Acids and Bases Strong Acid calculations Weak Acid

Acids and Bases Strong Acid calculations Weak Acid calculations

Finding the p. H of a Strong Acid • Strong acids – Ionize completely • HA H+ + A- – Do it only one at a time – So? ? • The concentration of the strong acid is EXACTLY the same as the [H+]

Finding the p. H of a Strong Acid • Example – Find the p. H of 1. 0 M HCl. – What contributes to H+? • HCl is an acid, water could be an acid • [HCl] = 1. 0 M [H+] = 1. 0 M • [H+] in water = 1. 0 x 10 -7 M • So HCl is the only significant acid • p. H = - log [H+] = - log [1. 0] = 0. 00

Finding the p. H of a Strong Acid • Example – Find the p. H of 0. 10 M HNO 3. – What contributes to H+? • HNO 3 is an acid, water could be an acid • [HNO 3] = 0. 10 M [H+] = 0. 10 M • [H+] in water = 1. 0 x 10 -7 M • So HNO 3 is the only significant acid • p. H = - log [H+] = - log [0. 10] = 1. 00

Finding the p. H of a Strong Acid • Example – Find the p. H of 1. 0 x 10 -10 M HBr. – What contributes to H+? • HBr is an acid, water could be an acid • [HBr] = 1. 0 x 10 -10 M [H+] = 1. 0 x 10 -10 M • [H+] in water = 1. 0 x 10 -7 M • So WATER is actually a better acid • p. H = - log [H+] = - log [1. 0 x 10 -7] = 7. 00

Finding the p. H of a Weak Acid • Don’t ionize completely (only partially) – So they make an EQUILIBRIUM – Write the equation with water using the Bronsted-Lowry definition – They’ll have a Ka value associated with them

A Couple Things about Ka • The relative strength of acids compared to each other can be found by comparing the Ka values for the acids – Strong acids have a Ka of infinity – Weak acids have known Ka values (look them up) • You can also find the Kb of its conjugate base – Ka x Kb = 1. 00 x 10 -14 • same as [H+][OH-] = 1. 00 x 10 -14
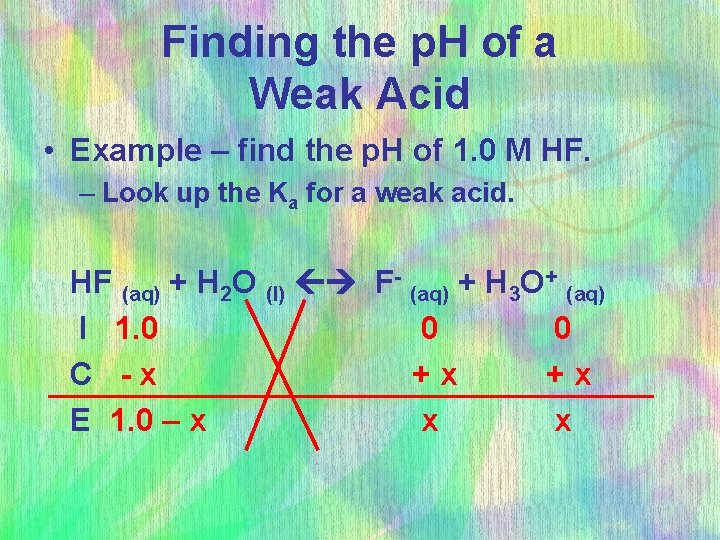
Finding the p. H of a Weak Acid • Example – find the p. H of 1. 0 M HF. – Look up the Ka for a weak acid. HF (aq) + H 2 O (l) F- (aq) + H 3 O+ (aq) I 1. 0 0 0 C -x +x +x E 1. 0 – x x x

Finding the p. H of a Weak Acid 7. 4 x 10 -4 = [x][x] [1. 0 -x] very small x = 2. 7 x 10 -2 5% rule? – What’s x? • So [H 3 O+] = 2. 7 x 10 -2 • p. H = - log [0. 027] = 1. 57

A Little Different Weak Acid calculation • Find the molarity of acetic acid is the p. H of the solution is 4. 15. HC 2 H 3 O 2 (aq) + H 2 O (l) C 2 H 3 O 2 - (aq) + H 3 O+ (aq) I ? ? ? C -x E ? ? ? – x 0 +x x

A Little Different Weak Acid calculation • p. H = 4. 15 – So [H 3 O+] = 10 -4. 15 = 7. 1 x 10 -5 M – So x = 7. 1 x 10 -5 M • 1. 8 x 10 -5 = [7. 1 x 10 -5] [? ? ? – x] – Solve for ? ? ? • 2. 8 x 10 -4 M

Last thing they could do • Give you the initial concentration of an unknown acid and its p. H, find Ka – 0. 0500 M HQ, p. H of 5. 31

Mixtures of Acids • When two or more acids are mixed together, you must calculate them in order from best “strongest” to weakest – Based on Ka

Mixtures of Acids • If both are strong…. – Basically finding the new molarity of H+ and taking the p. H – Ex – calculate the p. H of a mixture if 50. 0 m. L of 1. 00 M HCl and 100. 0 m. L of 3. 00 M HNO 3 are mixed.

