Acids and Bases Theyre Everywhere Definitions Arrhenius Acids

Acids and Bases They’re Everywhere

Definitions • Arrhenius Acids – Acids produce hydrogen ions in solution – Bases produce hydroxide ions in solution – Only have one type of base, hydroxide – First description of what an acid/base was

Definitions • Bronsted-Lowry Model – Acid is a proton donor – Base is a proton acceptor H---Cl + H---O---H (H---O---H)+1 H + Water is the base (proton acceptor) HCl is the acid (proton donor) + Cl-1 +1 -1

Conjugate Pairs • The reaction that occurs is really an equilibrium • The H 3 O+1 is called the hydronium ion H---Cl + H---O---H HCl Acid + H 2 O Base (H---O---H)+1 H + Cl-1 H 3 O+1 + + Cl-1 Acid Base These are conjugate acid base pairs. Water is a base on one side and a acid on the other, this is called amphoteric

Quick Practice • Strong acid HNO 3 + H 2 O ↔ H 3 O+ + NO 3– What are the conjugate pairs? • Weak acid HF + H 2 O ↔ H 3 O+ + F– What are the conjugate pairs?

Weak Acids • Weak acids partially dissociate in water: • HA(aq) H+(aq) + A- (aq) – Bronsted-Lowry: HA + H 2 O H 3 O+ + A- • What is Kc for this dissociation? • Kc in a weak acid case is better known as Ka, or the acid-dissociation constant. • The larger the value of Ka, the stronger the acid.

Acid Dissociation Constant • HA + H 2 O H 3 O+1 + A • Generalized expression – The equilibrium expression for this process Ka = [H 3 O+1 ] [A-] = acid dissociation constant [HA] Note: the [water] is a constant (concentration of a pure solid or pure liquid is not included in an equilibrium expression) Water does play an important part in the dissociation of the acid!

Acid Strength • HA(aq) + H 2 O H 3 O+1(aq) + A-(aq) – A strong acid lies to the right Ka >>1 • [H+1] [HA]0 – A weak acid lies to the left Ka<<1 • [H+1] << [HA]0 – Typical Ka (dissociation) constants for weak acids • HF • HC 2 H 3 O 2 7. 2 x 10 -7 1. 8 x 10 -5
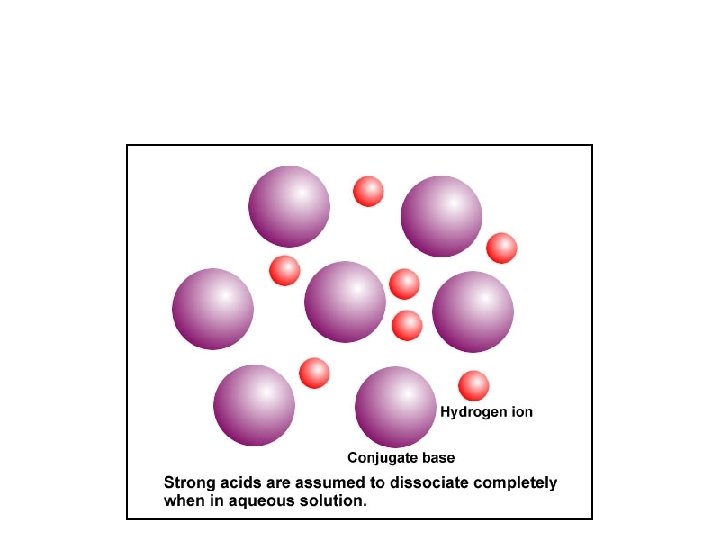
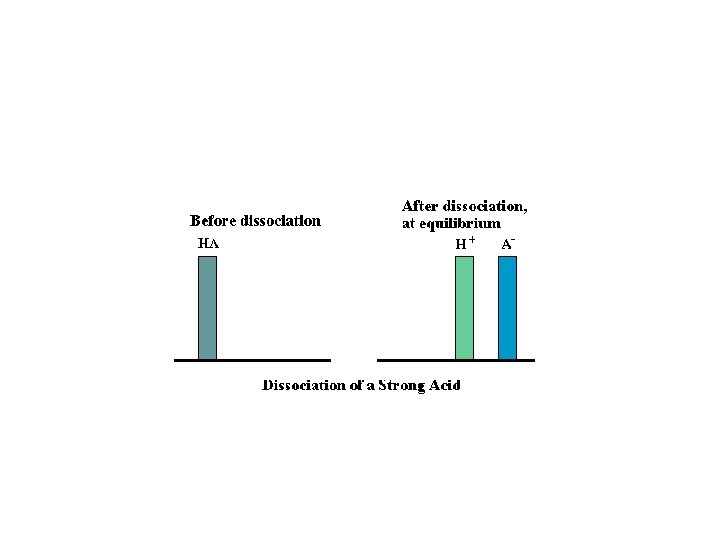
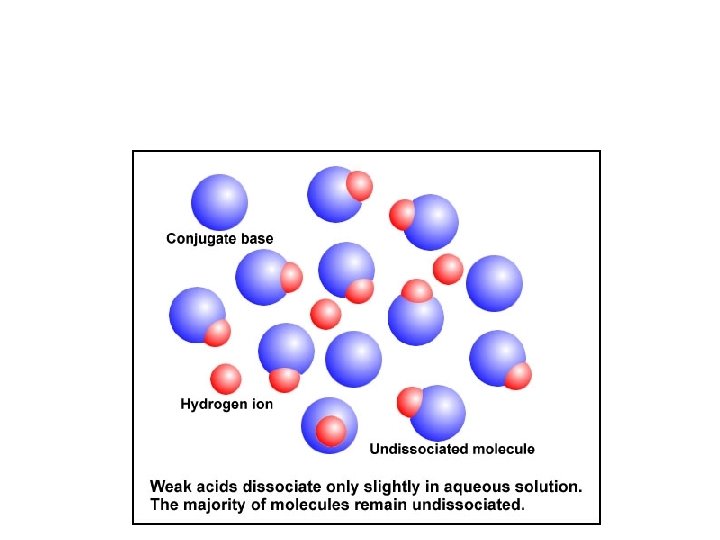

