Acids and bases Properties of acids bases 15

Acids and bases
Properties of acids & bases

15 -1 objectives • List 5 general properties of aqueous acids and bases • Name common binary acids and oxyacids • List 5 common industrial and laboratory acids and give 2 properties of each • Define Arrhenius acids and bases • Explain the difference between strong and weak acids and bases 9/18/2021 3

Properties of Acids 1. Taste sour 2. Change the color of p. H indicators (turn blue litmus paper red) 3. React with active metals to produce hydrogen gas 4. React with bases to produce salts and water 5. Conduct an electric current 9/18/2021 4

Properties of Bases 1. Taste bitter 2. Change the color of acid-base indicators (turn red litmus paper blue) 3. Feel slippery 4. React with acids to produce salts and water 5. Conduct an electric current 9/18/2021 5

Commonly Used Acids Sulfuric acid (H 2 SO 4) dehydrating agent; batteries; metals, paper; dyes, paints & detergents Nitric acid (HNO 3) stains proteins yellow; very smelly; explosives Phosphoric acid (H 3 PO 4) fertilizer; ceramics; detergents; flavoring Hydrochloric acid (HCl) “pickling” metals; food processing; pools; cleaning masonry Acetic acid (CH 3 COOH) smelly; vinegar; freezes at 17 o. C; plastics; fungicide 9/18/2021 6

Arrhenius Acid & Bases Arrhenius acid – increases the concentration of hydrogen ions in aqueous solution HNO 3(l) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq) The H 3 O+ ion is called the hydronium ion Arrhenius base – increases the concentration of hydroxide ions in aqueous solutions 9/18/2021 7

Strong Acids Strong acids ionize completely in aqueous solutions and are strong electrolytes Examples: H 2 SO 4 HCl HNO 3 HBr HI 9/18/2021 8

Weak Acids Do not ionize completely and are weak electrolytes Examples: H 3 PO 4 CH 3 COOH H 2 CO 3 H 2 S HCN Clip 9/18/2021 9

9/18/2021 10

Strong Bases A strong base completely dissociates in water to produce OH- ions Solutions are called alkaline Examples: Na. OH KOH 9/18/2021 11

Weak Bases Weak bases do not dissociate completely in solution Example: NH 3 C 6 H 5 NH 2 (aniline) Clip 9/18/2021 12

9/18/2021 13

Acid-Base Theories

15 -2 Objectives • Define and recognize Brønsted-Lowry acids and bases • Define a Lewis acid and a Lewis base • Name compounds that are acids under the Lewis definition but are not acids under the Brønsted-Lowry definition 9/18/2021 15
Brønsted-Lowry Acids • A molecule or ion that is a proton donor is Brønsted-Lowry acid Example: HCl dissolved in ammonia HCl dissolved in water H 2 O dissolved in ammonia 9/18/2021 16

Brønsted-Lowry Bases • A molecule or ion that accepts a proton is a Brønsted-Lowry base Example: HCl dissolved in ammonia 9/18/2021 17
Monoprotic Acids Monoprotic acids can donate only one proton Example: + HCl(g) + H 2 O(l) H 3 O (aq) + Cl (aq) 9/18/2021 18
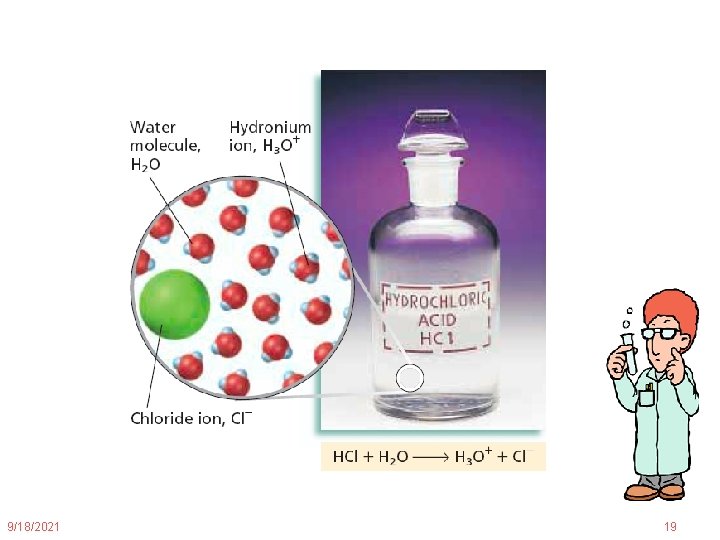
9/18/2021 19

