Acids and Bases Properties of Acids sour taste

Acids and Bases

Properties of Acids: • sour taste • produces H+ ions when dissolved in water Properties of Bases: o bitter taste o feels slippery o produces OH- ions when dissolved in water
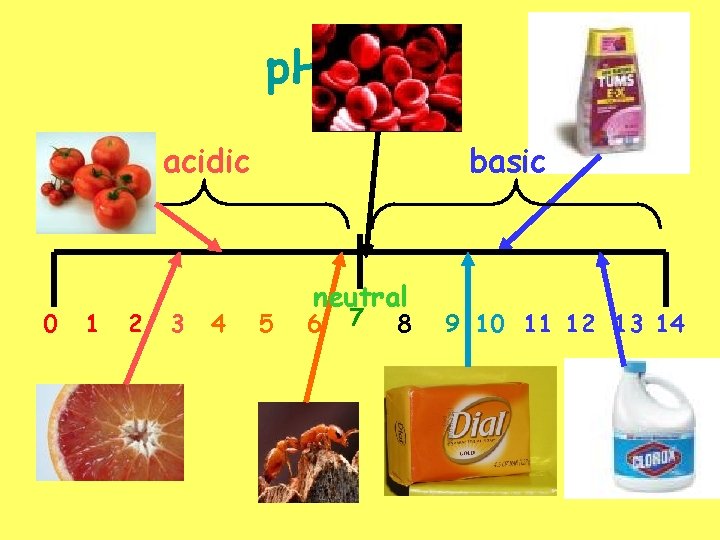
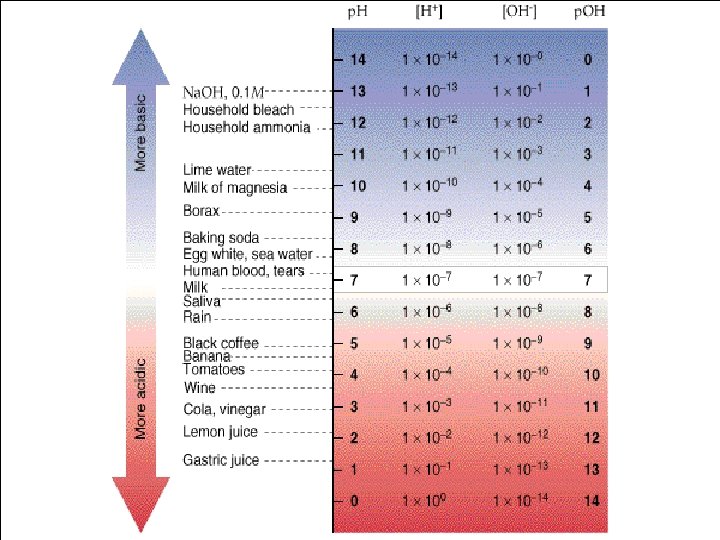
p. H scale acidic 0 1 2 3 4 basic 5 neutral 6 7 8 9 10 11 12 13 14
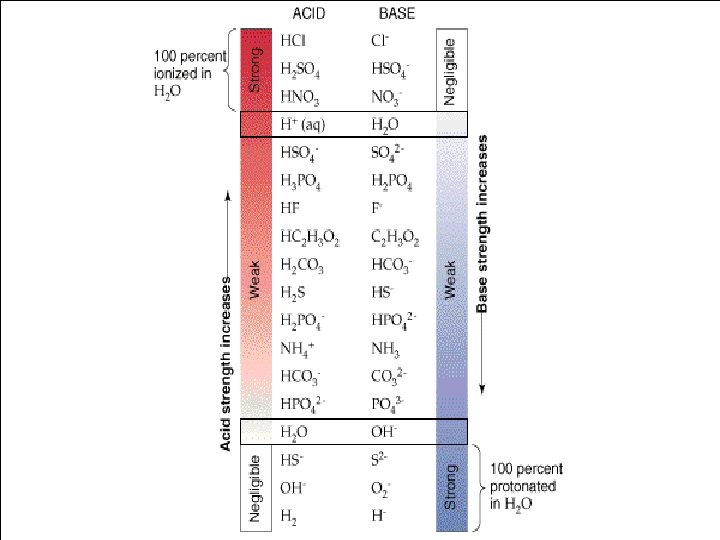
7 strong acids and 8 strong bases • Acids - HI, HBr, HCl. O 3, HCl. O 4, H 2 SO 4, HNO 3 • Bases – Li. OH, Na. OH, KOH, Rb. OH, Cs. OH, Ca(OH)2, Sr(OH)2, Ba(OH)2

