ACIDS AND BASES Properties of Acids q Acids

ACIDS AND BASES

Properties of Acids q Acids taste sour q Acids effect indicators q Blue litmus turns red q Methyl orange turns red q Acids have a p. H lower than 7 q Acids are proton (hydrogen ion, H+) donors q Acids react with active metals, produce H 2 q Acids react with carbonates q Acids neutralize bases

Acids you must know: Strong Acids Weak Acids Sulfuric acid, H 2 SO 4 Phosphoric acid, H 3 PO 4 Hydrochloric acid, HCl Acetic acid, HC 2 H 3 O 2 Nitric acid, HNO 3

Properties of Bases q Bases taste bitter q Bases effect indicators q Red litmus turns blue q Phenolphthalein turns purple q Bases have a p. H greater than 7 q Bases are proton (hydrogen ion, H+) acceptors q Solutions of bases feel slippery q Bases neutralize acids

Examples of Bases Ø Ø Sodium hydroxide (lye), Na. OH Potassium hydroxide, KOH Magnesium hydroxide, Mg(OH)2 Calcium hydroxide (lime), Ca(OH)2

Acid Definitions Lewis Acid Bronsted-Lowry Arrhenius acids Arrhenius Acids and Bases Acids release hydrogen ions in water. Bases release hydroxide ions in water. An acid is a substance that produces hydronium ions, H 3 O+, when dissolved in water. Bronsted-Lowry Definitions A Bronsted-Lowry acid is a proton donor; it donates a hydrogen ion, H+. A Bronsted-Lowry base is a proton acceptor; it accepts a hydrogen ion, H+. Lewis Definitions A Lewis acid is a substance than can accept (and share) an electron pair. A Lewis base is a substance than can donate (and share) an electron pair.
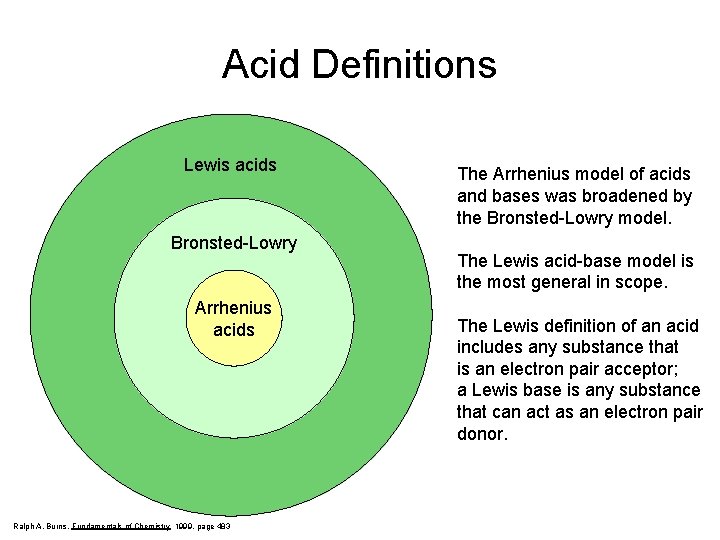
Acid Definitions Lewis acids Bronsted-Lowry Arrhenius acids Ralph A. Burns, Fundamentals of Chemistry 1999, page 483 The Arrhenius model of acids and bases was broadened by the Bronsted-Lowry model. The Lewis acid-base model is the most general in scope. The Lewis definition of an acid includes any substance that is an electron pair acceptor; a Lewis base is any substance that can act as an electron pair donor.
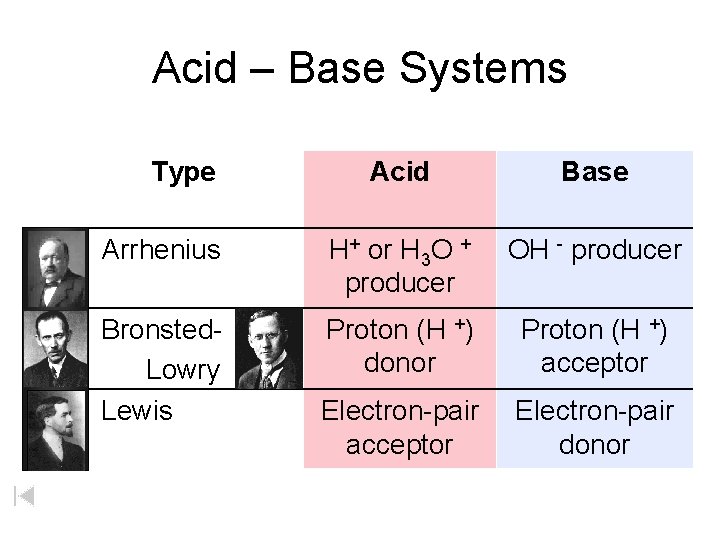
Acid – Base Systems Type Acid Base Arrhenius H+ or H 3 O + producer OH - producer Bronsted. Lowry Lewis Proton (H +) donor Proton (H +) acceptor Electron-pair donor
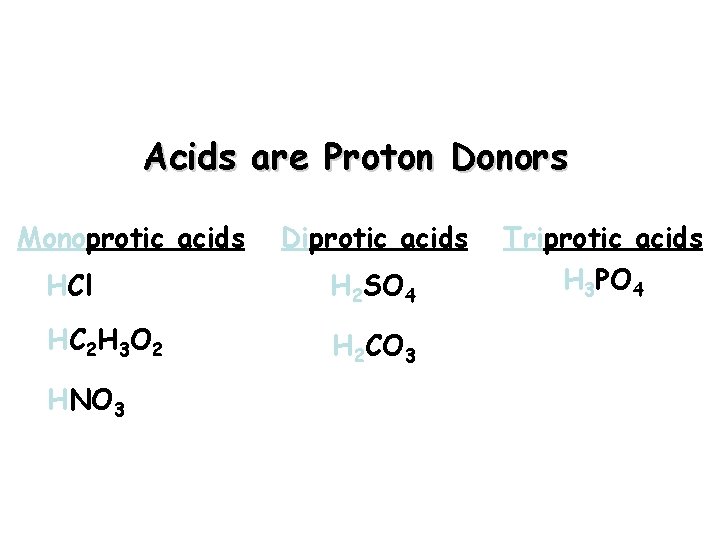
Acids are Proton Donors Monoprotic acids Diprotic acids HCl H 2 SO 4 HC 2 H 3 O 2 H 2 CO 3 HNO 3 Triprotic acids H 3 PO 4

