Acids and Bases Properties and Theories Acids in

Acids and Bases Properties and Theories

Acids in Ancient Times Latin “acidus” (sour) was the original name for vinegar Hydrochloric acid (aka muriatic acid) was discovered around the year 800 by Islamic alchemist Jabir Ibn Hayyan (Gaber). Nitric acid was known to the alchemists as aqua fortis Aqua regia, a mixture of nitric and hydrochloric acids, contributed to the effort of alchemists tried to find the philosopher's stone

Bases in Ancient Times Milk of magnesia—magnesium hydroxide l. Used as antacid, laxative Lye—sodium hydroxide lused in tanning, cloth production, and soapmaking Potash—crude mixture of potassium carbonate and potassium hydroxide l. Used as fertilizer Limewater—solution of calcium hydroxide or calcium carbonate l. Used as a test for CO 2, in calamine lotion, and as an antacid

How do acids behave? Sour taste Acids turn blue litmus paper red (but have no effect on red litmus paper)

Properties of Acids react with certain metals to release hydrogen gas Mg + HCl Mg. Cl 2 + H 2 Form electrolytes in aqueous solutions l l Ionize in solution support electric currents in water

How do bases behave? Bases turn red litmus paper blue (but have no effect on blue litmus paper) Bitter taste and slippery feel Electrolytes in solution Turns phenolphthalein pink

When acids and bases mix Neutralization reaction occurs Acid + base water + salt First studied quantitatively by German chemist Jeremias Richter
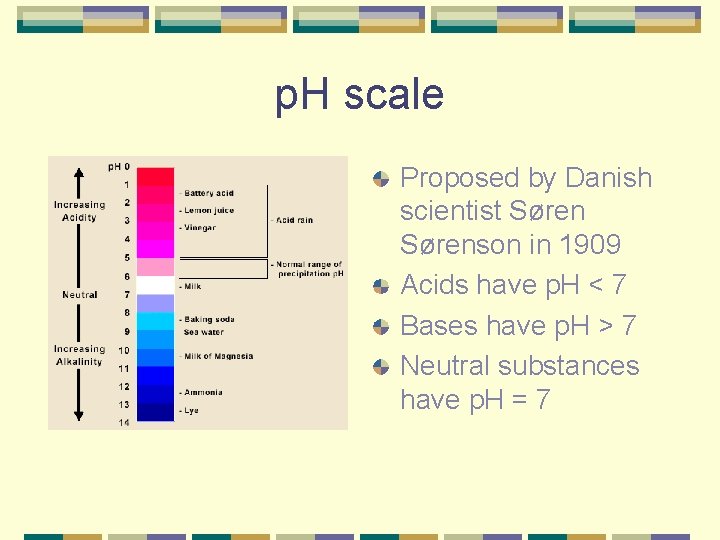
p. H scale Proposed by Danish scientist Sørenson in 1909 Acids have p. H < 7 Bases have p. H > 7 Neutral substances have p. H = 7

Operational Definitions Acids l l p. H < 7 React with metals to release H 2(g) Sour taste Turn litmus paper red Bases l l l p. H > 7 Turn phenolphthalein pink Turns litmus paper blue Bitter taste Slippery feel

1 st Acid-Base Theory Proposed by Swedish chemist Svante Arrhenius in 1887 1903 Nobel Prize in Chemistry Studied dissociation of substances in solution

Arrhenius Theory Acids release H+ ions in aqueous solution l Examples of Arrhenius acids l l l HNO 3, HCl, : monoprotic acids H 2 SO 4 : diprotic acid H 3 PO 4: triprotic acid Bases produce OH- ions in aqueous solution l Examples of Arrhenius bases l Na. OH, KOH, Mg(OH)2

What about ammonia? NH 3 Turns litmus paper blue Neutralizes acid Behaves as a base Arrhenius theory doesn’t explain it well

Brønsted-Lowry Theory Johannes Brønsted—Danish chemist Thomas Lowry—English chemist Both independently proposed this theory in 1923 Refined definition of bases Acid definition very similar to Arrhenius’ l All Arrhenius acids are also B-L acids
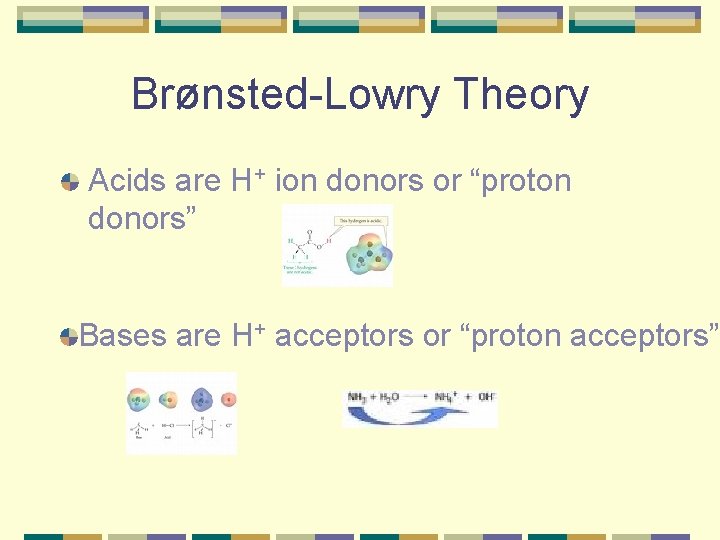
Brønsted-Lowry Theory Acids are H+ ion donors or “proton donors” Bases are H+ acceptors or “proton acceptors”

Conjugate Acid-Base Pairs Acids and bases occur in conjunction Conjugate acid-base pairs l Differ only by a proton

Amphiprotic Substances Some substances can act as an acid and as a base Also called “amphoteric”

Gilbert Lewis Massachusetts native and MIT chemistry professor Lewis structures Lewis acid-base theory an extension of his theory of electron pairs
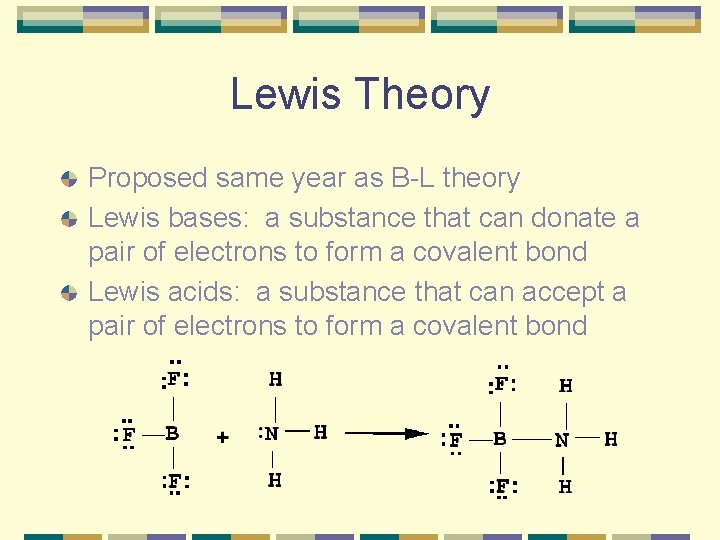
Lewis Theory Proposed same year as B-L theory Lewis bases: a substance that can donate a pair of electrons to form a covalent bond Lewis acids: a substance that can accept a pair of electrons to form a covalent bond
- Slides: 18