The Arrhenius Definition of an Acid and a Base An acid is a substance that produces H+ ions in water solutions HCl H+ + Cl. A base is a substance that produces OH- ions in a water solution Na. OH Na+ + OH-
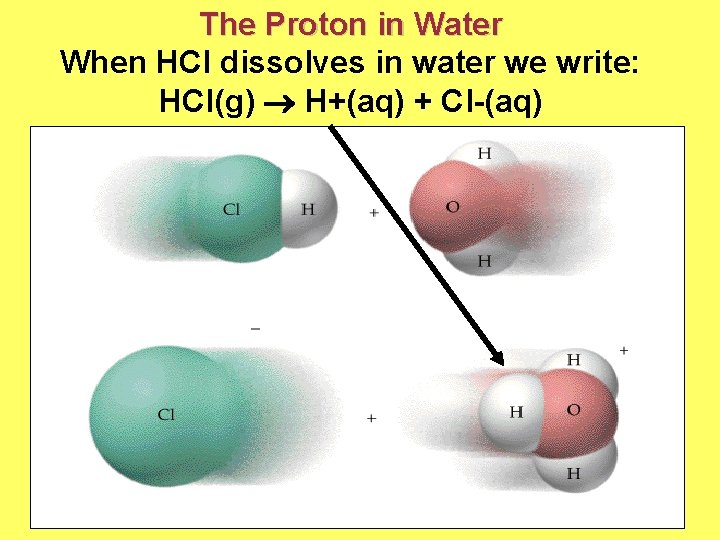
The Proton in Water When HCl dissolves in water we write: HCl(g) H+(aq) + Cl-(aq)

Acidic solutions are formed by a chemical reaction in which and acid transfers a proton (H+) to water, so we can write them either way. + HCl(aq) + H 2 O(aq) H 3 O (aq) + Cl (aq) or HCl(aq) H+(aq) + Cl-(aq)

The Bronsted-Lowry definition for Acids and Bases Acids may be defined as a substance that is capable of donating protons Bases may be defined as substance that accepts protons. HCl + NH 3 acid base NH 4+ + conjugate acid Clconjugate base
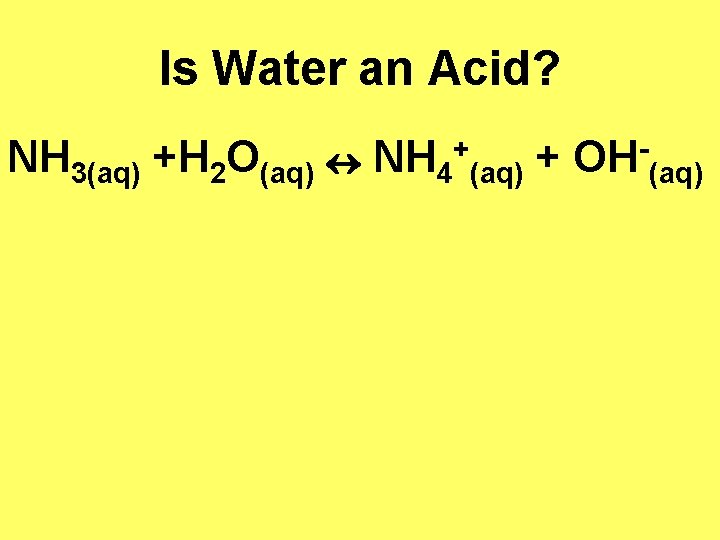
Is Water an Acid? NH 3(aq) +H 2 O(aq) NH 4 + (aq) + OH (aq)
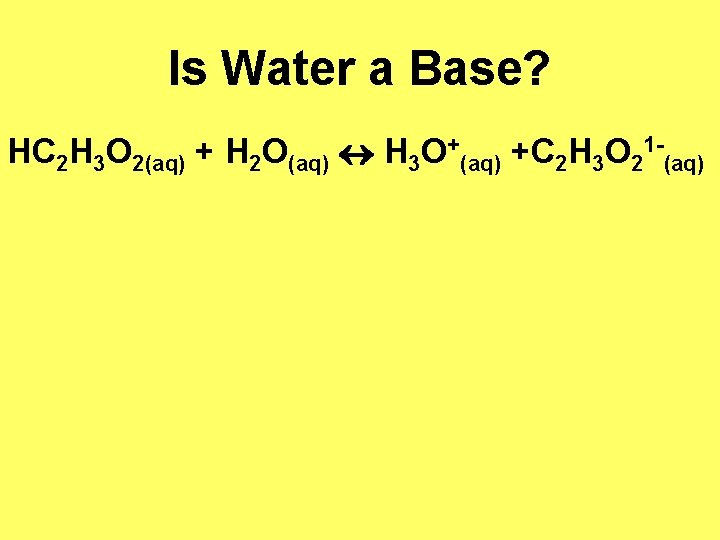
Is Water a Base? HC 2 H 3 O 2(aq) + H 2 O(aq) H 3 O+(aq) +C 2 H 3 O 21 -(aq)
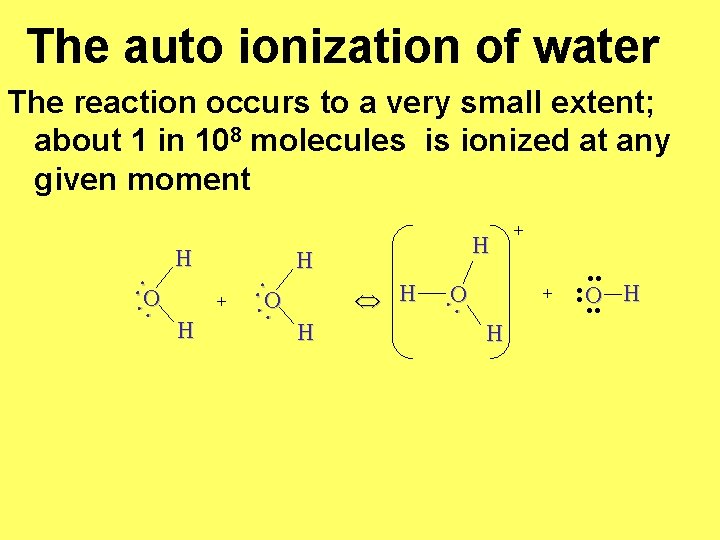
The auto ionization of water The reaction occurs to a very small extent; about 1 in 108 molecules is ionized at any given moment H H H O + : O : : : O H + H . . : O. . H
![Dissociation of Water, p. H Scale H 2 O(l) H+(aq) + OH-(aq) K =[H+] Dissociation of Water, p. H Scale H 2 O(l) H+(aq) + OH-(aq) K =[H+]](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-13.jpg)
Dissociation of Water, p. H Scale H 2 O(l) H+(aq) + OH-(aq) K =[H+] [OH-] [H 2 O] since water is a liquid and its concentration is therefore constant, this expression may be written as: Kw = [H+] [OH-] = 1. 0 x 10 -14 [H+] = [OH-] = 1. 0 x 10 -7 M
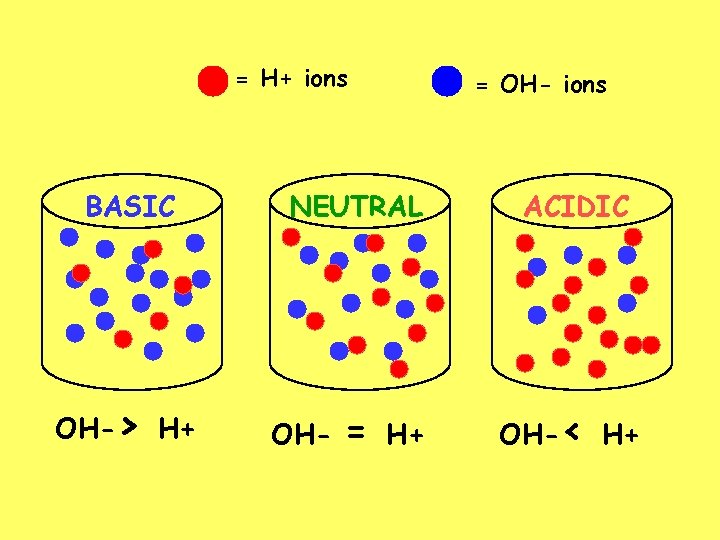
= H+ ions BASIC OH-> H+ = OH- ions NEUTRAL ACIDIC = OH-< H+ OH- H+

