Acids and Bases Review of Electrolytes A substance

Acids and Bases

Review of Electrolytes A substance that dissolves in water and forms positive and negative ions. § conducts an electrical current due to mobile charged ions § Aqueous solutions of acids and bases conduct electricity

Acids § conduct electricity. when aqueous § react with metals to produce a hydrogen gas Ex: HCl + Zn Zn. Cl 2 + H 2(g) § cause color change with indicators Table M § react with bases to form H 2 O and a salt (neutralization) § have a sour taste (ex: lemons) § p. H below 7 § Begins with H+

Table K § Lists common names of acids and their formulas

Table J for Acids Any metal above the H 2** on Table J will react with an acid to produce H 2 (g)

Bases § § § § conduct electricity when aqueous have a slippery or soapy feel cause color change with indicators Table M react with acids to form H 2 O and a salt (neutralization) have a bitter taste (ex: Baking Soda) p. H above 7 Ends with OH-

Table L § Gives names of common bases and their formulas

Arrhenius Theory § Acids and Bases have charged particles called Hydronium ions (H+ or H 3 O+) and Hydroxide (OH-)ions

Arrhenius Acids § Substances whose water solutions contain H+ ions (protons) as the only cation § H+ is attracted to the H 2 O unshared pair and will covalently bond with O to form H 3 O+ Hydronium ion § Properties of an acid are determined by the Hydronium ion - the more hydronium ions, the more acidic it is

Mono and Polyprotic Acids § Monoprotic – acids that produce a single (mono) H+ Ex: HCl H+ + Cl- § Polyprotic – acids that produce more than 1 H+(Diprotic – 2, Triprotic – 3) Ex: Ionizes in 2 steps H 2 SO 4 H+ + HSO 4 H+ + SO 42 -

Arrhenius Bases § Substances whose water solutions contain OH- ions (hydroxide ions) as the only anion § Hydroxide ions are responsible for the slippery or soapy feel and bitter taste § Bases yield the OH- in solution Ex: NH 3 + H 2 O NH 4+ + OH-

Neutralization Reaction § Acids and bases can neutralize each other to form water and a salt (ionic compound) § The H+ from the acid combines with the OH- in bases § During the reaction, the H+ will gain the electron to become H 2 O § Properties of acid and base are destroyed Ex: HCl(aq) + Na. OH(aq) H 2 O(l) + Na. Cl(aq)

Alternate Theory § Bronstead-Lowery – acid is a proton (H+) donor and bases are proton acceptors
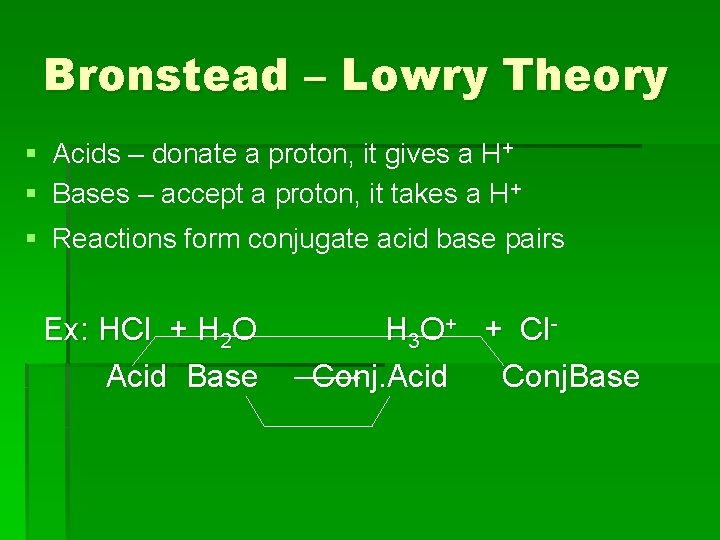
Bronstead – Lowry Theory § Acids – donate a proton, it gives a H+ § Bases – accept a proton, it takes a H+ § Reactions form conjugate acid base pairs Ex: HCl + H 2 O Acid Base H 3 O+ + Cl. Conj. Acid Conj. Base

Naming Acids and Bases § Acids general formula HX Ion Ending Prefix -ide hydro. Ex: HCl – hydrochloric Acid -ate NONE Ex: HCl. O 3 – Chloric Acid -ite NONE Ex: HCl. O 2 - Chlorous Acid Ending -ic -ous § Bases General Formula XOH § Name the cation and then use hydroxide

Amphoteric § When some substances act like an acid or a base § Ex: Amino acids and water § Water is usually neutral but can show characteristics in an equation of acids and bases. It produces it’s own ionization.

Titration § Used to find the molarity (concentration) of an unknown solution § Process of adding measured volumes of acid or base of known molarity (standard solution) to an unknown solution until neutralization occurs § Endpoint titration is the point when the titration is complete § Moles of acid = moles of base § Moles of H+ = moles of OH-

Formula From Table T § Listed as Titration MA V A = M B V B MA = molarity of H+ VA = Volume of Acid MB = Molarity of OHVB = Volume of Base Is used to calculate the molarity or volume of both acids and bases in neutralization reactions

Titration Formula Ex: What is the concentration of hydrochloric acid solution if 50. 0 m. L of a 0. 250 M KOH are needed to neutralize 20. 0 m. L of the HCl solution of unknown concentration? MA V A = M B V B MA(20 m. L) = (0. 250 M)(50 m. L) MA = 0. 625 M

p. H § Indicates the strength of an acid or a base § Scaled from 0 to 14 § p. H scale is logarithmic (increases tenfold) § p. H below 7 is acidic § p. H above 7 is basic § p. H of 7 is neutral

Acid Base Indicators § Table M gives a list of indicators and p. H ranges for that indicator with color changes Changes color when it gains or loses a proton (H+)

Concentration § § § § § p. H 0 strongest Acid p. H 1 – 2 strong acids p. H 3 – 5 medium/strong acid p. H 6 – 6. 9 weak acid p. H 7 neutral p. H 7. 1 – 8 weak base p. H 9 – 11 medium/strong base p. H 12 – 13 strong base p. H 14 strongest base § http: //www. quia. com/servlets/quia. activities. common. Activity. Player? AP _rand=1293143741&AP_activity. Type=11&AP_url. Id=1975&AP_continu e. Play=true&id=1975
- Slides: 22