Ionization of HCl and formation of hydronium ion, H 3 O+ H 2 O + Proton acceptor HCl Proton donor H 3 O+ + Cl-
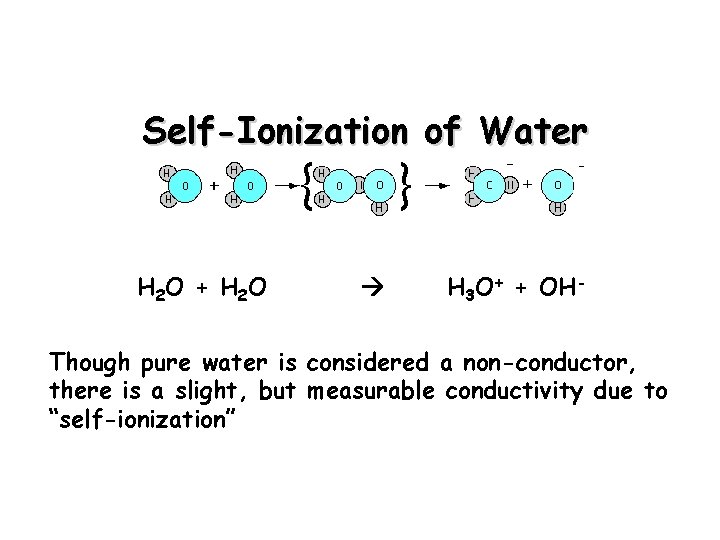
Self-Ionization of Water H 2 O + H 2 O H 3 O+ + OH- Though pure water is considered a non-conductor, there is a slight, but measurable conductivity due to “self-ionization”
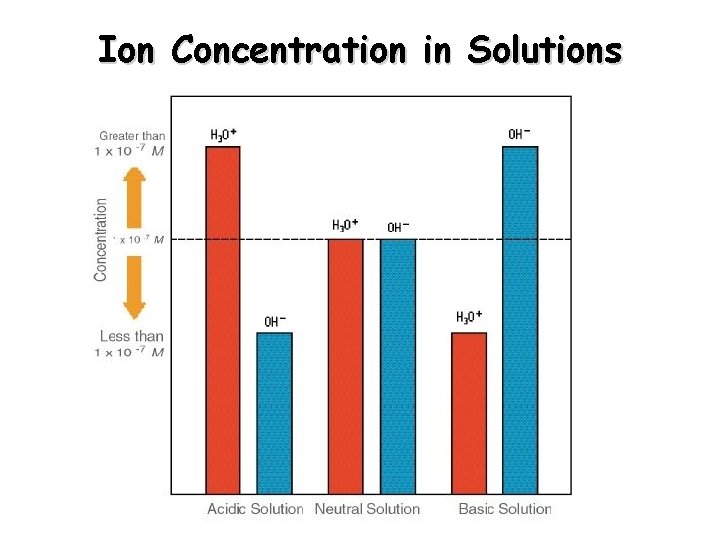
Ion Concentration in Solutions