Sample exercise: Indicate whether each of the following solutions is neutral, acidic, or basic: a) [H+] = 2 x 10 -5 M b) [OH-] = 0. 010 M c) [OH-] = 1. 0 x 10 -7 M
![Calculate the concentration of H+(aq) in (a)a solution in which the [OH-] is 0. Calculate the concentration of H+(aq) in (a)a solution in which the [OH-] is 0.](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-16.jpg)
Calculate the concentration of H+(aq) in (a)a solution in which the [OH-] is 0. 020 M (b)a solution in which the [OH-] = 2. 5 x 10 -6 M. Indicate whether the solution is acidic or basic
![The p. H Scale p. H = -log [H+] If [H+] = 2. 5 The p. H Scale p. H = -log [H+] If [H+] = 2. 5](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-18.jpg)
The p. H Scale p. H = -log [H+] If [H+] = 2. 5 x 105 the p. H is? -5 p. H = -log [2. 5 x 10 ] = 4. 6 + H If p. H is 3. 8 the concentration is Antilog -3. 8= 1. 58 x 10 -4 M
![In a sample of lemon juice, [H+] = 3. 8 x 10 -4 M. In a sample of lemon juice, [H+] = 3. 8 x 10 -4 M.](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-19.jpg)
In a sample of lemon juice, [H+] = 3. 8 x 10 -4 M. What is the p. H? A commonly available window cleaner has a [H+] = 5. 3 x 10 -9 M. What is the p. H? In a sample of freshly pressed apple juice has a p. H of 3. 76. Calculate the [H+]
![What if we took the –log of the Kw expression Kw = [H+] [OH-] What if we took the –log of the Kw expression Kw = [H+] [OH-]](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-20.jpg)
What if we took the –log of the Kw expression Kw = [H+] [OH-] = 1. 0 x 10 -14 p. Kw = p. H + p. OH = 14
![What is the p. H, [H+], [OH-], of a solution with a p. OH What is the p. H, [H+], [OH-], of a solution with a p. OH](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-21.jpg)
What is the p. H, [H+], [OH-], of a solution with a p. OH of 2. 5? Is the solution acidic or basic?
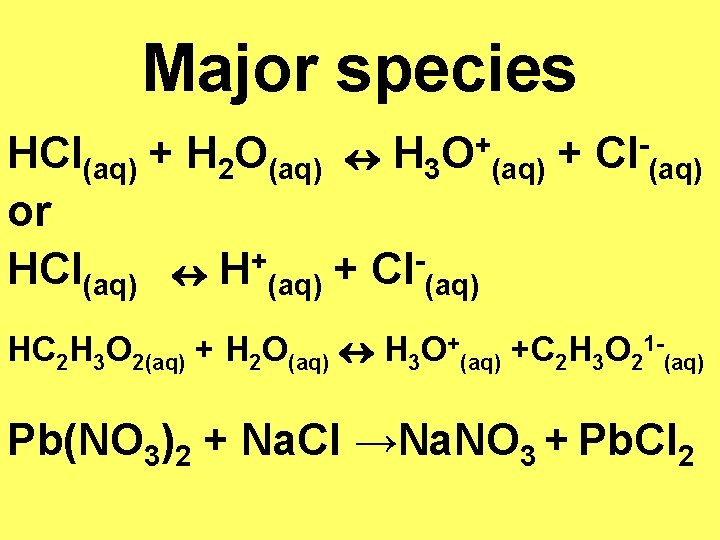
Major species HCl(aq) + H 2 O(aq) H 3 or + HCl(aq) H (aq) + Cl (aq) + O (aq) + Cl (aq) HC 2 H 3 O 2(aq) + H 2 O(aq) H 3 O+(aq) +C 2 H 3 O 21 -(aq) Pb(NO 3)2 + Na. Cl →Na. NO 3 + Pb. Cl 2

