Acids and Bases Titration Curves Strong AcidBase Titration

Acids and Bases Titration Curves
![Strong Acid-Base Titration Plotting Points 50. 0 ml of [0. 200] HNO 3 is Strong Acid-Base Titration Plotting Points 50. 0 ml of [0. 200] HNO 3 is](http://slidetodoc.com/presentation_image_h2/974172558f9abf4503885f741f4ee60b/image-2.jpg)
Strong Acid-Base Titration Plotting Points 50. 0 ml of [0. 200] HNO 3 is titrated with 0. 100 M Na. OH. Calculate the p. H points a. Na. OH has NOT been added. Eq: HNO 3 + Na. OH Na. Cl + Na. NO 3 Since HNO 3 is a strong acid p. H = 0. 70 b. 10. 0 ml of 0. 100 M Na. OH has been added. (or n=CV and 0. 0010 moles) Acid moles in flask = C V = [0. 200] x 0. 0500 L = 0. 0100 mol to START H+ left over = 0. 0100 mol Acid – 0. 0010 mol OH- added = 0. 0090 mol Acid Left C Acid left = 0. 0090 mol H+/ 0. 060 ml = 0. 15 M Acid left so p. H=0. 82 c. Now another 10 ml base added for total of 20. 0 m. L of 0. 100 M Na. OH H+left over = 0. 0100 mol – 0. 0020 mol of OH- added = 0. 0080 mol Acid Left C Acid left = 0. 0080 mol H+/ 0. 070 ml total volume = 0. 114 M Acid left so p. H=0. 942 • 50. 0 m. L of 0. 100 M Na. OH has been added (half way). H+ = 0. 0100 mol – 0. 0050 mol of OH- added = 0. 0050 mol Acid left C Acid left = 0. 0050 mol H+/ 0. 100 ml = 0. 0500 M Acid left p. H=1. 13

Strong Acid (titrant) - Strong Base Titration (analyte) e. After 150. 0 m. L of 0. 100 M Na. OH has been added, Excess Base p. H = 12. 40 Center vertical region, Graph Midpoint = equivalence point, or look at products

Important points: – p. H increases slowly far from the equivalence point – p. H changes quickly near the equivalence point – The equivalence point of a strong acid—strong base titration = 7. 00 The titration of a strong base with a strong acid is almost identical, just reversed

Acid-base indicators Must change immediately in the required p. H range over the addition of ‘half’ a drop of reagent. COLOUR CHANGES OF SOME COMMON INDICATORS p. H METHYL ORANGE LITMUS PHENOLPHTHALEIN 1 2 3 4 5 6 7 8 9 10 CHANGE 11 12 13 14

Titration Curves A titration curve is a graph of the p. H changes that occur during an acid -base titration versus the volume of acid or base added. You need to be able to recognize each and then choose a suitable indicator for that titration, by looking at the PRODUCTS or at the midpoint of the titration curves steepest section Also at that very same point is the equivalence point, and the end of a titration where the stoichiometry exactly satisfied, or moles H+ = moles OH-. It is also called the transition point or end point referring to where the indicator changes color and [HInd] = [Ind-].