Conjugate Pairs and Strength • HA(aq) + H 2 O H 3 O+1(aq) + A-(aq) – Strong acids have weak conjugate bases – Strong bases have weak conjugate acids http: //www. chem. ubc. ca/courseware/p. H/index. html
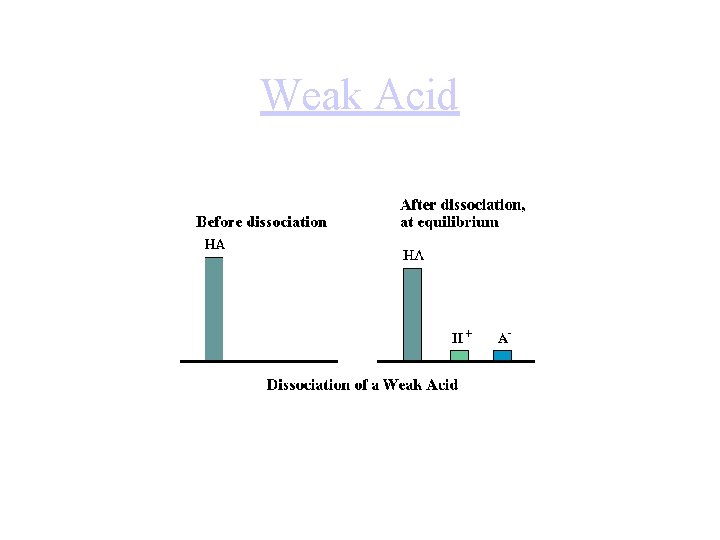
Weak Acid

Acid Strength • In an acid, the strength of the bond between the acidic hydrogen and the other atom (H-X) determines how strong the acid is. • In general, the strength of an H-X bond weakens as atoms get bigger. – So, going down a group, the strength of an acid increases. – HF < HCl < HBr < HI