Indicators
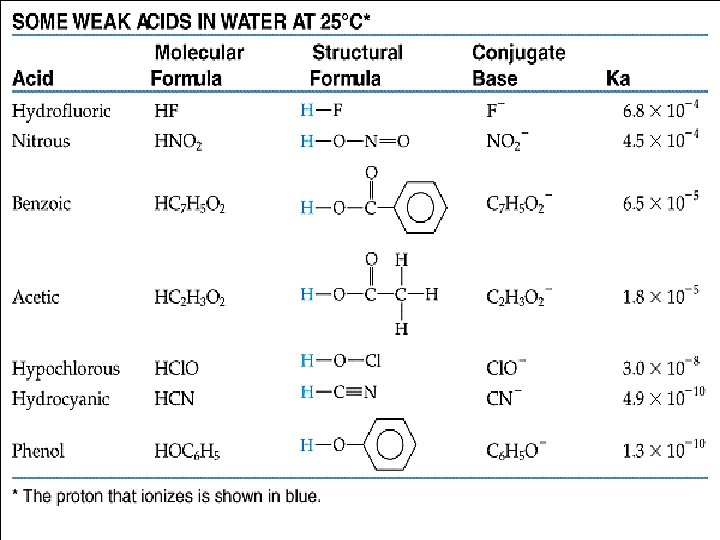

What is the p. H of 0. 010 M solution of HCl? If it ionizes completely which is what strong means then take the negative log of the concentration. HCl(aq) H+(aq) + Cl-(aq). 01 M p. H = 2