Choosing an Indicator The p. H of the equivalence point depends on type of salt in PRODUCT. Rule of thumb: Not exact p. H’s but good approximations If the salt in PRODUCT is neutral the equivalence point = HCl + K OH → K Cl + HOH 7 If the salt in PRODUCT is basic the equivalence point CH 3 COOH + K OH - H 2 O + K CH 3 COO - = 9 If the salt in PRODUCT is acidic the equivalence point HCl + NH 3 → NH 4+ + Cl- = 5
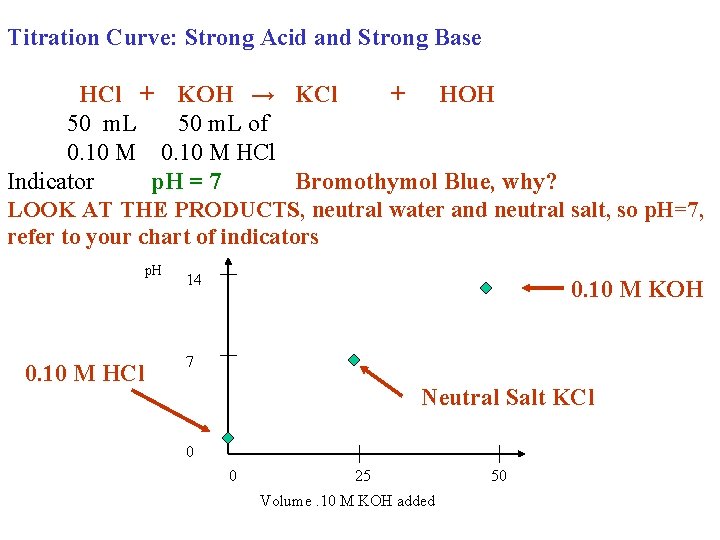
Titration Curve: Strong Acid and Strong Base HCl + KOH → KCl + HOH 50 m. L of 0. 10 M HCl Indicator p. H = 7 Bromothymol Blue, why? LOOK AT THE PRODUCTS, neutral water and neutral salt, so p. H=7, refer to your chart of indicators p. H 0. 10 M HCl 14 0. 10 M KOH 7 Neutral Salt KCl 0 0 25 Volume. 10 M KOH added 50
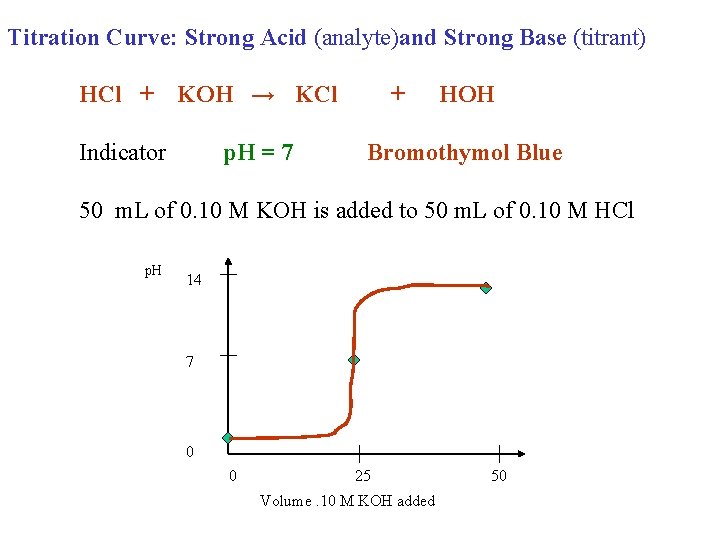
Titration Curve: Strong Acid (analyte)and Strong Base (titrant) HCl + KOH → KCl Indicator p. H = 7 + HOH Bromothymol Blue 50 m. L of 0. 10 M KOH is added to 50 m. L of 0. 10 M HCl p. H 14 7 0 0 25 Volume. 10 M KOH added 50

NEXT Strong Base into a Weak Acid

Strong Base (titrant) into a Weak Acid (analyte) What is the end point and why? CH 3 COOH + OH - H 2 O + CH 3 COO Indicator p. H = 9 Phenolphthalein, why? Weak base CH 3 COOH + OH - H 2 O + CH 3 COO Why is the p. H higher at the start? It is due to the weak acid. Why? CH 3 COOH + OH - H 2 O + CH 3 COO 0. 100 M of this weak acid is p. H = 2. 88
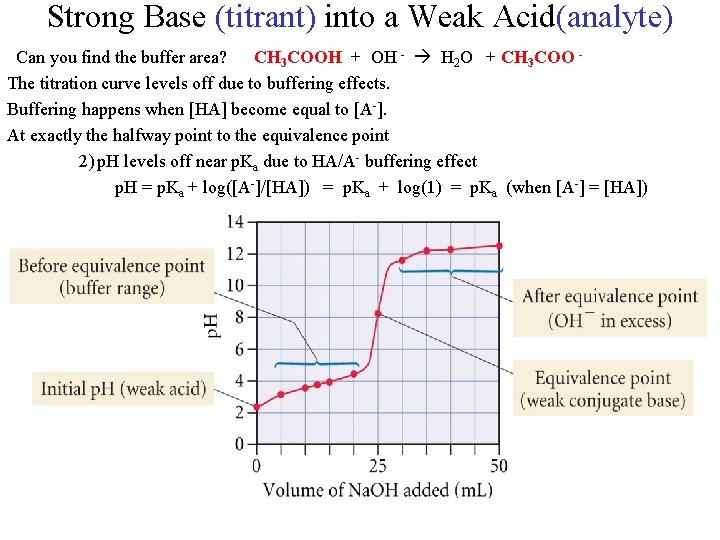
Strong Base (titrant) into a Weak Acid(analyte) Can you find the buffer area? CH 3 COOH + OH - H 2 O + CH 3 COO The titration curve levels off due to buffering effects. Buffering happens when [HA] become equal to [A-]. At exactly the halfway point to the equivalence point 2) p. H levels off near p. Ka due to HA/A- buffering effect p. H = p. Ka + log([A-]/[HA]) = p. Ka + log(1) = p. Ka (when [A-] = [HA])