Kw – Ionization Constant for Water In pure water at 25 C: [H 3 O+] = 1 x 10 -7 mol/L [OH-] = 1 x 10 -7 mol/L Kw is a constant at 25 C: Kw = [H 3 O+][OH-] Kw = (1 x 10 -7) = 1 x 10 -14
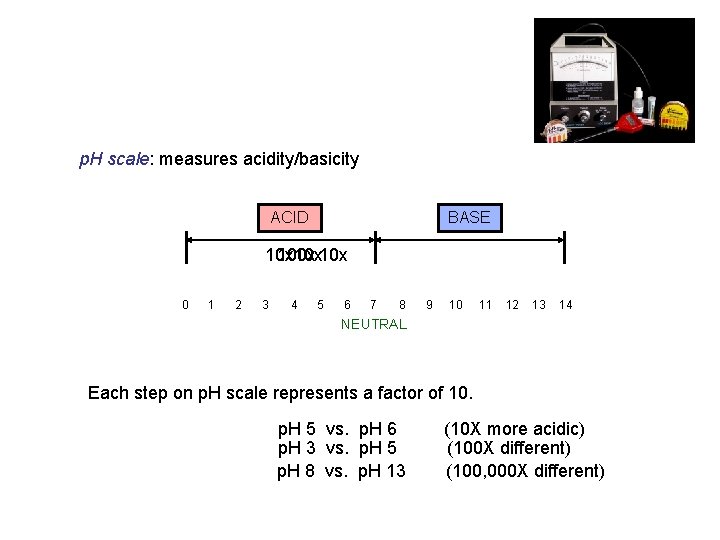
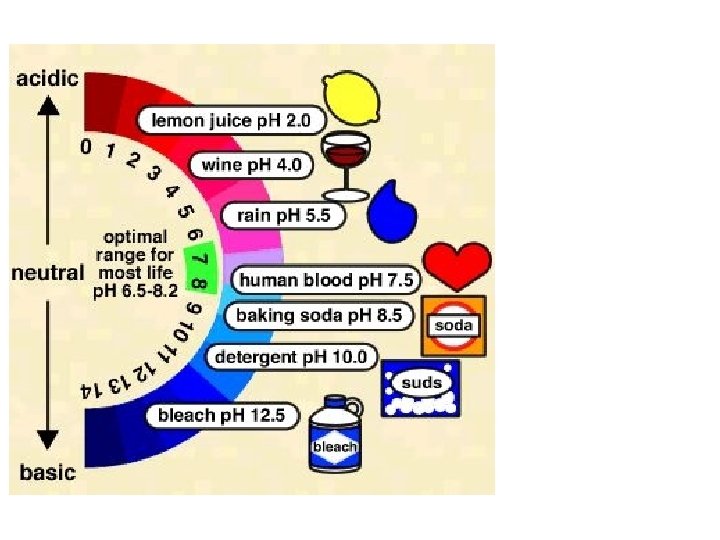
p. H scale: measures acidity/basicity ACID BASE 10 x 100 x 10 x 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 NEUTRAL Each step on p. H scale represents a factor of 10. p. H 5 vs. p. H 6 p. H 3 vs. p. H 5 p. H 8 vs. p. H 13 (10 X more acidic) (100 X different) (100, 000 X different)