Mixtures of Acids • Calculate the p. H of a solution that contains 1. 00 M HCN (Ka=6. 2 x 10 -10) and 5. 00 M HNO 2 (Ka=4. 0 x 10 -4). – Also calculate the [CN-] in this solution.

Mixtures of Acids • Start with the better acid HNO 2 (aq) + H 2 O (l) NO 2 - (aq) + H 3 O+ (aq) I 5. 00 0 0 C -x +x +x E 5. 00 – x x x 4. 0 x 10 -4 = [x][x] x = 0. 045 = [H 3 O+] [5. 00 -x]

Mixtures of Acids • Then do the next acid (BUT YOU ALREADY HAVE A CONCENTRATION OF H 3 O+ FROM THE FIRST ACID) HCN (aq) + H 2 O (l) CN- (aq) + H 3 O+ (aq) I 1. 00 0 0. 045 C -x +x +x E 1. 00 – x x 0. 045 + x 6. 2 x 10 -10 = [0. 045 +x][x] x = 1. 4 x 10 -8 = [CN-] [1. 00 -x] [H 3 O+] = 0. 045 p. H = - log [0. 045] = 1. 35
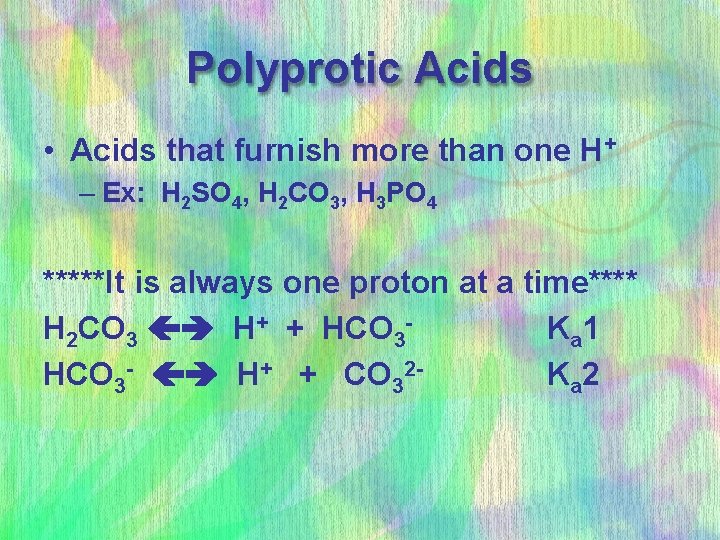
Polyprotic Acids • Acids that furnish more than one H+ – Ex: H 2 SO 4, H 2 CO 3, H 3 PO 4 *****It is always one proton at a time**** H 2 CO 3 H+ + HCO 3 K a 1 HCO 3 - H+ + CO 32 K a 2

Polyprotic Acids • Typically: Ka 1 > Ka 2 > Ka 3 • Usually by at least a factor of 100 x – Why? • The conjugate base of a weak acid is a pretty “good” base – so it would rather act like a base than an acid in most cases

Polyprotic Acid Calculation • Find the p. H of 5. 0 M H 3 PO 4 and the equilibrium [ ] of each conjugate base. – What are we trying to find? • • [H 2 PO 4 -] [HPO 42 -] [PO 43 -] [H 3 O+] p. H

Polyprotic Acid Calculation • Start with the first H+ (Ka 1) H 3 PO 4 (aq) + H 2 O (l) H 2 PO 4 - (aq) + H 3 O+ (aq) I 5. 0 C -x E 5. 0 – x 0 +x x 7. 5 x 10 -3 = [x][x] x = 0. 038 = [H 3 O+] = [H 2 PO 4 -] [5. 0 -x]

Polyprotic Acid Calculation • Then do the next hydrogen (Ka 2)– use the values for the things you just calculated in this one too!! H 2 PO 4 - (aq) + H 2 O (l) HPO 42 - (aq) + H 3 O+ (aq) I 0. 038 C -x E 0. 038 – x 0 +x x 0. 038 + x 6. 2 x 10 -8 = [0. 038 +x][x] x = 6. 2 x 10 -8 = [HPO 42 -] [0. 038 -x] [H 3 O+] now = 0. 038 + 6. 2 x 10 -8 = 0. 038

Polyprotic Acid Calculation • Then do the last hydrogen (Ka 3) – use the values for the things you just calculated in this one too!! HPO 42 - (aq) + H 2 O (l) PO 43 - (aq) + H 3 O+ (aq) I 6. 2 x 10 -8 C -x E 6. 2 x 10 -8 – x 4. 8 x 10 -13 = [0. 038 +x][x] [6. 2 x 10 -8 -x] [H 3 O+] still 0. 038 M p. H = 1. 42 0 +x x 0. 038 + x x = 7. 8 x 10 -19 = [PO 43 -]
- Slides: 23