Acids and Bases Topics to be covered Definitions

Acids and Bases Topics to be covered: � Definitions of acids and bases; � Bronsted’s conjugate acid-base pairs concept; � Definition of strong vs. weak acid/base
![Acids and Bases � Arrhenius definition: Acid: substance that increases [H 3 O+] in Acids and Bases � Arrhenius definition: Acid: substance that increases [H 3 O+] in](http://slidetodoc.com/presentation_image_h2/0defc6377c8a460063a2b4ed4093a9bd/image-2.jpg)
Acids and Bases � Arrhenius definition: Acid: substance that increases [H 3 O+] in solution; Base: substance that increases [OH-] in solution; � Br nsted-Lowry definition: Acid: reactant that donates proton in a reaction; Base: reactant that accepts proton in a reaction;

Strong and Weak Acids � Strong acids ionize completely in aqueous solution: ○ HCl(aq) + H 2 O H 3 O+(aq) + Cl-(aq); ○ H 2 SO 4(aq) + H 2 O H 3 O+(aq) + HSO 4 -(aq); � Weak acids ionize only partially in aqueous solution: ⇌ H 3 O+(aq) + F-(aq); H 2 O ⇌ H 3 O+(aq) + Cl. O-(aq). ○ HF(aq) + H 2 O ○ HOCl(aq) +
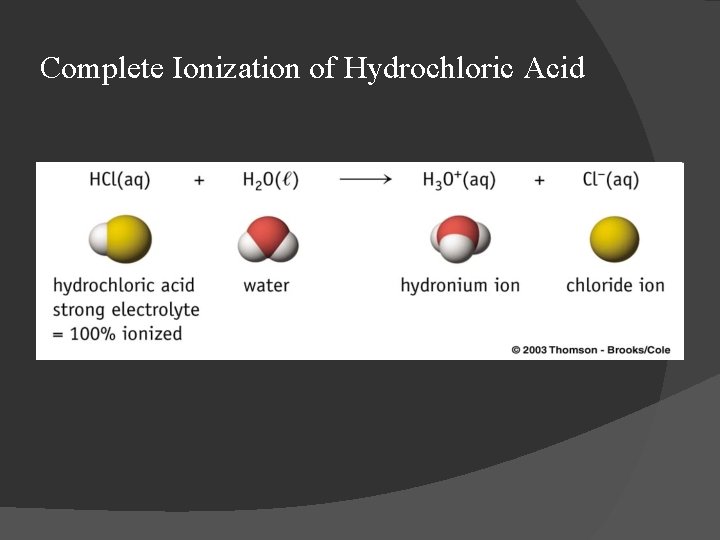
Complete Ionization of Hydrochloric Acid
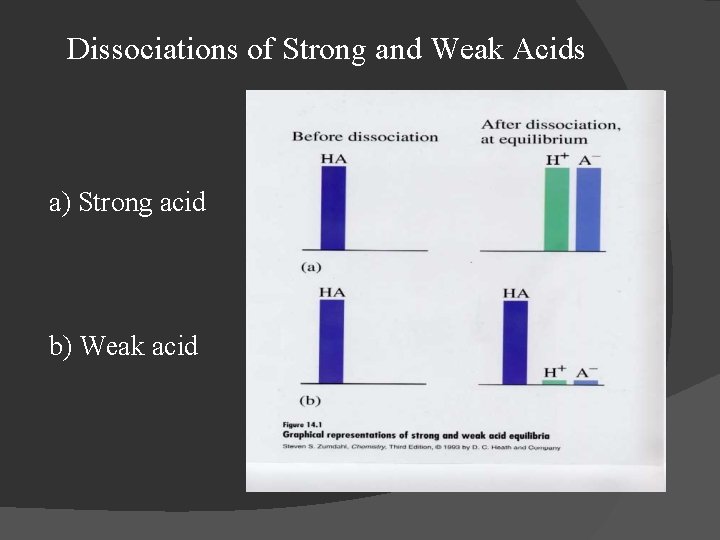
Dissociations of Strong and Weak Acids a) Strong acid b) Weak acid