H+, OH-, and p. H

Calculating p. H, p. OH p. H = -log 10(H 3 O+) p. OH = -log 10(OH-) Relationship between p. H and p. OH p. H + p. OH = 14 Finding [H 3 O+], [OH-] from p. H, p. OH [H 3 O+] = 10 -p. H [OH-] = 10 -p. OH

Strong Acids vs. Weak Acids Strong acids are assumed to be 100% ionized in solution (good proton donors). HCl H 2 SO 4 HNO 3 Weak acids are usually less than 5% ionized in solution (poor proton donors). H 3 PO 4 HC 2 H 3 O 2 Organic acids
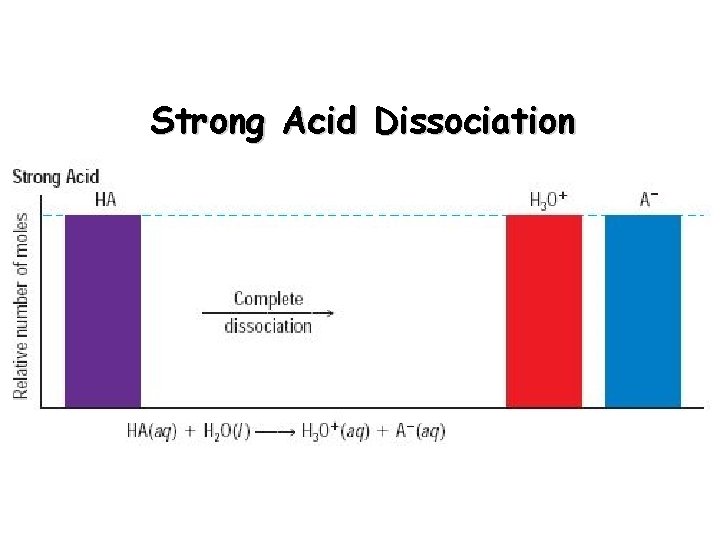
Strong Acid Dissociation
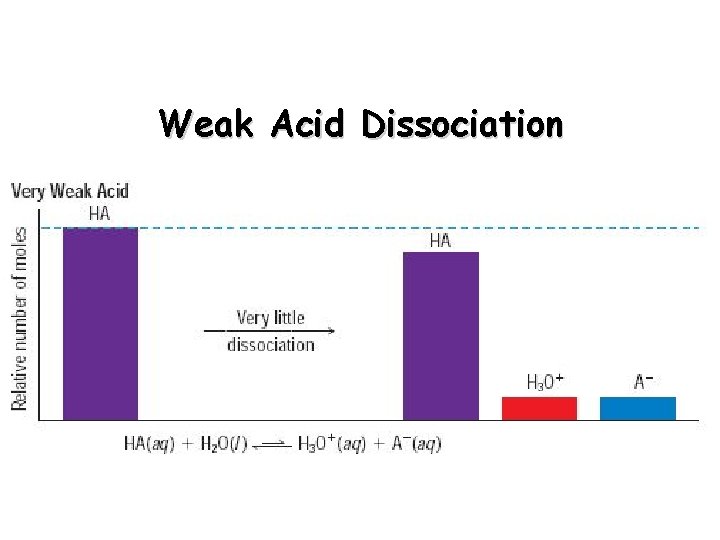
Weak Acid Dissociation

Acids Effect Indicators Blue litmus paper turns red in contact with an acid.
Acids Have a p. H less than 7

Effects of Acid Rain on Marble (calcium carbonate) George Washington: BEFORE George Washington: AFTER

Bases Effect Indicators Red litmus paper turns blue in contact with a base. Phenolphthalein turns purple in a base.
Bases have a p. H greater than 7

Bases Neutralize Acids Milk of Magnesia contains magnesium hydroxide, Mg(OH)2, which neutralizes stomach acid, HCl. 2 HCl + Mg(OH)2 Mg. Cl 2 + 2 H 2 O