Acid Strength • Going across a row, bond strengths don’t change all that much. So, bond polarity is the major factor – the more polar the bond, the stronger the acid • Period 2: CH 4 < NH 3 < H 2 O < HF
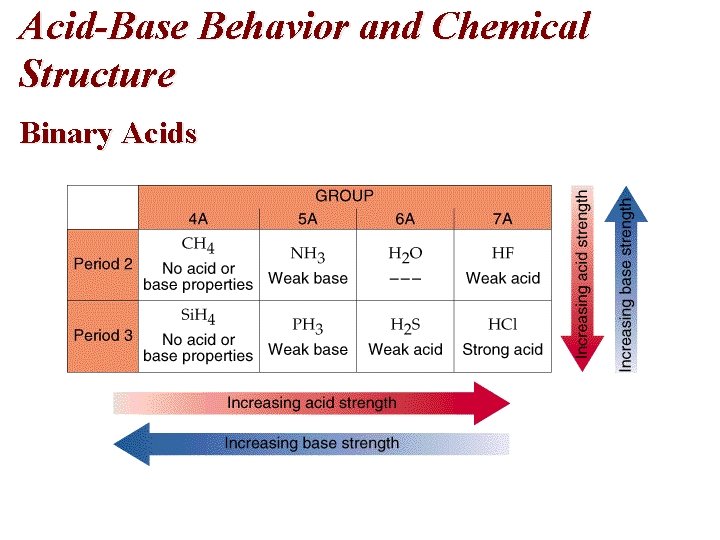
Acid-Base Behavior and Chemical Structure Binary Acids

Acid-Base Behavior and Chemical Structure Factors That Affect Acid Strength Consider H-X. For this substance to be an acid we need: • H-X bond to be polar with H + and X - (if X is a metal then the bond polarity is H -, X + and the substance is a base). • the H-X bond must be weak enough to be broken, • the conjugate base, X-, must be stable.

Acid-Base Behavior and Chemical Structure Binary Acids • Acid strength increases across a period and down a group. • Conversely, base strength decreases across a period and down a group. • What differences in atomic structure account for these variations? • The kernel charge increases across a period. Therefore, the nonmetallic element has a stronger pull on the shared electron pair and H is more easily ionized. • As you go down a group, the strength of the H-X bond weakens as the X has more shells and its size increases. Therefore, H is more easily ionized

Acid-Base Behavior and Chemical Structure Oxyacids • Oxyacids contain O-H bonds. • All oxyacids have the general structure Y-O-H. • The strength of the acid depends on Y and the atoms attached to Y. Y – If Y is a metal (low electronegativity), then the substances are bases. – If Y has intermediate electronegativity (e. g. I, EN = 2. 5), the electrons are between Y and O and the substance is a weak oxyacid

Acid-Base Behavior and Chemical Structure Oxyacids – If Y has a large electronegativity (e. g. Cl, EN = 3. 0), the electrons are located closer to Y than O and the O-H bond is polarized to lose H+. – The number of O atoms attached to Y increase the O-H bond polarity and the strength of the acid increases (e. g. HOCl is a weaker acid than HCl. O 2 which is weaker than HCl. O 3 which is weaker than HCl. O 4 which is a strong acid).
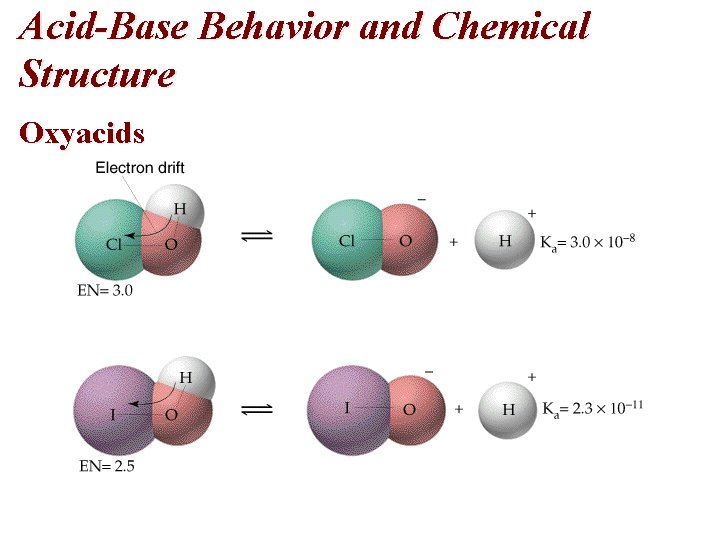
Acid-Base Behavior and Chemical Structure Oxyacids
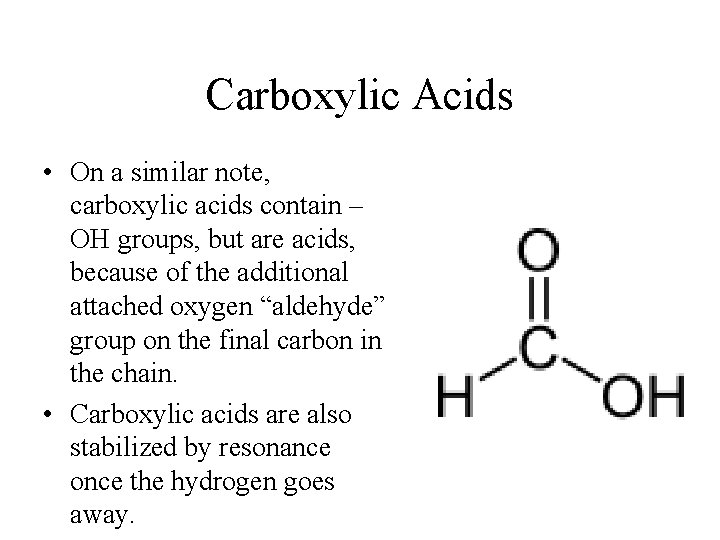
Carboxylic Acids • On a similar note, carboxylic acids contain – OH groups, but are acids, because of the additional attached oxygen “aldehyde” group on the final carbon in the chain. • Carboxylic acids are also stabilized by resonance once the hydrogen goes away.

