Acids and Bases Three Definitions Arrhenius acid produces

Acids and Bases

Three Definitions • Arrhenius § acid – produces H+ in soln § base – produces OH- in soln • Bronsted-Lowry § acid – H+ donor § base – H+ acceptor in soln, don’t really have free H+/protons, have H 3 O+ called hydronium

Three Definitions • Arrhenius § acid – produces H+ in soln § base – produces OH- in soln • Bronsted-Lowry § acid – H+ donor § base – H+ acceptor • Lewis § acid – electron pair acceptor § base – electron pair donor
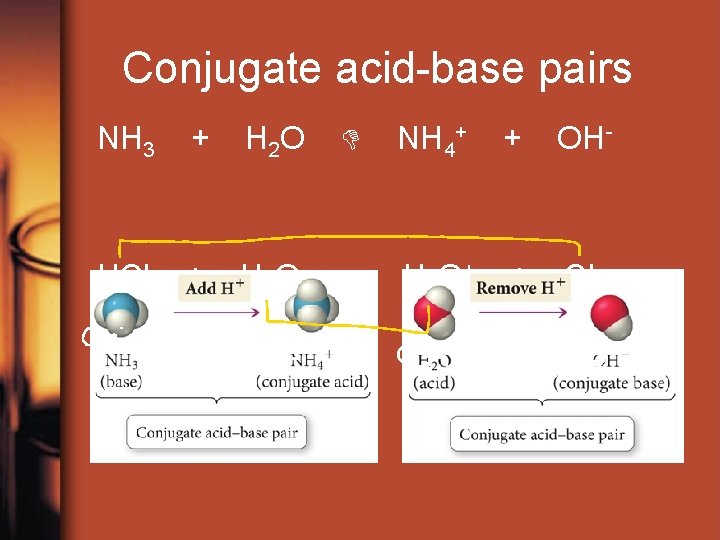
Conjugate acid-base pairs NH 3 + H 2 O NH 4+ + OH- HCl + H 2 O H 3 O + + Cl-

Conjugate acid-base pairs The stronger a Bronsted { }, the weaker its conjugate { }.
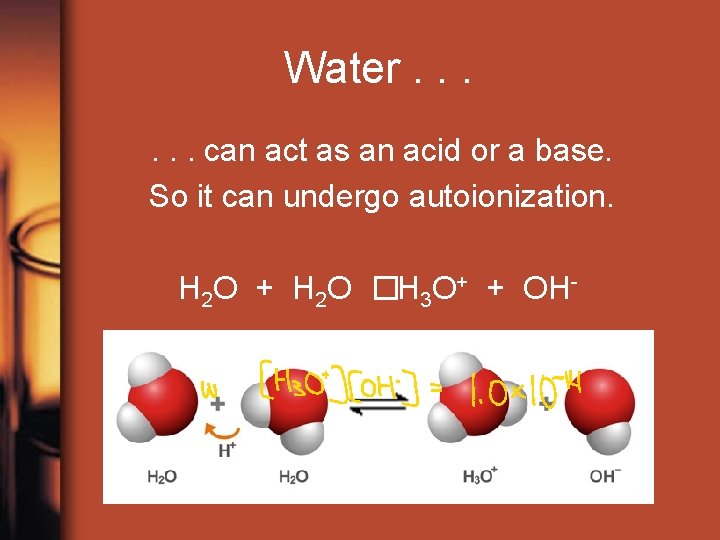
Water. . . can act as an acid or a base. So it can undergo autoionization. H 2 O + H 2 O �H 3 O+ + OHK =
![Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] = Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] =](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-7.jpg)
Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] = [OH-] < = 1. 0 x 10 -7 M
![Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] = Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] =](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-8.jpg)
Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 Neutral: [H+] = [OH-] = 1. 0 x 10 -7 M Acidic : [H+] > [OH-] [H+] > 1. 0 x 10 -7 M [OH-] < 1. 0 x 10 -7 M [H+] < [OH-] [H+] < 1. 0 x 10 -7 M [OH-] > 1. 0 x 10 -7 M Basic :
![p. H Scale p. H = -log [H+] p. OH = -log [OH-] p. p. H Scale p. H = -log [H+] p. OH = -log [OH-] p.](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-9.jpg)
p. H Scale p. H = -log [H+] p. OH = -log [OH-] p. H + p. OH = 14. 00
![p. H Scale Neutral: [H+] = [OH-] = 1. 0 x 10 -7 p. p. H Scale Neutral: [H+] = [OH-] = 1. 0 x 10 -7 p.](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-10.jpg)
p. H Scale Neutral: [H+] = [OH-] = 1. 0 x 10 -7 p. H = 7 Acidic : [H+] > 1. 0 x 10 -7 M p. H < 7 Basic : [H+] < 1. 0 x 10 -7 M p. H > 7
![p. H Scale Neutral: Acidic : Basic : [H+] = [OH-] = 1. 0 p. H Scale Neutral: Acidic : Basic : [H+] = [OH-] = 1. 0](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-11.jpg)
p. H Scale Neutral: Acidic : Basic : [H+] = [OH-] = 1. 0 x 10 -7 [H+] > 1. 0 x 10 -7 M [OH-] < 1. 0 x 10 -7 M [H+] < 1. 0 x 10 -7 M [OH-] > 1. 0 x 10 -7 M p. H = 7 p. OH = 7 p. H < 7 p. OH > 7 p. OH < 7
![Determine the p. OH, [OH-] and [H+] for a solution of vinegar with p. Determine the p. OH, [OH-] and [H+] for a solution of vinegar with p.](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-12.jpg)
Determine the p. OH, [OH-] and [H+] for a solution of vinegar with p. H = 3. 26. SIG FIGS!!!
![In ammonia, [OH-] = 4. 8 x 10 -2 M. Calculate its p. H, In ammonia, [OH-] = 4. 8 x 10 -2 M. Calculate its p. H,](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-13.jpg)
In ammonia, [OH-] = 4. 8 x 10 -2 M. Calculate its p. H, p. OH, and [H+].