What is the p. H of a solution made from 20 m. L of 2. 0 M HCl and 35 m. L of 3. 2 M HNO 3?
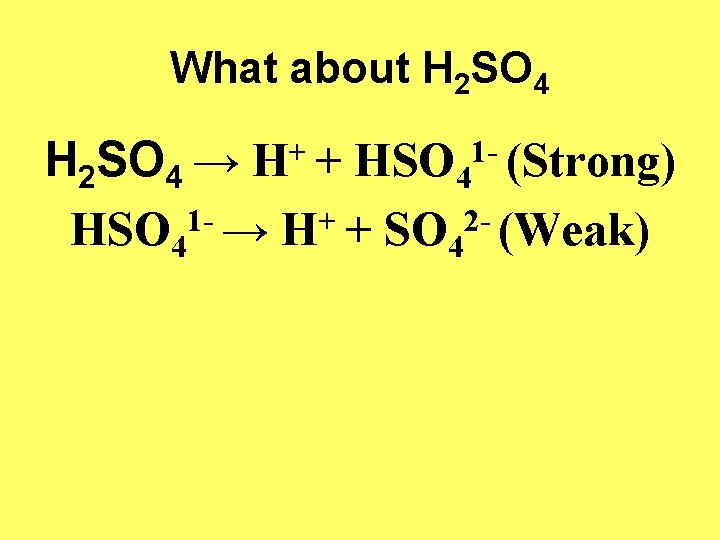
What about H 2 SO 4 → H+ + HSO 41 - (Strong) 1+ 2 HSO 4 → H + SO 4 (Weak)
![What about weak acids HX(aq) + (aq), then + Ka = [H ][X ] What about weak acids HX(aq) + (aq), then + Ka = [H ][X ]](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-28.jpg)
What about weak acids HX(aq) + (aq), then + Ka = [H ][X ] [HX] The smaller the value of the acid dissociation constant Ka, the weaker the acid + H X
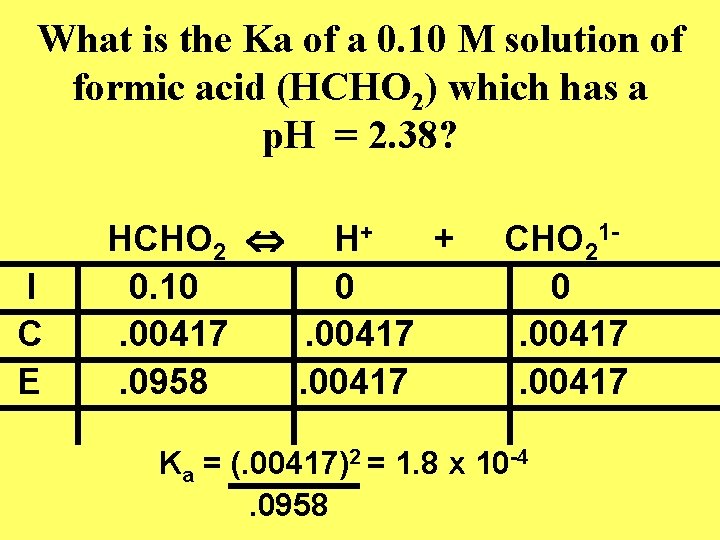
What is the Ka of a 0. 10 M solution of formic acid (HCHO 2) which has a p. H = 2. 38? I C E HCHO 2 H+ + 0. 10 0. 00417. 0958. 00417 CHO 210. 00417 Ka = (. 00417)2 = 1. 8 x 10 -4. 0958

What is the concentration of H+ ions in a 0. 10 M solution of HC 2 H 3 O 2 (Ka -5 = 1. 8 x 10 )? p. H? % ionization? HC 2 H 3 O 2 I. 10 C -X E. 10 – X 1. 8 x 10 -5 = X 2. 10 – X H+ + C 2 H 3 O 210 0 +X +X X = 1. 3 x 10 -3 p. H = 2. 87

percent dissociation 1. 3 X. 10 -3 10 x 100 = 1. 3%

What is the p. H and percent ionization of a 0. 20 M solution of HCN? Ka = 4. 9 x 10 -10

Acid-Base Equilibria: Strong Bases The most common soluble strong Bases are the hydroxides of group IA and Ca, Ba and Sr What is the p. H of a 0. 010 M solution of Ba(OH)2?
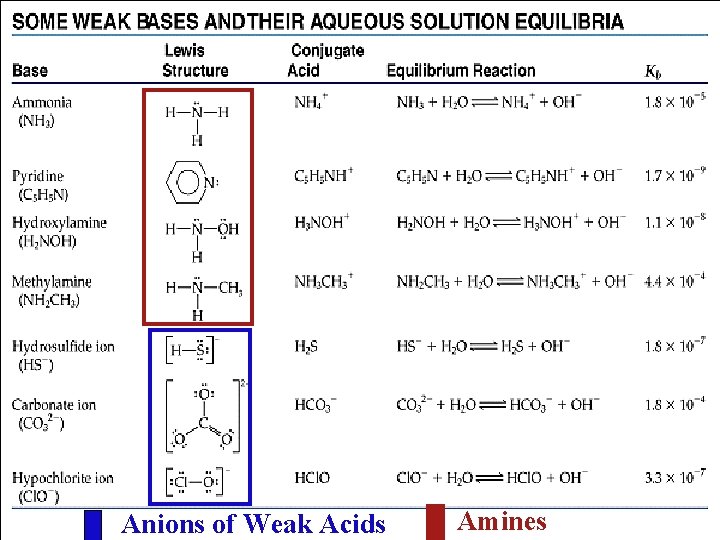
Anions of Weak Acids Amines

