it is water H+ and OH- will be equal so if H = 1. 0 x 10 -7 then OH = 1. 0 x 10 -7 making water 1. 0 x 10 -14 • Kw = 1. 0 x 10 -14

Kw: Water Constant • If H+ is larger then it is an acid • If OH- is larger then it is a base H+ OH- 4. 7 x 10 -5 7. 0 x 10 -7 2. 3 x 10 -12 1. 0 x 10 -7 A/B

What is a log? A logarithm (log) is the exponent to which a power of 10 must be raised to get the number. -log 1 x 10 -3 log = 3

p. H • The negative logarithm of the hydrogen ion concentration p. H = -log[H+] • p. H can not be less than 0 nor greater than 14 • If it is below 7 it is an acid, above 7 a base, and 7 is neutral
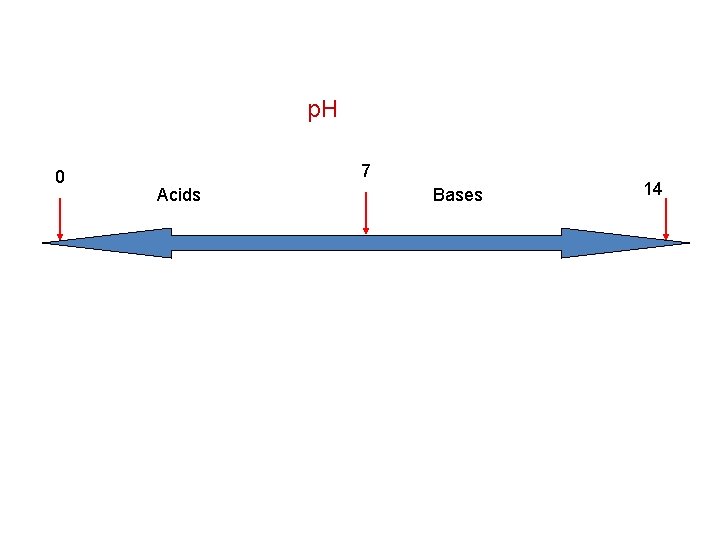
p. H 0 7 Acids Bases 14
![Ion Concentrations from p. H • Antilog (-p. H) = [H+] Ion Concentrations from p. H • Antilog (-p. H) = [H+]](http://slidetodoc.com/presentation_image_h/2e4954e2600d7af3182c0ca69874d2e9/image-24.jpg)
Ion Concentrations from p. H • Antilog (-p. H) = [H+]
![p. OH • Negative logarithm of the hydroxide ion concentration p. OH = -log[OH-] p. OH • Negative logarithm of the hydroxide ion concentration p. OH = -log[OH-]](http://slidetodoc.com/presentation_image_h/2e4954e2600d7af3182c0ca69874d2e9/image-25.jpg)
p. OH • Negative logarithm of the hydroxide ion concentration p. OH = -log[OH-] • p. OH less than 7 are basic, greater than 7 are acidic, and 7 is neutral
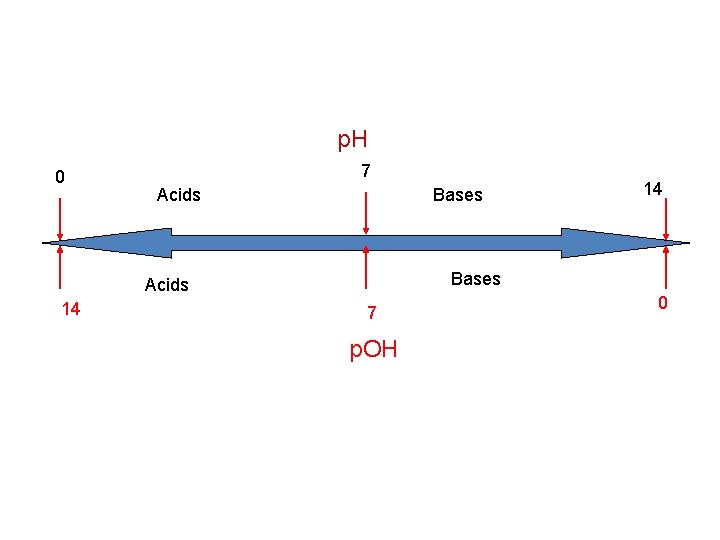
p. H 0 7 Acids Bases Acids 14 14 7 p. OH 0
![Ion Concentration from p. OH • Antilog (-p. OH) = [OH-] Ion Concentration from p. OH • Antilog (-p. OH) = [OH-]](http://slidetodoc.com/presentation_image_h/2e4954e2600d7af3182c0ca69874d2e9/image-27.jpg)
Ion Concentration from p. OH • Antilog (-p. OH) = [OH-]
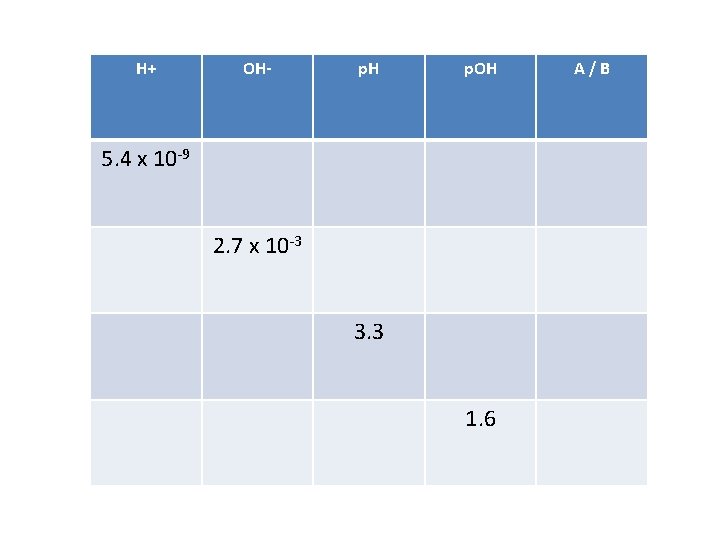
H+ OH- p. H p. OH 5. 4 x 10 -9 2. 7 x 10 -3 3. 3 1. 6 A/B