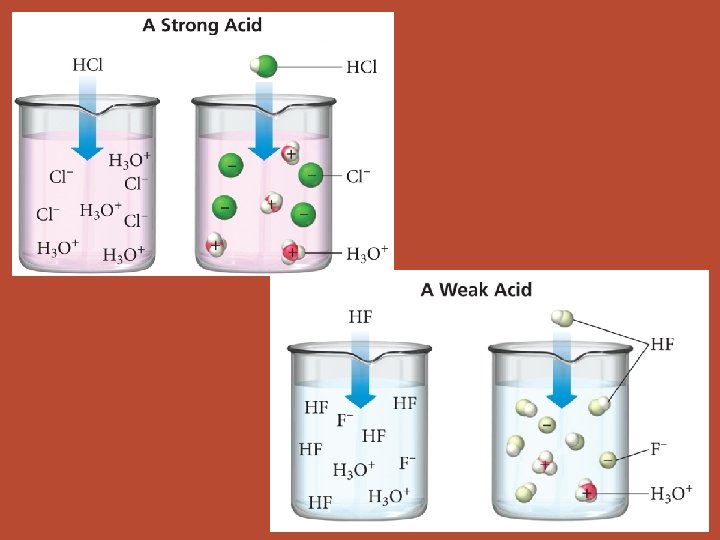
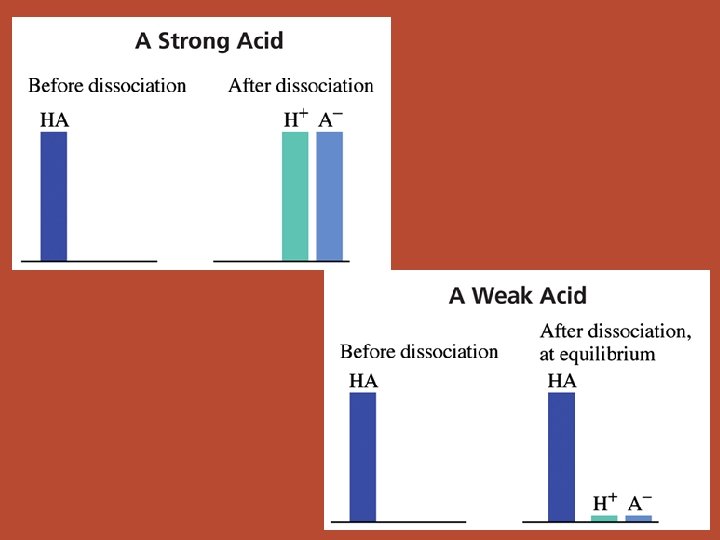
Strong Acids • ionize completely Weak Acids • only partially ionize • memorize them!!! • ALL other acids HCl, HBr, HI, HCl. O 4, H 2 SO 4, HNO 3 id c a r e ? b ? m ? e e r m u e t a R l c n e m no
![Strong Acids • ionize completely. [H+] = [HA]o • memorize them!!! HCl, HBr, HI, Strong Acids • ionize completely. [H+] = [HA]o • memorize them!!! HCl, HBr, HI,](http://slidetodoc.com/presentation_image_h/e6a55adbf539fbb2dcc368219783946c/image-17.jpg)
Strong Acids • ionize completely. [H+] = [HA]o • memorize them!!! HCl, HBr, HI, HCl. O 4, H 2 SO 4, HNO 3 • strong electrolytes Weak Acids • only partially ionize. need ICE table to calc [H+] • ALL other acids • weak electrolytes

Weak Acids • achieve equilibrium. soln mix contains mostly nonionized acid molecules, with some H 3 O+ and conjugate base ions. (ex) HCN Ka = 6. 2 x 10 -10

Weak Acids What does the Ka value tell us? Ka (HNO 2) = 4. 6 x 10 -4 Ka (HC 2 H 3 O 2) = 1. 8 x 10 -5 Ka (HCN) = 6. 2 x 10 -10