B-l Acids and Bases • Weak acids and Bases • Know the difference between a Bronsted. Lowry acid/base and an Arrhenius acid and base

Strong Acid • Example 0. 10 M HNO 3 • H 2 O H 3 O+ + OH • There are two sources of H+ – The nitric acid and water • Since [H+] >>[OH-] in 0. 1 M nitric – Autoionization of water is insignificant – All the H+ is from HNO 3 • [H+] = 0. 10 M p. H = -log 0. 1 = 1. 0
![Strong Acid • The acid contributes all the [H+] • Example 1. 0 x Strong Acid • The acid contributes all the [H+] • Example 1. 0 x](http://slidetodoc.com/presentation_image_h2/48528c2cbeb8c14148d0a92d035c6885/image-25.jpg)
Strong Acid • The acid contributes all the [H+] • Example 1. 0 x 10 -10 HCl – The [H+] from autoionization (1 x 10 -7 M) is much higher. p. H = - log 1 x 10 -7 = 7

Weak Acids • • Treat like any equilibrium problem What is p. H of a 1. 00 M HF solution Kc = 7. 2 x 10 – 4 Kw = 1. 0 x 10 -14 – Since the Kc is so much bigger than the Kw, – HF is the major source of H+

p. H of 1. 0 M HF Solution I C E HF + H 2 O 1. 0 -x 1. 0 – x H 3 O+ + F 0(1 x 10 -7) 0 +x +x x x Ka = 7. 2 x 10 -4 = [F- ] [H+] = x • x = x 2 [HF] 1–x 1 Assume x is small (5% rule) X = 2. 7 x 10 -2 = [H+]
![Check for 5% Rule x 2 [HA]o – x [HA] • Ka = x Check for 5% Rule x 2 [HA]o – x [HA] • Ka = x](http://slidetodoc.com/presentation_image_h2/48528c2cbeb8c14148d0a92d035c6885/image-28.jpg)
Check for 5% Rule x 2 [HA]o – x [HA] • Ka = x 2 Ka • (Ka • [HA])1/2 X [HA]0 x 2 x x 100 5% • X = 2. 7 x 10 -2 x 100 1 5%

Weak Acid Equilibrium • List the Major species in solution – Don’t forget water as an acid source! • Choose the species that can produce H+ – Write balanced equation • • • Using the K values, choose dominate source H+ Write the equilibrium expression for dominate H+ Do “ICE” and solve using 5% rule Verify 5% Calculate [H+ ] and p. H

Polyprotic Acids • When acids are polyprotic, like the triprotic H 3 PO 4, where all three protons are weak acids, different Ka values are used. • H 3 PO 4 H 2 PO 4 - + H+ • H 2 PO 4 - HPO 42 - + H+ • HPO 42 - PO 43 - + H+ Ka 1 = 7. 1 x 10 -3 Ka 2 = 6. 3 x 10 -8 Ka 3 = 4. 5 x 10 -13 • If Ka 1 is more than 103 larger than Ka 2, you can ignore Ka 2 and treat it like a monoprotic acid.