Titration • Using a KNOWN concentration to figure out an UNKNOWN concentration of an acid or base
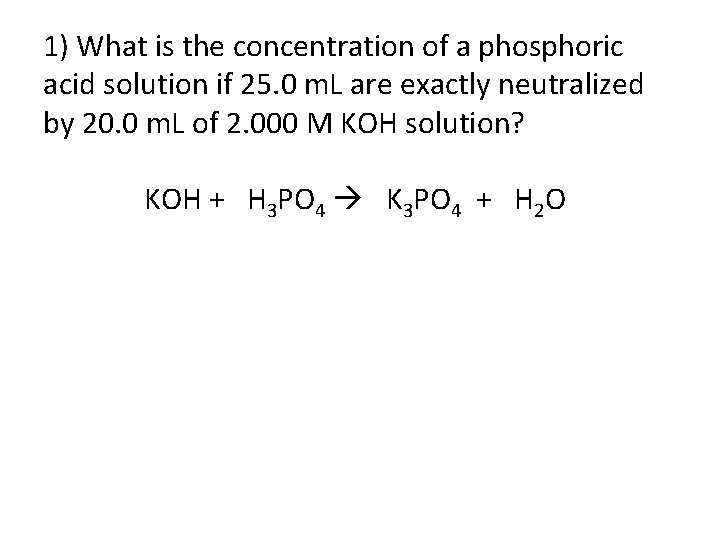
1) What is the concentration of a phosphoric acid solution if 25. 0 m. L are exactly neutralized by 20. 0 m. L of 2. 000 M KOH solution? KOH + H 3 PO 4 K 3 PO 4 + H 2 O

2) How many m. L of a 0. 1 M calcium hydroxide solution are needed to completely neutralize 10. 00 m. L of 0. 250 M HCl solution? Ca(OH)2 + HCl Ca. Cl 2 + H 2 O