Dealing with Weak Bases The base dissociation constant Kb refers to the equilibrium in which a base reacts with H 2 O to form the conjugate acid and OHWeak base + H 2 O conjugate acid + OHNH 3 (aq) + H 2 O (l) NH 4 +(aq) + OH-(aq) +] [OH-] [NH 4 Kb = [NH 3] Calculate the [OH-] in a 0. 15 M solution of NH 3 + H 2 O NH 4+ + OHI C E
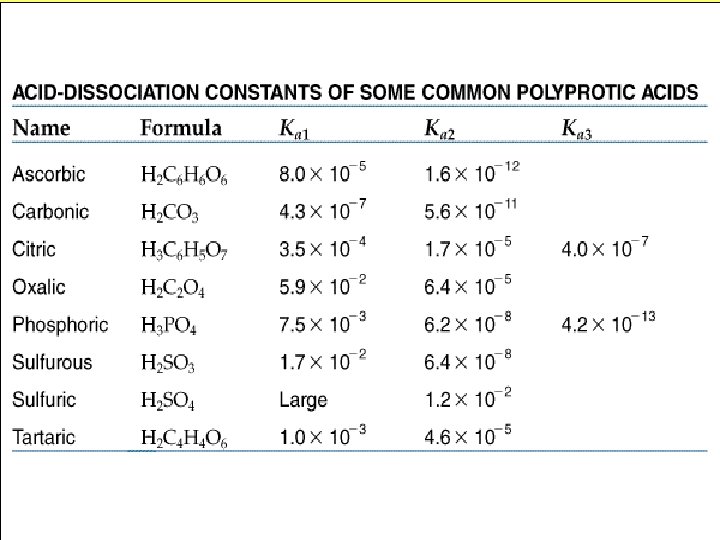
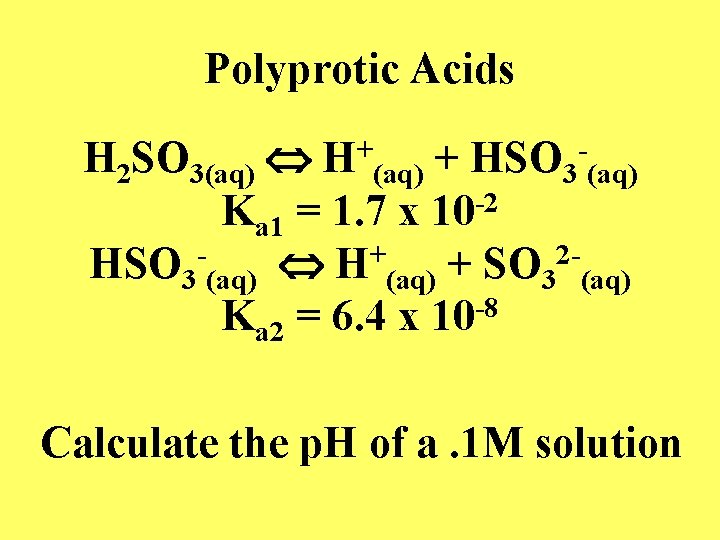
Polyprotic Acids H 2 SO 3(aq) + HSO 3 (aq) -2 Ka 1 = 1. 7 x 10 HSO 3 -(aq) H+(aq) + SO 32 -(aq) Ka 2 = 6. 4 x 10 -8 + H - Calculate the p. H of a. 1 M solution
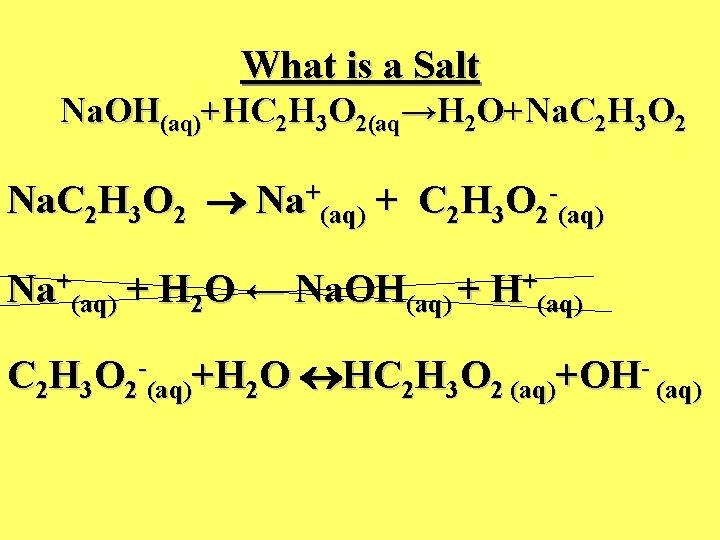
What is a Salt Na. OH(aq)+HC 2 H 3 O 2(aq→H 2 O+Na. C 2 H 3 O 2 Na+(aq) + C 2 H 3 O 2 -(aq) Na+(aq) + H 2 O ← Na. OH(aq) + H+(aq) C 2 H 3 O 2 -(aq)+H 2 O HC 2 H 3 O 2 (aq)+OH- (aq)
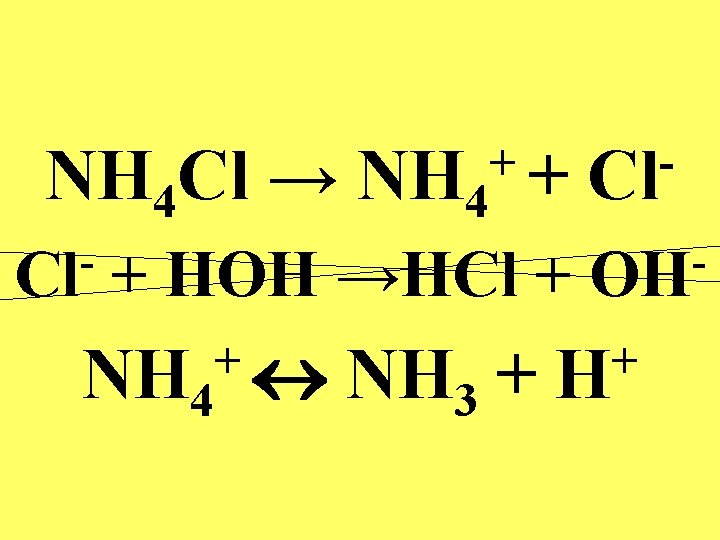
NH 4 Cl → NH 4 Cl ++ + HOH →HCl + + NH 4 NH 3 + Cl OH + H
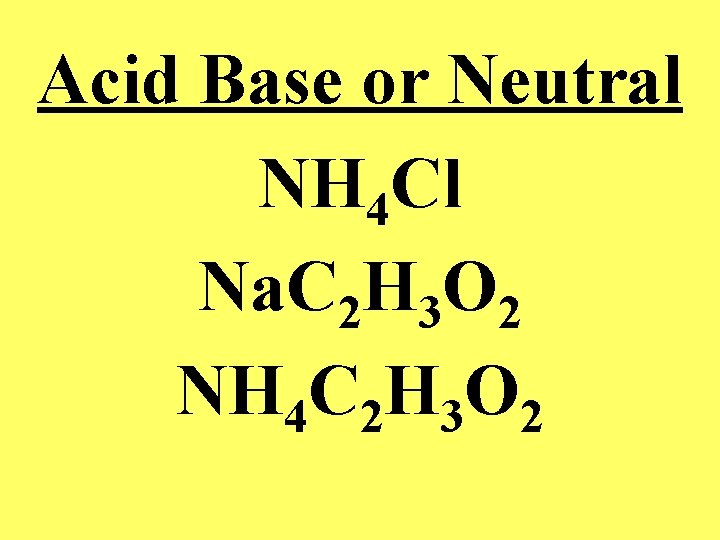
Acid Base or Neutral NH 4 Cl Na. C 2 H 3 O 2 NH 4 C 2 H 3 O 2