Calculate p. H of 0. 100 M HOCl • Ka = 3. 5 x 10 -8 • You Calculate the p. H – Pg 675 if you need book • p. H = 4. 23

p. H of Weak Acid Mixture • Calculate the p. H of a mixture of 1. 00 M HCN, 5. 00 M HNO 2 and the equilibrium concentration of [CN-1] • HCN Ka = 6. 2 x 10 -10 • HNO 2 Ka = 4. 0 x 10 -4 • H 20 Kw = 1 x 10 -14 How do you approach this?
![Calculate p. H • Ka = 4. 0 x 10 -4 = [H+][NO 2 Calculate p. H • Ka = 4. 0 x 10 -4 = [H+][NO 2](http://slidetodoc.com/presentation_image_h2/48528c2cbeb8c14148d0a92d035c6885/image-33.jpg)
Calculate p. H • Ka = 4. 0 x 10 -4 = [H+][NO 2 -] [HNO 2] HNO 2 H+ + NO 2 I 5. 00 0 0 C -x +x +x E 5. 00 – x x x

Calculate p. H • Ka = 4. 0 x 10 -4 = = x 2 5. 00 - x x = [H+] = 4. 5 x 10 -2 M x 2 5 p. H = - log [H+] = 1. 35 Now calculate [CN-], you now [HCN] and [H+]
![Calculate [CN ] • Ka = 6. 2 x 10 – 10 = [CN-][H+] Calculate [CN ] • Ka = 6. 2 x 10 – 10 = [CN-][H+]](http://slidetodoc.com/presentation_image_h2/48528c2cbeb8c14148d0a92d035c6885/image-35.jpg)
Calculate [CN ] • Ka = 6. 2 x 10 – 10 = [CN-][H+] = [CN-][4. 5 x 10 -2] [CN] 1. 00 Solve for [CN-] = 1. 4 x 10 -8 M
![Percent Dissociation • % dissociation = [amount disassociated] x 100 [initial concentration] In the Percent Dissociation • % dissociation = [amount disassociated] x 100 [initial concentration] In the](http://slidetodoc.com/presentation_image_h2/48528c2cbeb8c14148d0a92d035c6885/image-36.jpg)
Percent Dissociation • % dissociation = [amount disassociated] x 100 [initial concentration] In the HF example [H+] = 1. 27 x 10 -2 M x 100 = 1. 27% [HF] 1. 00 M

Calculate the Percent Dissociation • 1. 00 M HC 2 H 3 O 2 • Left side of room • 0. 100 M HC 2 H 3 O 2 • Right side of room – Write on board

Percent Dissociation • For solutions of weak acids: – the more dilute the solution – The greater the percent dissociation General Proof Suppose have acid HA, with [HA]0 Dilute it to 1/10 th initial concentration Q = (x/10) = x 2 = 1/10 Ka [HA]/10 10 [HA] Since Q < Ka, the reaction moves to the right And you get a greater percent dissociation

Ka from % Dissociation • Lactic acid is 3. 7% dissociated @ 0. 100 M • HC 3 H 5 O H+ + C 3 H 5 O • Ka = [H+][C 3 H 5 O -] [HC 3 H 5 O] 3. 7% = x x 100 x = 3. 7 x 10 -3 [HC 3 H 5 O] Ka = [H+][C 3 H 5 O -] = (3. 7 x 10 – 3) [HC 3 H 5 O] 0. 100 Ka = 1. 4 x 10 -4

Example Weak acid equilibria Determine the p. H of a 0. 10 M benzoic acid solution at 25 o. C if Ka = 6. 5 x 10 -5 HBz(aq) + H 2 O(l) H+(aq) + Bz-(aq) The first step is to write the equilibrium expression. Ka = [H+] [Bz-] [HBz]