RICE diagram • RICE diagrams are needed when asked to find p. H or p. OH • Reaction • Initial • Change • Excess
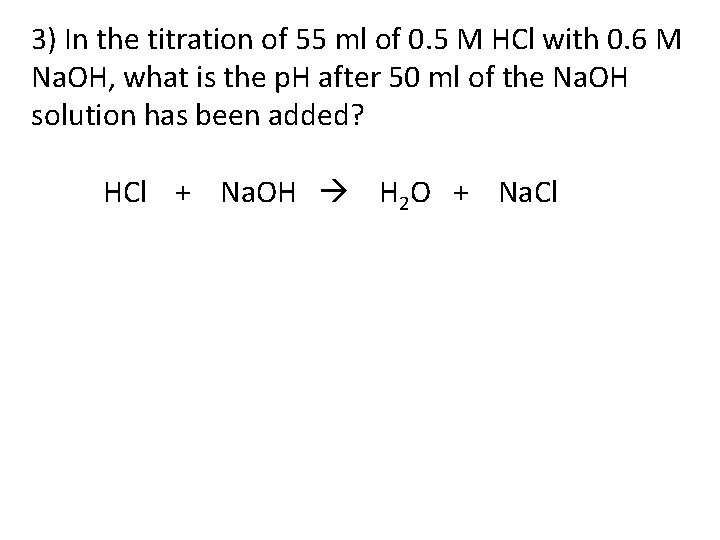
3) In the titration of 55 ml of 0. 5 M HCl with 0. 6 M Na. OH, what is the p. H after 50 ml of the Na. OH solution has been added? HCl + Na. OH H 2 O + Na. Cl
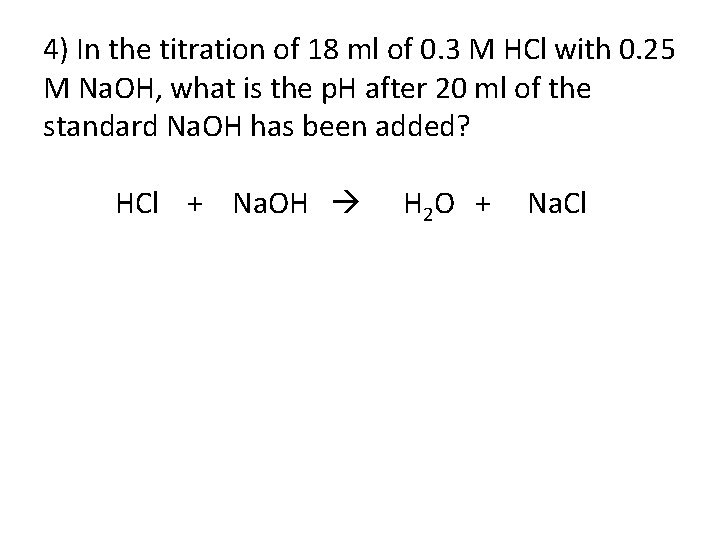
4) In the titration of 18 ml of 0. 3 M HCl with 0. 25 M Na. OH, what is the p. H after 20 ml of the standard Na. OH has been added? HCl + Na. OH H 2 O + Na. Cl
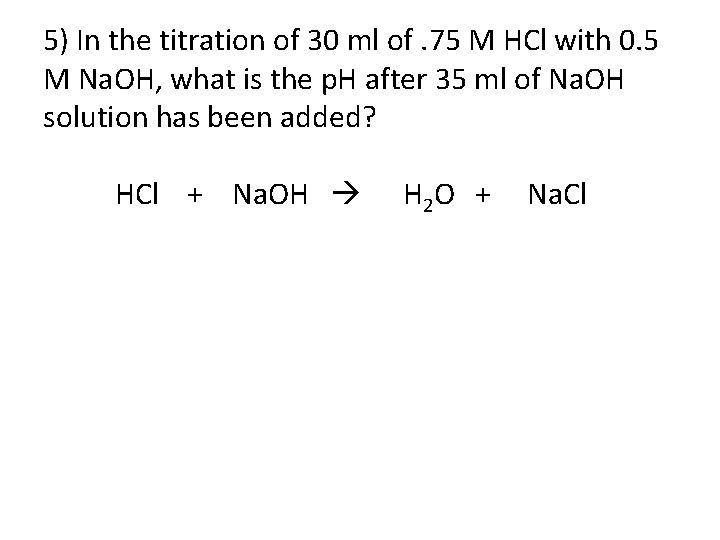
5) In the titration of 30 ml of. 75 M HCl with 0. 5 M Na. OH, what is the p. H after 35 ml of Na. OH solution has been added? HCl + Na. OH H 2 O + Na. Cl

Vocabulary to Add • Amphoteric: substance that can act as an acid or base (Water most common example) • Saponification: mixing a base and fat to make soap • Indicator: any substance that gives a visible sign, usually by a color change, of the presence or absence of a threshold concentration of an acid or base solution

Electrochemistry OIL RIG Oxidation is loss (of e-) Reduction is gain (of e-)

LEO the lion goes GER • Lose electrons = Oxidation • Gain electrons = Reduction

Balancing Acid Redox Reactions • Split the reaction into two halves • Balance the common element on the two sides. • Balance the Oxygen by adding water to the opposing side • Balance the hydrogen by adding H+ ions to the opposing side • Balance the charge by adding electrons