Polyprotic Acids Polyprotic acids can donate more than one proton Example: + H 2 SO 4(l) + H 2 O(l) H 3 O (aq) + HSO 4 (aq) + H 2 O(l) H 3 O (aq) + SO 42 -(aq) This is a diprotic acid (2 ionizations) 9/18/2021 20

9/18/2021 21

Lewis Acids and Bases An atom, ion, or molecule that accepts an electron pair to form a covalent bond is a Lewis acid An atom, ion, or molecule that donates an electron pair to form a covalent bond is a Lewis base 9/18/2021 22

Acid-base reactions

15 -3 Objectives • Describe a conjugate acid, conjugate base, and amphoteric compound • Explain the process of neutralization • Explain how acid rain damages marble surfaces 9/18/2021 24
Amphoteric • A molecule or ion that can be classified as an acid or a base. • Ex – HSO 4 - 9/18/2021 25

Conjugate Acids & Bases The species that remains after a Brønsted. Lowry acid has given up a proton is the conjugate base of that acid. Example: 9/18/2021 26

Conjugate Acids & Bases The species that is formed when a Brønsted. Lowry base gains a proton is the conjugate acid of that base. Example: 9/18/2021 27
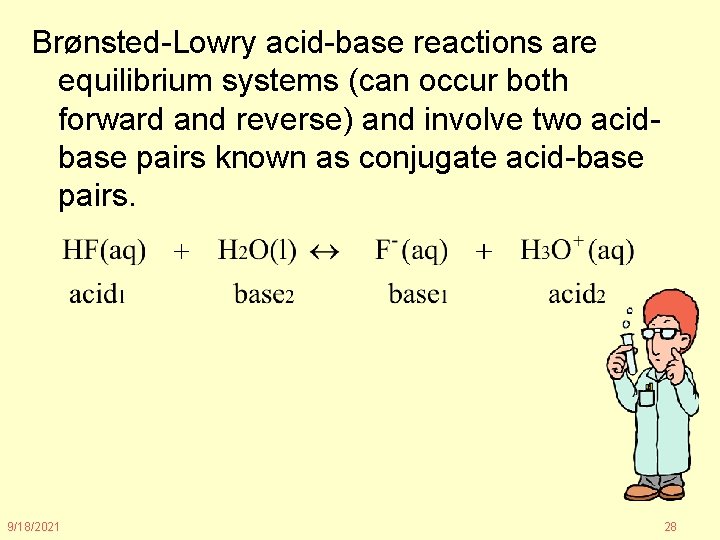
Brønsted-Lowry acid-base reactions are equilibrium systems (can occur both forward and reverse) and involve two acidbase pairs known as conjugate acid-base pairs. 9/18/2021 28

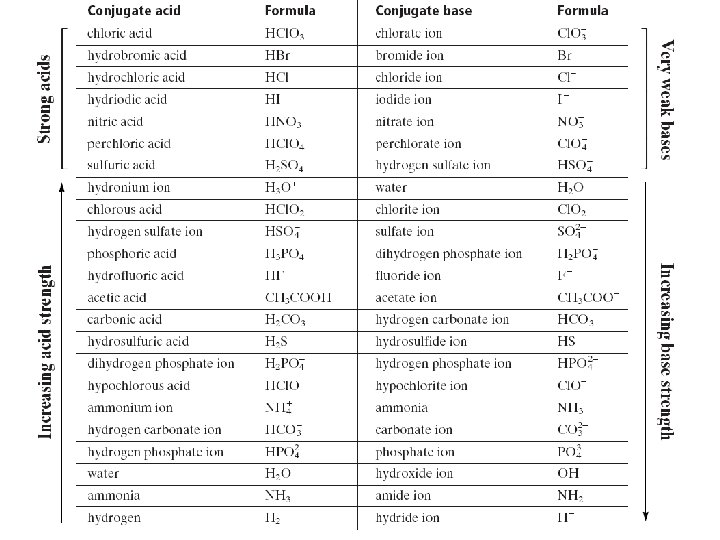
Strength of Conjugate Acids and Bases The stronger the acid is the weaker its conjugate base The stronger the base, the weaker its conjugate acid 9/18/2021 29
9/18/2021 30

Neutralization Reactions Are reactions between hydronium ions and hydroxide ions to form water Another product is a salt – a cation from an acid an anion from a base 9/18/2021 31
- Slides: 31