Acids Neutralize Bases HCl + Na. OH Na. Cl + H 2 O Neutralization reactions ALWAYS produce a salt and water.
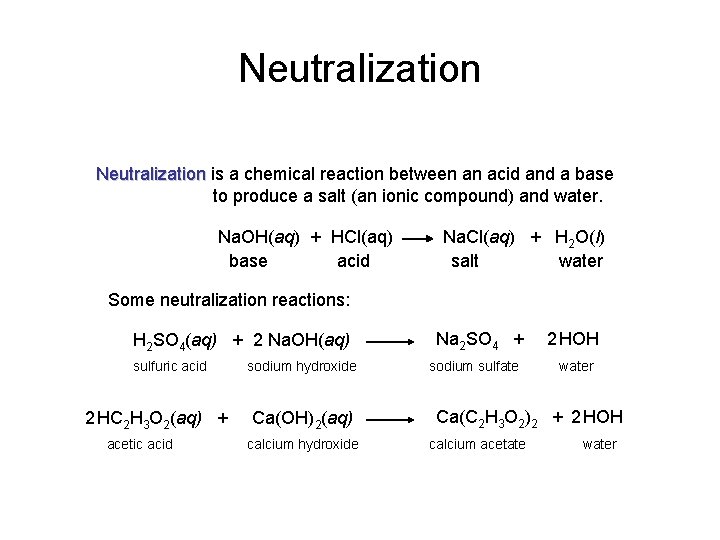
Neutralization is a chemical reaction between an acid and a base to produce a salt (an ionic compound) and water. Na. OH(aq) + HCl(aq) base acid Na. Cl(aq) + H 2 O(l) salt water Some neutralization reactions: H 2 SO 4(aq) + 2 Na. OH(aq) sulfuric acid 2 HC 2 H 3 O 2(aq) + acetic acid sodium hydroxide Ca(OH)2(aq) calcium hydroxide Na 2 SO 4 + sodium sulfate 2 HOH water Ca(C 2 H 3 O 2)2 + 2 HOH calcium acetate water

Neutralization ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic – Salts can be neutral, acidic, or basic. – Neutralization does not mean p. H = 7. Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
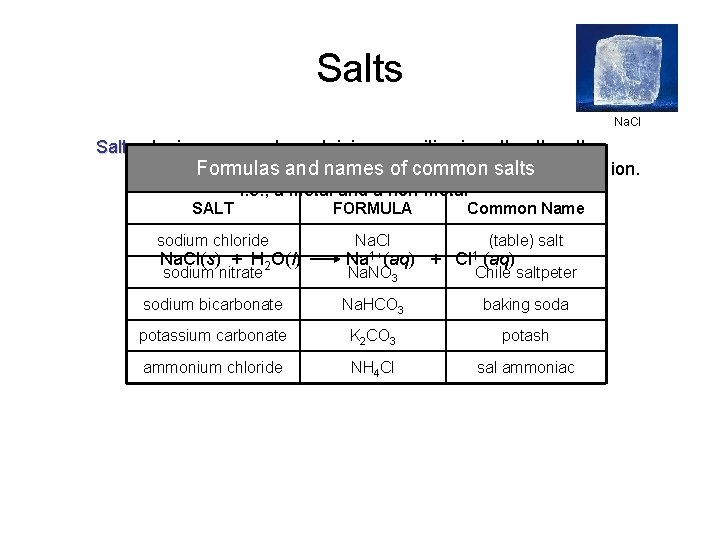
Salts Na. Cl Salts - Ionic compounds containing a positive ion other than the hydrogen ion andand a negative than salts the hydroxide ion. Formulas namesion of other common i. e. , a metal and a non-metal SALT FORMULA sodium chloride Na. Cl(s) + H 2 O(l) sodium nitrate Na 1+(aq) Na. NO 3 Common Name + (table) salt Cl 1 -(aq) Chile saltpeter sodium bicarbonate Na. HCO 3 baking soda potassium carbonate K 2 CO 3 potash ammonium chloride NH 4 Cl sal ammoniac
- Slides: 30