• Salt derived from a strong base and a strong acid will have a p. H of 7 • Salt derived from a strong base and a weak acid will have a p. H above 7 • Salt derived from a weak acid and a weak base depends upon whether the dissolved ion acts as an acid or a base as determined by the size of the Ka or Kb
![Ka and Kb NH 4 + (aq) Ka = NH 3(aq)+ H+ [H+][NH 3] Ka and Kb NH 4 + (aq) Ka = NH 3(aq)+ H+ [H+][NH 3]](http://slidetodoc.com/presentation_image_h2/98dfce0362ee91f4ef5f7b28f8aa7b3a/image-42.jpg)
Ka and Kb NH 4 + (aq) Ka = NH 3(aq)+ H+ [H+][NH 3] (aq) NH 3(aq)+ H 2 O NH 4+ (aq)+ OH-(aq) Kb = [NH 4+] [NH 4][OH- ] [NH 3] NH 4+(aq) NH 3(aq) + H+ (aq) NH 3(aq) + H 2 O(l) NH 4+(aq) + OH- (aq) H 2 O H+(aq) + OH-(aq) When two reactions are added to give a third reaction, the equilibrium constant for the third reaction is given by the product of the equilibrium constants for the two added reactions p. Ka + p. Kb = p. Kw K x K =K a b w

p. H of Salt Solutions Calculate the p. H of a 0. 01 M solution of sodium hypochlorite (Na. Cl. O) Cl. OI C E + H 2 O HCl. O + OH-
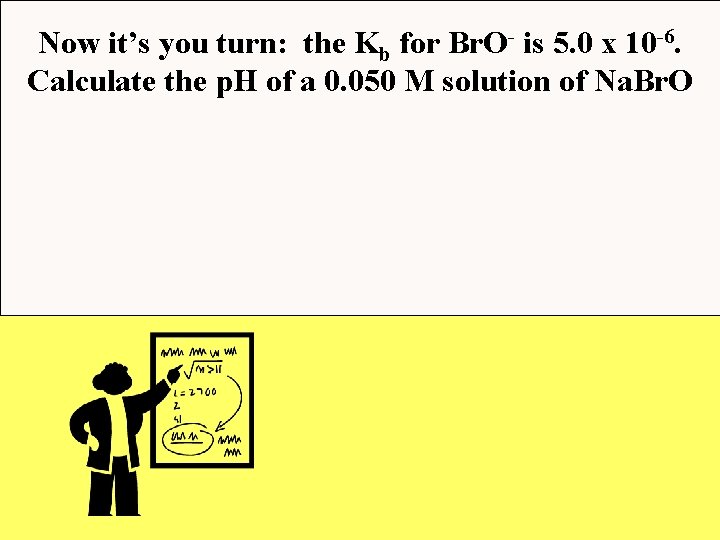
Now it’s you turn: the Kb for Br. O- is 5. 0 x 10 -6. Calculate the p. H of a 0. 050 M solution of Na. Br. O