Redox Reactions in Acid • Al + Mn. O 4 -1 Al 3+ + Mn 2+
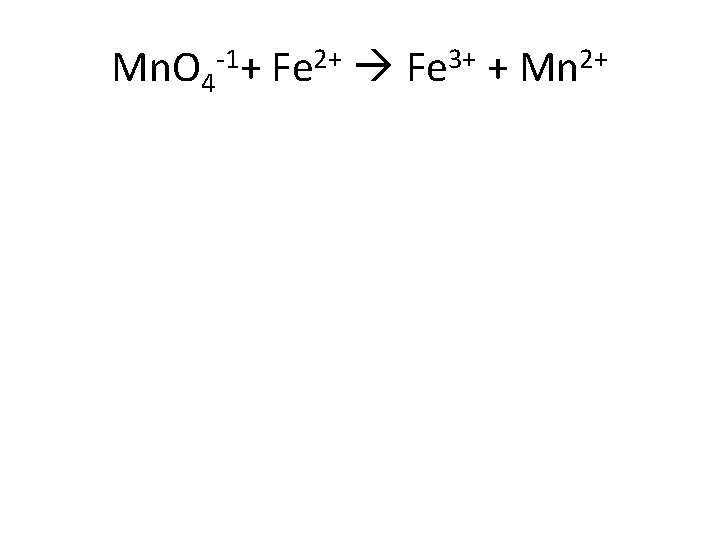
Mn. O 4 -1+ Fe 2+ Fe 3+ + Mn 2+
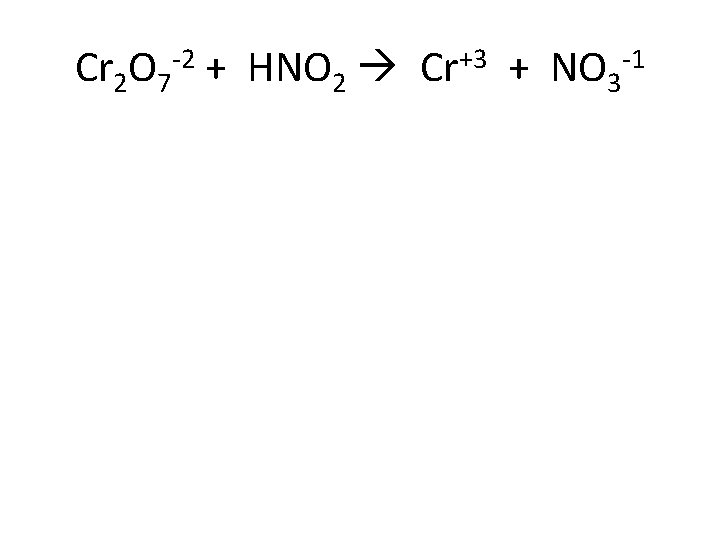
Cr 2 O 7 -2 + HNO 2 Cr+3 + NO 3 -1
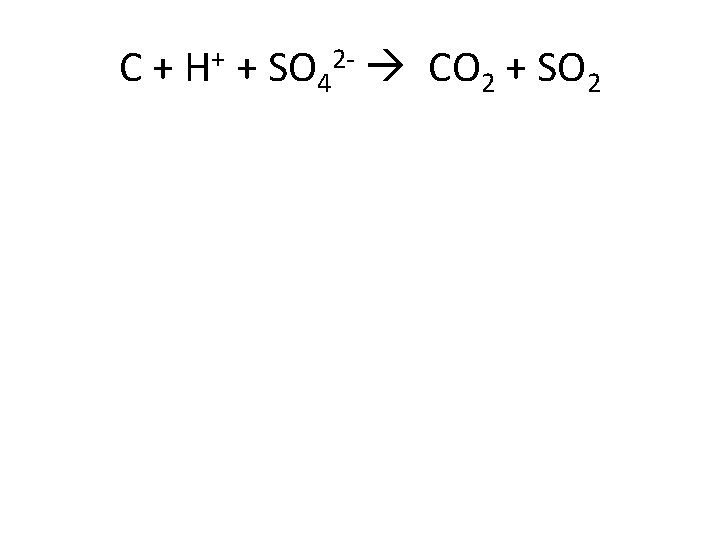
C + H+ + SO 42 - CO 2 + SO 2

Balancing Basic Redox Reactions • Split into two half reactions • Balance the common ion • Balance the charge of the common ion by using electrons • Balance the overall charge by using OH • Balance the hydrogen by adding H 2 O