Bases • “Strong” and “weak” are used in the same way to describe bases. • memorize list of strong bases!!! Li. OH, Na. OH, KOH, Rb. OH, Cs. OH, Ca(OH)2*, Sr(OH)2, Ba(OH)2 *borderline
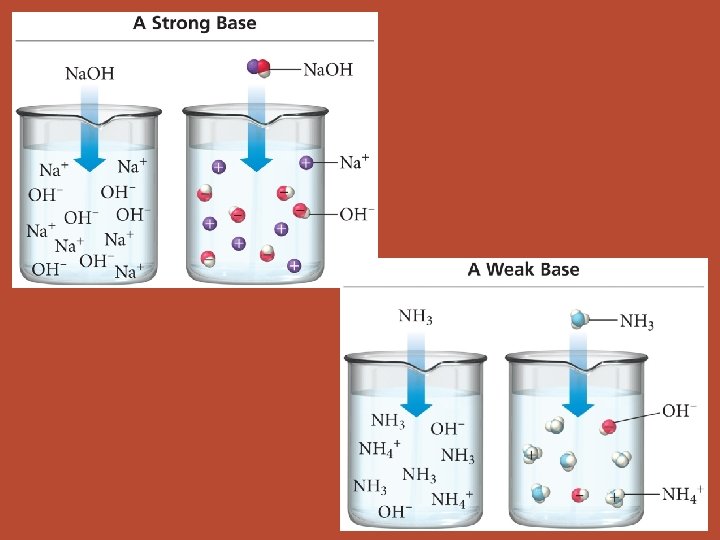
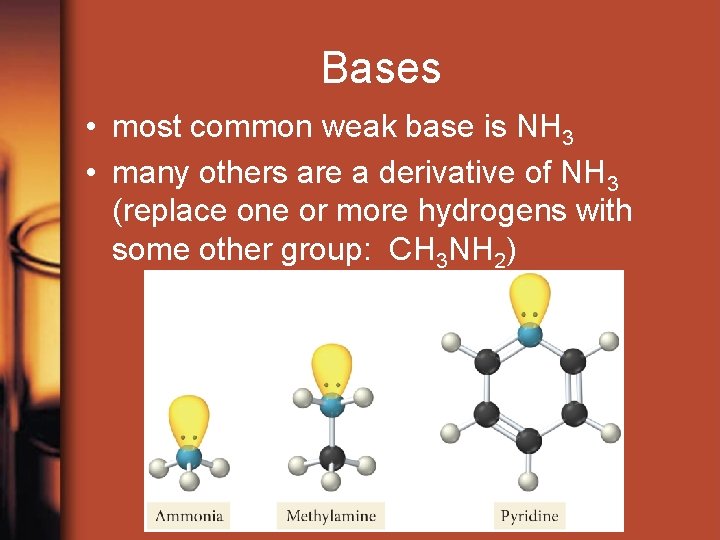
Bases • most common weak base is NH 3 • many others are a derivative of NH 3 (replace one or more hydrogens with some other group: CH 3 NH 2)

Conjugate Acid-Base Pairs Ka x Kb = Kw = 1. 0 x 10 -14 NH 3 / NH 4+ Kb (NH 3) = 1. 8 x 10 -5 Ka (NH 4+) = 5. 6 x 10 -10 Shows mathematically that the stronger an acid, the weaker its conjugate base.
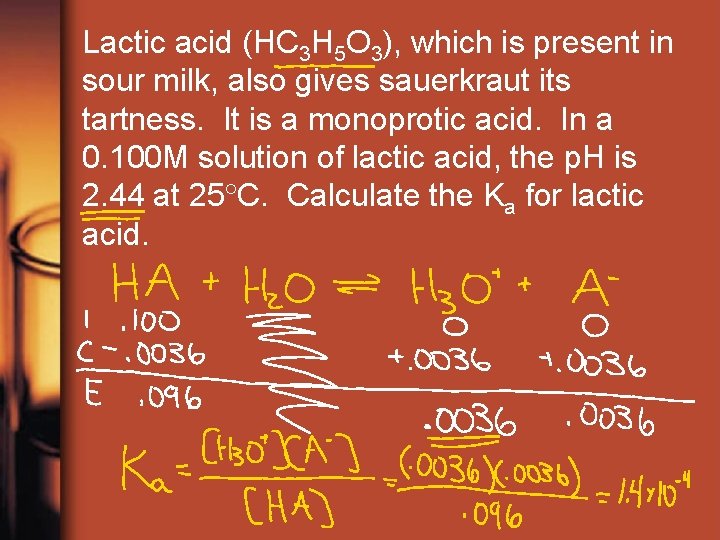
Lactic acid (HC 3 H 5 O 3), which is present in sour milk, also gives sauerkraut its tartness. It is a monoprotic acid. In a 0. 100 M solution of lactic acid, the p. H is 2. 44 at 25 C. Calculate the Ka for lactic acid.

A student planned an experiment that would use 0. 10 M propionic acid, HC 3 H 5 O 2. Calculate the values of [H+] and p. H for this solution. For propionic acid, Ka = 1. 4 x 10 -5.

A solution of hydrazine, C 2 H 4, has a concentration of 0. 25 M. What is the p. H of the solution? Kb (C 2 H 4) = 1. 7 x 10 -6.
- Slides: 26