Calculate the (a) base-dissociation constant, Kb, for the fluoride ion, if the p. Ka of HF = 3. 17 p. Ka = -log Ka 3. 17 = -log Ka Antilog -3. 17 = 6. 76 x 10 -4 Since Ka x Kb = Kw (6. 76 x 10 -4)x Kb = 1. 0 x 10 -14/ 6. 76 x 10 -4 = 1. 5 x 10 -11 or p. Ka + p. Kb = 14 so p. Kb = 10. 83 10 -10. 83 = 1. 5 x 10 -11

Calculate the p. Kb for carbonic acid (Ka = 4. 3 x 10 -7) Now it’s your turn

• Anions of polyprotic acids, such as HCO 3 -, that still have ionizable protons are capable of acting as either proton donors or acceptors depending upon the magnitudes of the Ka or Kb Predict whether the salt Na 2 HPO 4 will form an acidic or basic solution on dissolvingin water. Na 2 HPO 4 2 Na+ (aq) + HPO 4 - acting like an acid HPO 4 -(aq) + H 2 O H 3 O+ + PO 43 -(aq) HPO 4 - acting like an base HPO 4 - (aq) + H 2 O H 2 PO 42 -(aq) + K 3 = 4. 2 x 10 -13 OH-(aq) So HPO- is the conjugate base of H 2 PO 4 -. Since the K 2 of H 2 PO 4 - = 6. 2 x 10 -8 then: = 1. 0 x 10 -14 Kw = Kb = 6. 2 x 10 -8 Ka 1. 6 x 10 -7 Since Kb is larger than Ka, HPO 4 - will act like a base
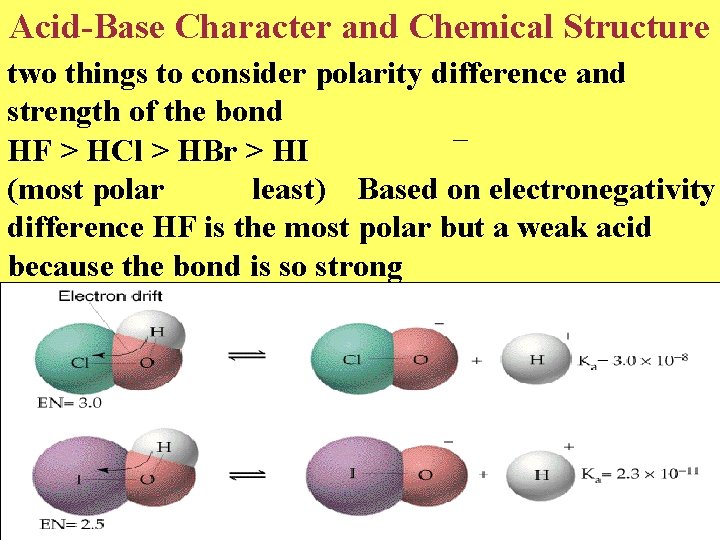
Acid-Base Character and Chemical Structure two things to consider polarity difference and strength of the bond HF > HCl > HBr > HI (most polar least) Based on electronegativity difference HF is the most polar but a weak acid because the bond is so strong


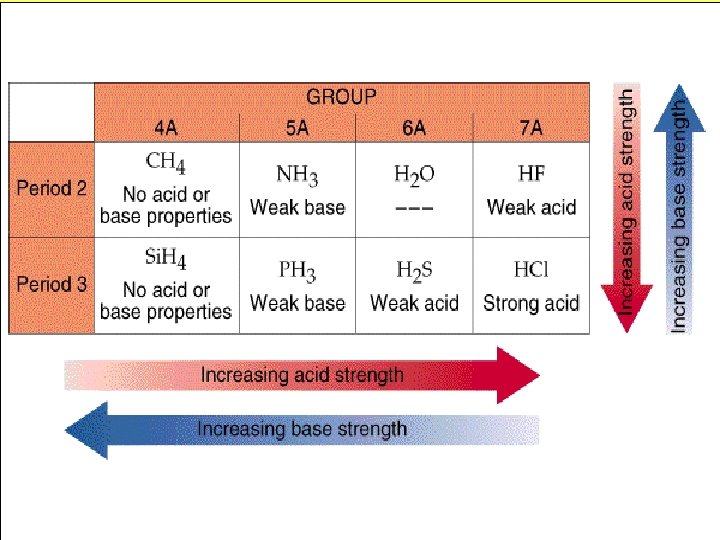

Acid strength of oxyacids The more oxygen's the stronger the acid because of the oxygen pulling the electrons towards themselves. HCl. O 4> HCl. O 3> HCl. O 2> HOCl
- Slides: 52