Weak acid equilibria HBz H+ Bz- Initial conc. , M 0. 10 0. 00 Change, DM -x +x Eq. Conc. , M 0. 10 - x x +x x [H+] = [Bz-] = x We’ll assume that [Bz-] and [H+] are negligible compared to [HBz], since the value of the Ka<< [HBz]. (6. 5 x 10 -5 << 0. 10 M)

Weak acid equilibria Solve the equilibrium equation in terms of x 2 x Ka = 6. 5 x 10 -5 =0. 10 x = (6. 5 x 10 -5 )(0. 10) = 0. 00255 M H+ p. H = - log (0. 0025 M) = 2. 6

Weak acid example Now, lets go for the exact solution Earlier, we found that for 0. 10 M benzoic acid 2 x Ka = 6. 5 x 10 -5 = 0. 10 -x X 2 + 6. 5 x 10 -5 X - 6. 5 x 10 -6 = 0 Use the quadratic equation to solve for x. X= -b + b 2 - 4 ac 2 a

Weak acid example -6. 5 x 10 -5 + [(6. 5 x 10 -5)2 +4 x 6. 5 x 10 -6]1/2 X= 2 X = 0. 00252 M H+ versus p. H = - log (0. 00252 M) = 2. 599 p. H = - log (0. 00255 M) = 2. 593 In this case, there is no significant difference between our two answers. If the Ka value is more than 2 powers of 10 different than the [acid], you can ignore the change in [acid].

Dissociation of bases, Kb The ionization of a weak base can also be expressed as an equilibrium. B (aq) + H 2 O(l) BH+(aq) +OH- (aq) The strength of a weak base is related to its equilibrium constant, Kb. [OH-] [BH+] Kb = [B]

Example Weak base equilibria The Bz-(aq) formed in the benzoic acid solution is a weak conjugate base. Determine the p. H of a 0. 10 M sodium benzoate solution Na. Bz(aq), at 25 o. C Bz-(aq) + H 2 O(l) HBz(aq) + OH-(aq) The Na+(aq) are spectator ions, and are not part of the equilibrium expression. Kb = [OH-] [HBz] [Bz-]

Example Weak base equilibria The Kb value is related to the Ka value by the equation Ka x Kb = Kw = 1. 0 x 10 -14 [H+] [Bz-] [HBz] [OH-] [HBz] [Bz-] = [H+] [OH-] Kb = Kw / Ka = 1. 0 x 10 -14 / 6. 5 x 10 -5 = 1. 5 x 10 -10

Weak base equilibria Bz - OH- HBz Initial conc. , M 0. 10 0. 00 Change, DM -x +x +x Eq. Conc. , M 0. 10 - x x x [OH-] = [HBz] = x We’ll assume that [HBz] and [OH-] are negligible compared to [Bz -], since the value of the Ka << [Bz -]. (1. 5 x 10 -10 << 0. 10 M)
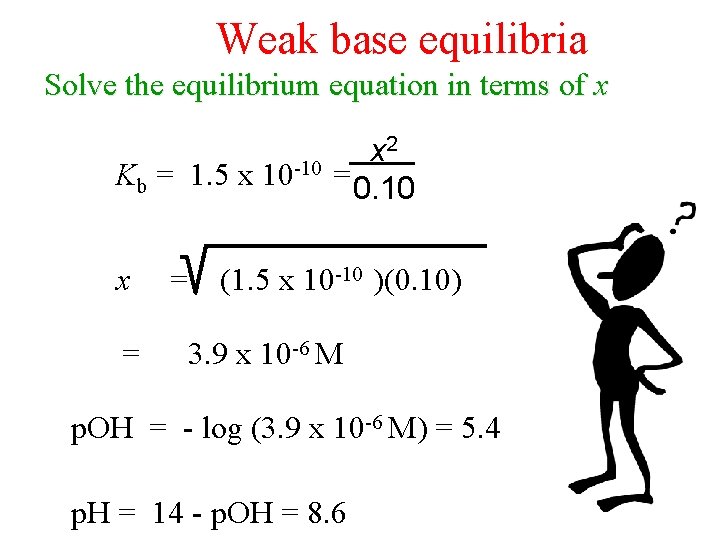
Weak base equilibria Solve the equilibrium equation in terms of x Kb = 1. 5 x 10 -10 x = = x 2 = 0. 10 (1. 5 x 10 -10 )(0. 10) 3. 9 x 10 -6 M p. OH = - log (3. 9 x 10 -6 M) = 5. 4 p. H = 14 - p. OH = 8. 6