NEXT Weak Acid into a Strong Base
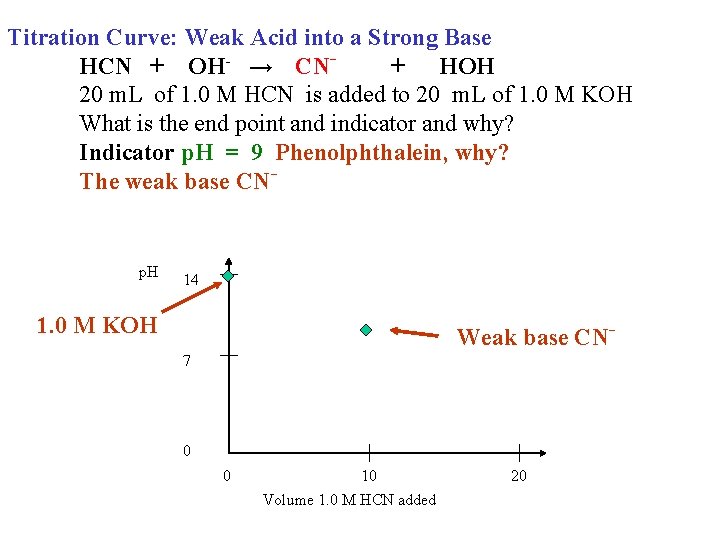
Titration Curve: Weak Acid into a Strong Base HCN + OH- → CN+ HOH 20 m. L of 1. 0 M HCN is added to 20 m. L of 1. 0 M KOH What is the end point and indicator and why? Indicator p. H = 9 Phenolphthalein, why? The weak base CN- p. H 14 1. 0 M KOH Weak base CN 7 0 0 10 Volume 1. 0 M HCN added 20
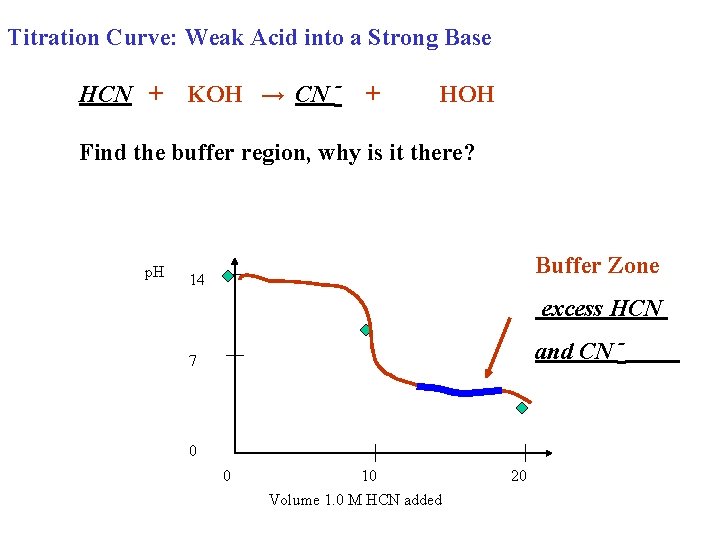
Titration Curve: Weak Acid into a Strong Base HCN + KOH → CN - + HOH Find the buffer region, why is it there? p. H Buffer Zone 14 excess HCN and CN - 7 0 0 10 Volume 1. 0 M HCN added 20