Strong and Weak Bases � Strong bases ionize completely in aqueous solution: Na. OH(aq) Na+(aq) + OH-(aq); Ba(OH)2(aq) Ba 2+(aq) + 2 OH-(aq); � Weak bases ionize only partially in aqueous solution: NH 3(aq) + H 2 O NH 4+(aq) + OH-(aq); PO 43 -(aq) + H 2 O HPO 42 -(aq) + OH-(aq)

Brønsted-Lowry: Conjugate Acids & Bases Let “HA” represent hydrogen bonded to a random negative ion, A � Consider the following equilibrium: HA + B ⇌ BH+ + AAcid Base Conjugate acid base � A- is the conjugate base to acid HA; HA & A- are conjugate acid-base pair; � BH+ is the conjugate acid to base B; BH+ & B are also conjugate acid-base pair.
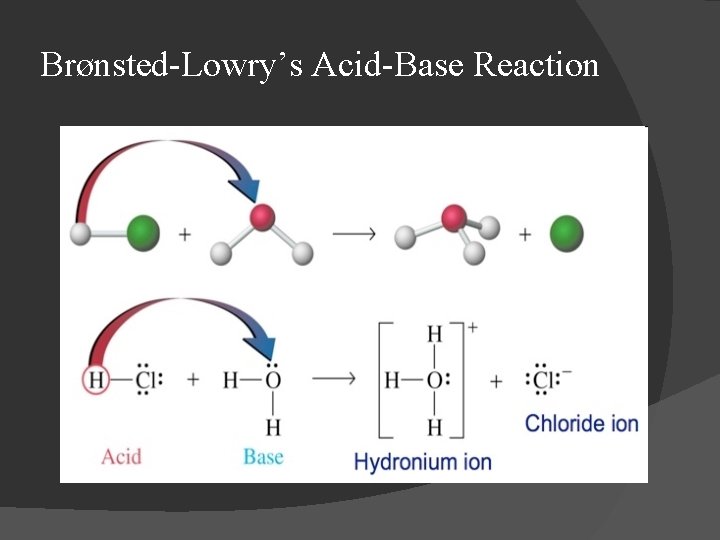
Brønsted-Lowry’s Acid-Base Reaction
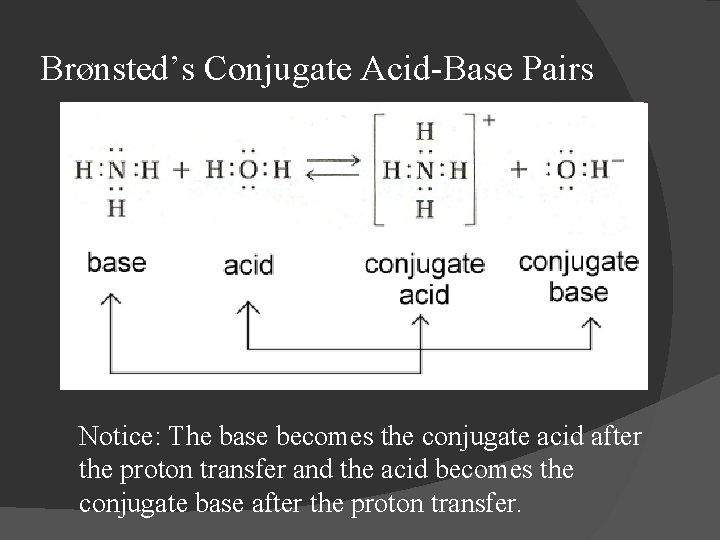
Brønsted’s Conjugate Acid-Base Pairs Notice: The base becomes the conjugate acid after the proton transfer and the acid becomes the conjugate base after the proton transfer.