Relationship Between Ka and Kb • What happens when you multiply Ka and Kb together? • Ka x Kb = [H+][OH-] = Kw = 1. 0 x 10 -14 • And, just like p. H + p. OH = 14. 00 for strong acids/bases at standard temperature… • p. Ka + p. Kb = p. Kw = 14. 00

Acidic and Basic Salts • Certain ions in solution can exhibit acid/base properties. • For example, consider the weak base, ammonia: – NH 3 + H 2 O NH 4+ + OH- • What if you dissolve the salt, ammonium sulfate, in water? – (NH 4)2 SO 4 2 NH 4+ + SO 42 - • Because NH 4+ is the conjugate acid of NH 3, when this salt dissolves in water, the solution will become slightly acidic.

Anions and Water • An anion that is a conjugate base of a weak acid raises the p. H of a solution: – X- + H 2 O HX + OH- • Example – sodium acetate in water – Ionic: Na+ + CH 3 COO- + H 2 O Na+ + CH 3 COOH + OH- – Net ionic: CH 3 COO- + H 2 O CH 3 COOH + OH • The Kb of this reaction can be found using the Ka of acetic acid (Ka x Kb = Kw)

Cations and Water • An cation that is a conjugate acid of a weak base that contains hydrogen lowers the p. H of a solution: – HX+ + H 2 O X + H 3 O+ • Example – ammonium chloride – Ionic: NH 4+ + Cl- + H 2 O NH 3 + Cl- + H 3 O+ – Net ionic: NH 4+ + H 2 O NH 3 + H 3 O+ • The Ka of this reaction can be found using the Kb of ammonia (Ka x Kb = Kw)

Some Rules • An anion that is the conjugate base of a strong acid will not affect the p. H of a solution. (Ex: Br- from HBr) • An anion that is the conjugate base of a weak acid will cause an increase in p. H (CN- from HCN) • A cation that is the conjugate acid of a weak base will cause a decrease in p. H (NH 4+ from NH 3)

Some Rules • Alkali metal cations and Ca 2+, Sr 2+, and Ba 2+ will not affect p. H (they are conjugate acids of strong bases). • Other metals (Al 3+, etc. ) will cause a decrease in p. H. • When a solution contains both a cation and anion that will affect p. H, the ion with the larger equilibrium constant (Ka or Kb will have the greater influence on p. H).

Lewis Acids and Bases • There is yet a third definition of acids and bases – the Lewis definition. • A Lewis base is defined to be an electron pair donor. • A Lewis acid is defined to be an electron pair receiver. • Understanding Lewis acids and bases requires the use of Lewis diagrams.

Lewis Acid-Base Reactions • Example 1: H+ and NH 3 – Empty s orbital on hydrogen • Example 2: NH 3 and BF 3 – Empty p orbital on boron • In order for a Lewis acid to receive an electron pair, there must be an empty orbital in the electron configuration.

Coordination Complexes • Certain transition metal cations can act as multiple Lewis acids (where the empty orbitals are is beyond the scope of AP Chemistry) • Fe 3+ has the ability to attract six electron pairs to itself: – Reaction of Fe 3+ and CN- • This kind of compound is known as a coordination complex.

Brief Preview of Organic Reactions • Lots of organic chemistry relies on “bonding sites” – or determining and predicting reaction mechanisms based on chemical structures. • Example – Lewis acid/base reaction between CO 2 and H 2 O to create H 2 CO 3

Hydrolysis of Metal Ions • When salts dissolve in water, the metal ions become hydrolyzed, or the water acts like a Lewis base and forms coordination compounds with the metal ions. • This is the mechanism behind metal ions acting like acids: • [Fe(H 2 O)6]3+ Fe(H 2 O)5(OH)2+ + H+

Lewis Acid/Base Review • Which of the following compounds can act as Lewis acids? • NH 3 • H 2 O • H+ • SO 42 • BCl 3

Hydride Bases • We are used to seeing hydrogen ions (H+) as acids… but there a class of compounds called hydrides (H-), which act as bases. – Ex: Na. H (ionic bond w/ H-)
- Slides: 62