NEXT Weak BASE into a Strong Acid
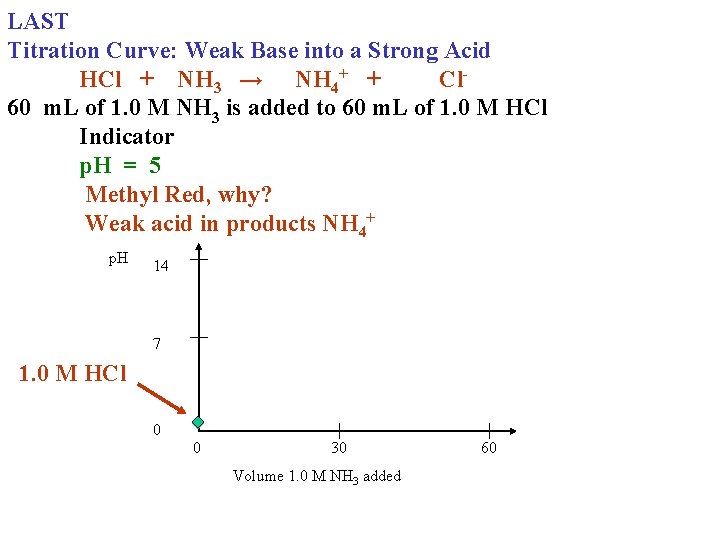
LAST Titration Curve: Weak Base into a Strong Acid HCl + NH 3 → NH 4+ + Cl 60 m. L of 1. 0 M NH 3 is added to 60 m. L of 1. 0 M HCl Indicator p. H = 5 Methyl Red, why? Weak acid in products NH 4+ p. H 14 7 1. 0 M HCl 0 0 30 Volume 1. 0 M NH 3 added 60
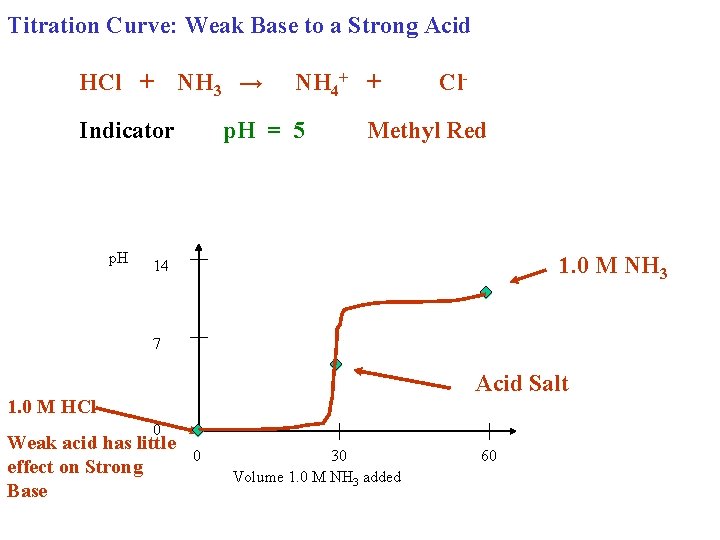
Titration Curve: Weak Base to a Strong Acid HCl + NH 3 → Indicator p. H NH 4+ + p. H = 5 Cl- Methyl Red 1. 0 M NH 3 14 7 Acid Salt 1. 0 M HCl 0 Weak acid has little effect on Strong Base 0 30 Volume 1. 0 M NH 3 added 60
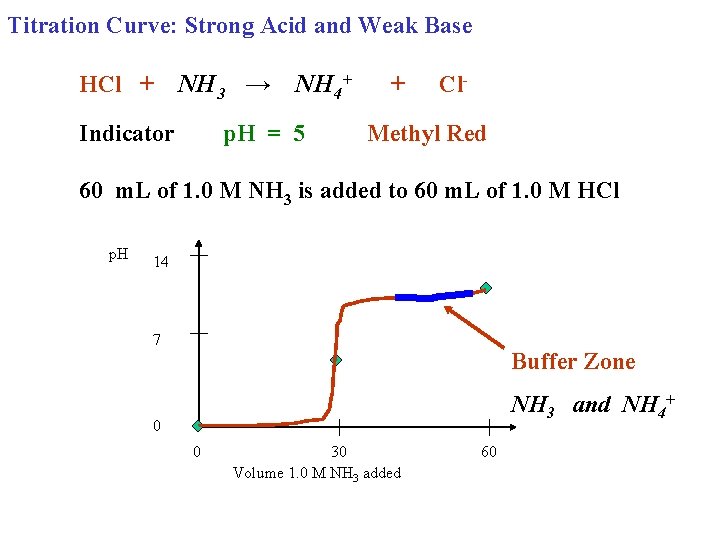
Titration Curve: Strong Acid and Weak Base HCl + NH 3 → NH 4+ Indicator p. H = 5 + Cl- Methyl Red 60 m. L of 1. 0 M NH 3 is added to 60 m. L of 1. 0 M HCl p. H 14 7 Buffer Zone NH 3 and NH 4+ 0 0 30 Volume 1. 0 M NH 3 added 60
Next Weak with Weak Non-Stoichiometric
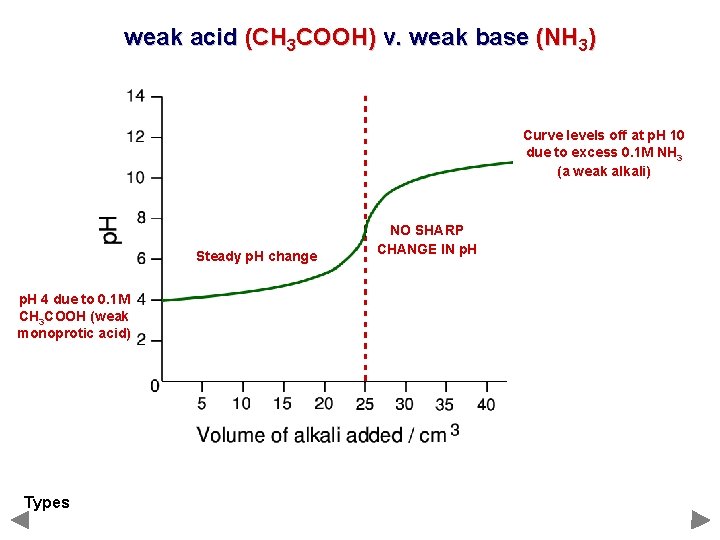
weak acid (CH 3 COOH) v. weak base (NH 3) Curve levels off at p. H 10 due to excess 0. 1 M NH 3 (a weak alkali) Steady p. H change p. H 4 due to 0. 1 M CH 3 COOH (weak monoprotic acid) Types NO SHARP CHANGE IN p. H