Examples of Conjugate Acid-Base Pairs HNO 3 – NO 3 H 2 SO 4 – HSO 4 H 3 O+ – H 2 O HF – FH 3 PO 4 – H 2 PO 4 CH 3 COOH – CH 3 COOH 2 PO 4 - – HPO 42 NH 4+ – NH 3 H 2 O – OH-

Exercise #1: Conjugate Acids & Bases Write the formulas of the conjugate bases for the following acids: (a) HF (b) HSO 4(c) H 2 CO 3 (d) HPO 42 -

Exercise #1: Conjugate Acids & Bases Answers: (a) F(b) SO 42(c) HCO 3 (d) PO 43 -

Exercise #2: Conjugate Acids and Bases Write the formulas of the conjugate acids for the following bases: (a) NH 3 (b) CO 32(c) C 5 H 5 N (d) HPO 32 -

Exercise #2: Conjugate Acids and Bases Answers (a) NH 4+ (b) HCO 3(c) HC 5 H 5 N+ (d) H 2 PO 3 -

Acid Strength and Ionization Constants Identify the acid, base, conjugate acid, and conjugate base in each of the following reactions and identify the conjugate acid base pairs. (a) HSO 4 - + NH 3 ↔ (b) SCN- + HF ↔ NH 4+ + SO 42 - HSCN + F-
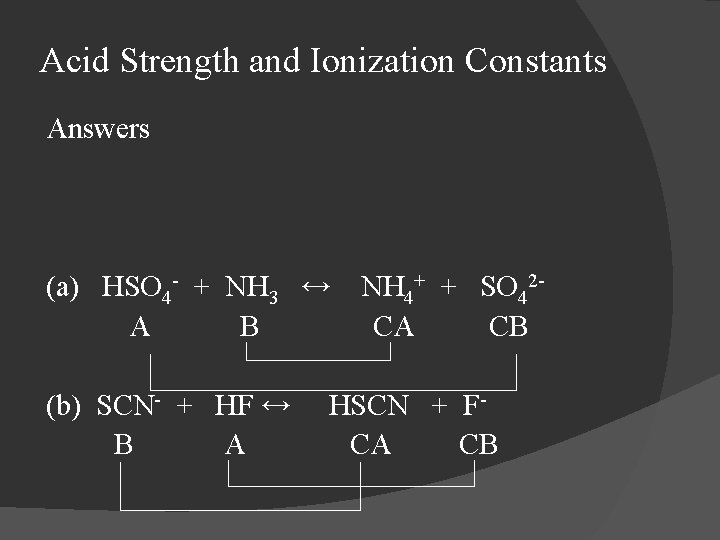
Acid Strength and Ionization Constants Answers (a) HSO 4 - + NH 3 ↔ A B (b) SCN- + HF ↔ B A NH 4+ + SO 42 CA CB HSCN + FCA CB

Relative Strength of Acids and Their Conjugate Bases Acids Conjugate Bases Very Strong Very Weak Strong Very Weak Very Strong _______________ � Strong acids lose protons very readily weak conjugate bases; � Weak acids do not lose protons very readily strong conjugate bases.

Relative Strength of Acids and Their Conjugate Bases

Acid-Base Properties of Water � Water auto-ionizes (ionizes with itself) to produce both H 3 O+ and OH, thus it has both acid and base properties. � Auto-ionization of water: 2 H 2 O H 3 O + (aq) - + OH (aq)

Polyprotic Acids � Acids with more than one ionizable hydrogen ion are called polyprotic acids. Examples: H 2 SO 3, H 2 SO 4, H 3 PO 4, H 3 C 6 H 5 O 7, etc. � Their hydrogen atoms ionize in stages. For example, H 3 PO 4(aq) + H 2 O ⇄ H 3 O+(aq) + H 2 PO 4 -(aq); H 2 PO 4 -(aq) + H 2 O ⇄ H 3 O+(aq) + HPO 42 -(aq); HPO 42 -(aq) + H 2 O ⇄ H 3 O+(aq) + PO 43 -(aq); Acid strength decreases in the order: H 3 PO 4 >> H 2 PO 4 - >> HPO 42 -;

Work time! � Finish part 2, part 6, and part 7 of the “Acid-Base Homework Packet” given to you last Tuesday. The whole packet will be due next Monday. Don’t forget that your Titration lab is due tomorrow.
- Slides: 21