Basic Redox • Calculate the charge of P in PO 4 -3 • Calculate the charge of Cr in Cr 2 O 7 -2

Redox Reactions in Basic Solutions • Mg + OCl- Mg(OH)2 + Cl-
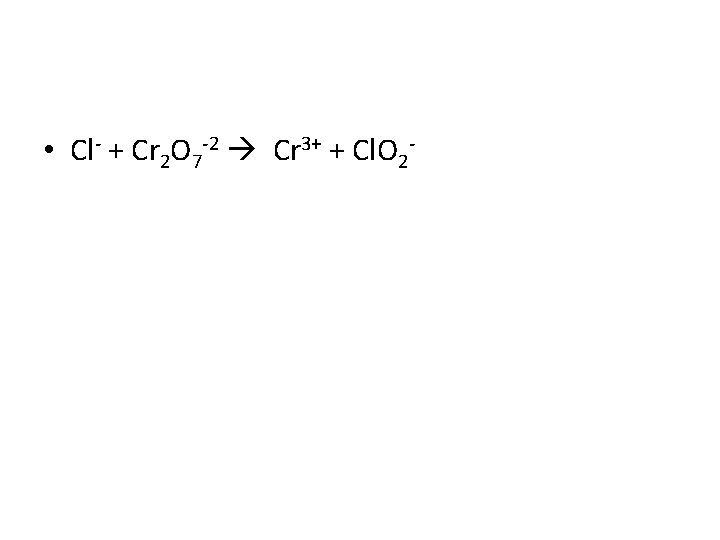
• Cl- + Cr 2 O 7 -2 Cr 3+ + Cl. O 2 -
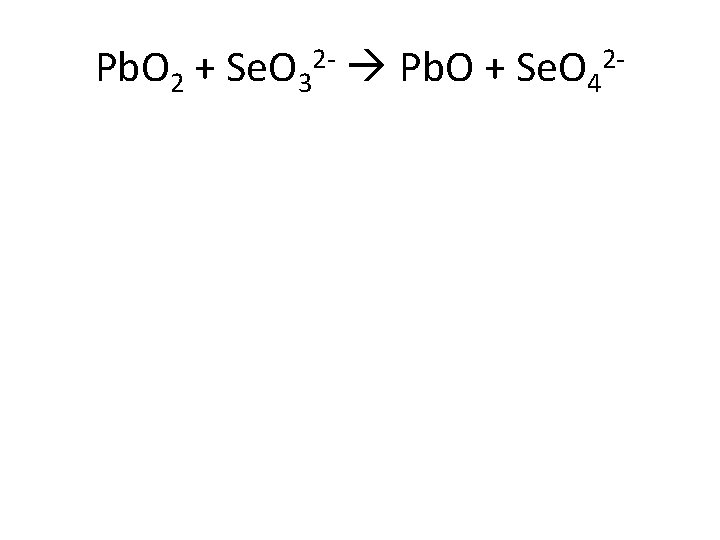
Pb. O 2 + Se. O 32 - Pb. O + Se. O 42 -
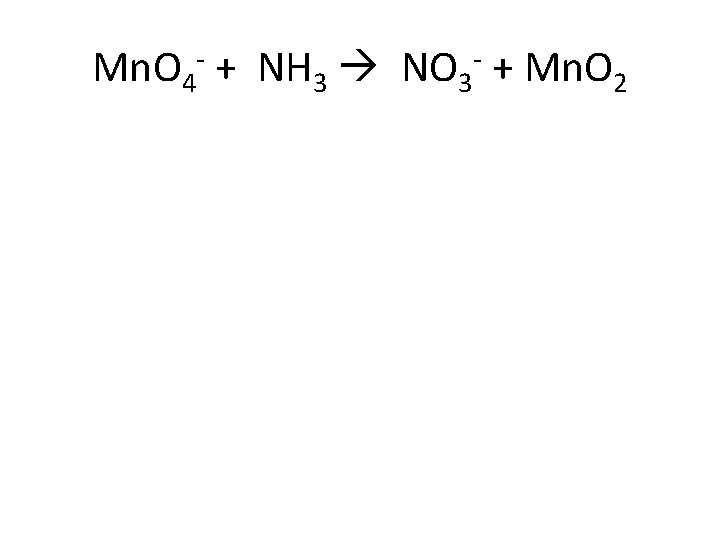
Mn. O 4 - + NH 3 NO 3 - + Mn. O 2
- Slides: 50