weak acid (CH 3 COOH) v. weak base (NH 3) PHENOLPHTHALEIN LITMUS METHYL ORANGE NOTHING SUITABLE There is no suitable indicator- none change in the ‘vertical’ portion. The end point can be detected by plotting a curve using a p. H meter.

POLYPROTIC ACID AND BASE TITRATION CURVES
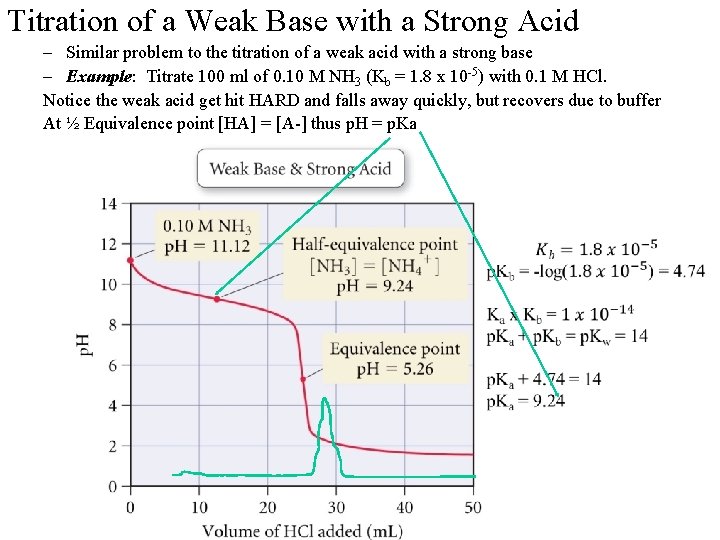
Titration of a Weak Base with a Strong Acid – Similar problem to the titration of a weak acid with a strong base – Example: Titrate 100 ml of 0. 10 M NH 3 (Kb = 1. 8 x 10 -5) with 0. 1 M HCl. Notice the weak acid get hit HARD and falls away quickly, but recovers due to buffer At ½ Equivalence point [HA] = [A-] thus p. H = p. Ka
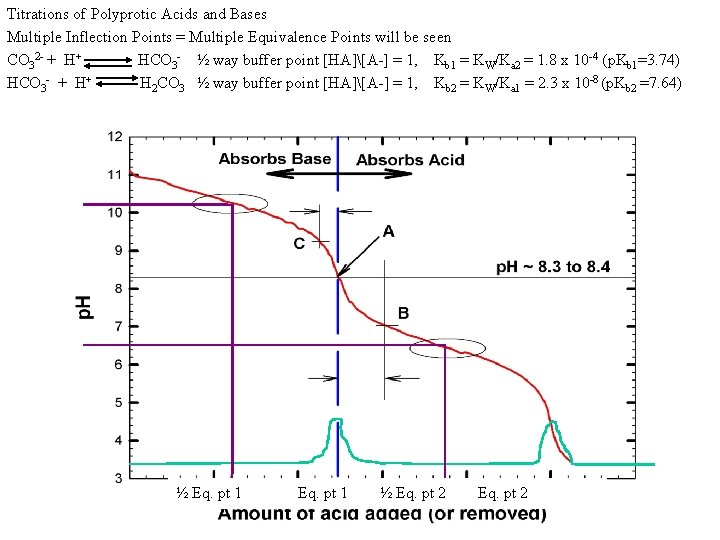
Titrations of Polyprotic Acids and Bases Multiple Inflection Points = Multiple Equivalence Points will be seen CO 32 - + H+ HCO 3 - ½ way buffer point [HA][A-] = 1, Kb 1 = KW/Ka 2 = 1. 8 x 10 -4 (p. Kb 1=3. 74) HCO 3 - + H+ H 2 CO 3 ½ way buffer point [HA][A-] = 1, Kb 2 = KW/Ka 1 = 2. 3 x 10 -8 (p. Kb 2 =7. 64) p. Ka 2 = 10. 26 (p. Kb 1 = 3. 74) p. Ka 1 = 6. 36 p. Kb 2 = 7. 64 ½ Eq. pt 1 ½ Eq. pt 2

Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O
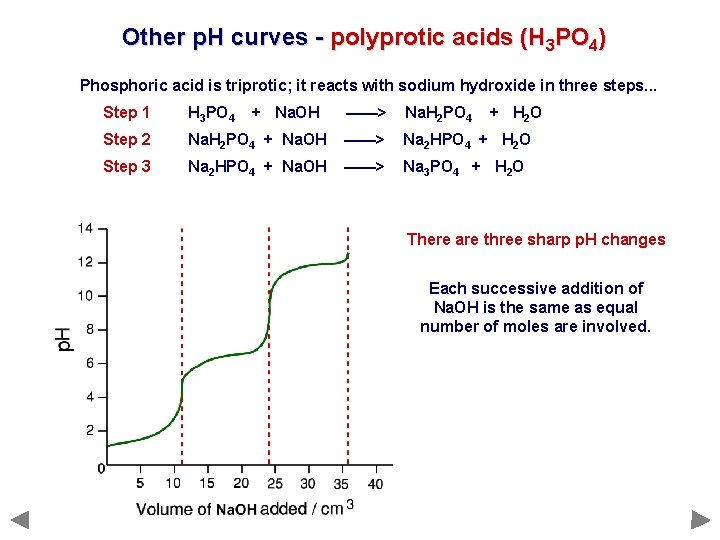
Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O There are three sharp p. H changes Each successive addition of Na. OH is the same as equal number of moles are involved.
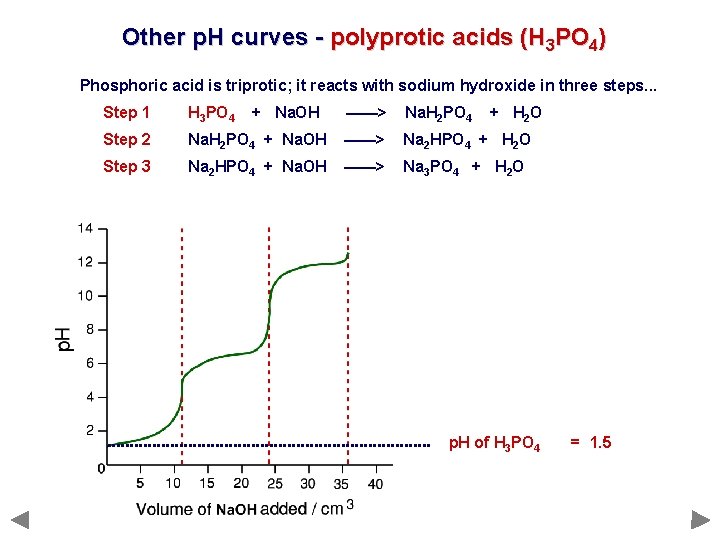
Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O p. H of H 3 PO 4 = 1. 5
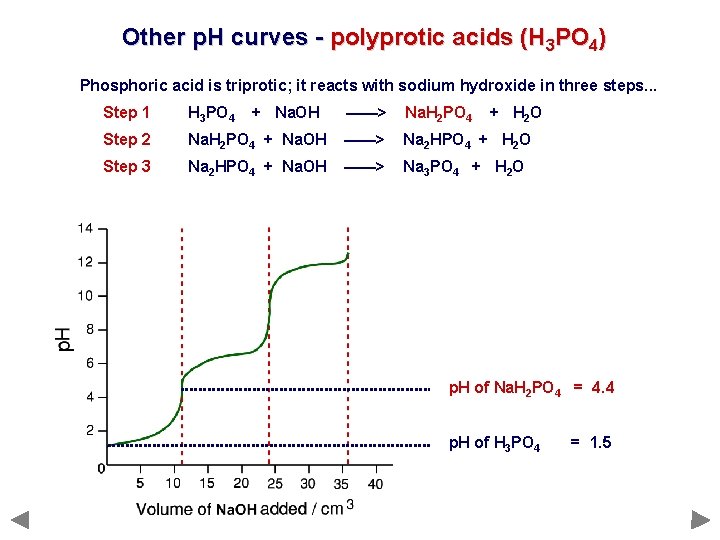
Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O p. H of Na. H 2 PO 4 = 4. 4 p. H of H 3 PO 4 = 1. 5

Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O p. H of Na 2 HPO 4 = 9. 6 p. H of Na. H 2 PO 4 = 4. 4 p. H of H 3 PO 4 = 1. 5
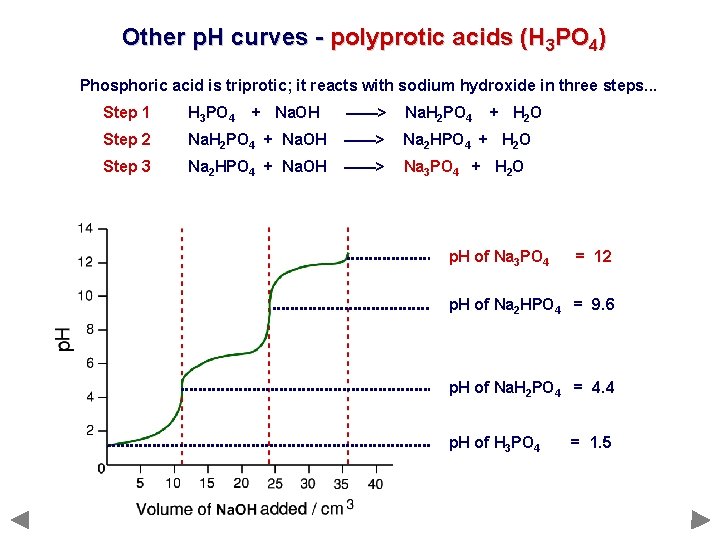
Other p. H curves - polyprotic acids (H 3 PO 4) Phosphoric acid is triprotic; it reacts with sodium hydroxide in three steps. . . Step 1 H 3 PO 4 Step 2 Step 3 + Na. OH ——> Na. H 2 PO 4 + H 2 O Na. H 2 PO 4 + Na. OH ——> Na 2 HPO 4 + H 2 O Na 2 HPO 4 + Na. OH ——> Na 3 PO 4 + H 2 O p. H of Na 3 PO 4 = 12 p. H of Na 2 HPO 4 = 9. 6 p. H of Na. H 2 PO 4 = 4. 4 p. H of H 3 PO 4 = 1. 5
- Slides: 31