Acids and Bases Power Point 5 1 Classification

Acids and Bases Power. Point 5. 1

Classification of Elements How many different ways are individual elements categorized? ØMetals, non-metals, and metalloids. ØTransition metals grouped together. ØLanthanoids and Actinoids, usually separated from the rest of the table. ØBy Group, alkali metals, halogens, noble gases, and so on. ØBy Period, indicating the number of shells of an element’s neutral atom. ØDiatomic elements, H 2, N 2, O 2, S 2, F 2, Cl 2, Br 2, I 2

Classification of Compounds How many different ways are individual compounds categorized? Ø Ionic compounds versus Covalent compounds Ø Acids and bases

General Differences between Acids and Bases • • • Acids Sour taste Corrosive, corrode metals Both acids and bases Formula generally • Corrosive begin with H or CH • Conduct electricity Produces H+ p. H < 7 • • Bases Bitter taste Slippery feel Caustic less reactive with metals Formula generally ends with OH Produces OHp. H > 7

p. H Scale Ø A scale from 0 to 14 utilized to indicate how acidic, or how basic, a substance is. Ø A change in one unit is actually a 10 times increase in acidity or basicity. acidic basic or alkaline

Acid-Base Indicators Ø Acid-Base indicators are chemical that change colour depending on the p. H of a solution. Ø They are often utilized to determine the p. H of a solution or to determine the concentration of H+ or OH-.
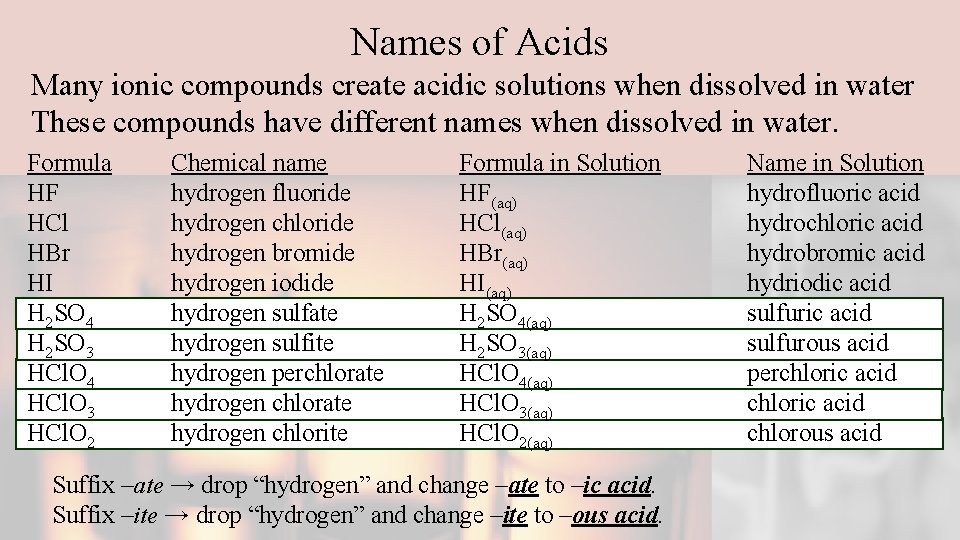
Names of Acids Many ionic compounds create acidic solutions when dissolved in water These compounds have different names when dissolved in water. Formula HF HCl HBr HI H 2 SO 4 H 2 SO 3 HCl. O 4 HCl. O 3 HCl. O 2 Chemical name hydrogen fluoride hydrogen chloride hydrogen bromide hydrogen iodide hydrogen sulfate hydrogen sulfite hydrogen perchlorate hydrogen chlorite Formula in Solution HF(aq) HCl(aq) HBr(aq) HI(aq) H 2 SO 4(aq) H 2 SO 3(aq) HCl. O 4(aq) HCl. O 3(aq) HCl. O 2(aq) Suffix –ate → drop “hydrogen” and change –ate to –ic acid. Suffix –ite → drop “hydrogen” and change –ite to –ous acid. Name in Solution hydrofluoric acid hydrochloric acid hydrobromic acid hydriodic acid sulfurous acid perchloric acid chlorous acid
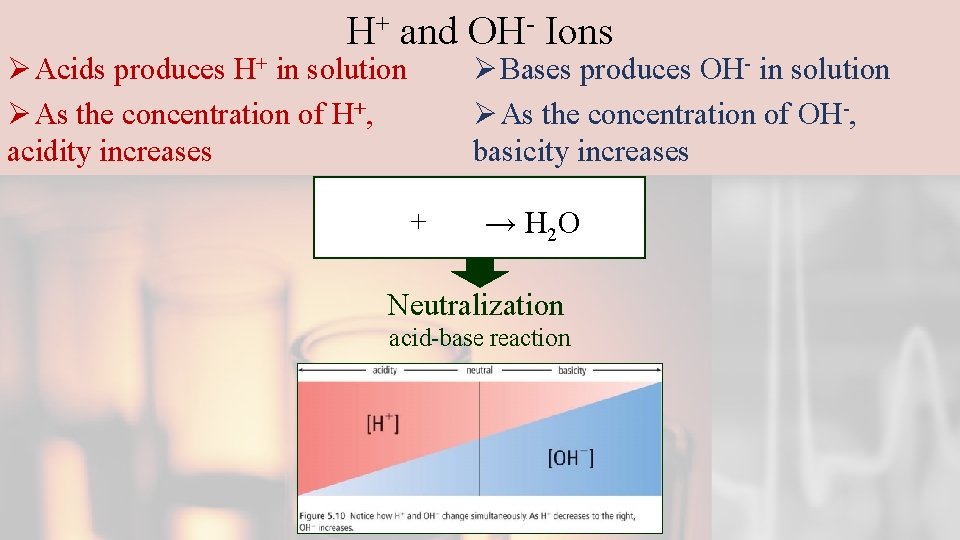
H+ and OH- Ions Ø Acids produces H+ in solution Ø As the concentration of H+, acidity increases Ø Bases produces OH- in solution Ø As the concentration of OH-, basicity increases + → H 2 O Neutralization acid-base reaction
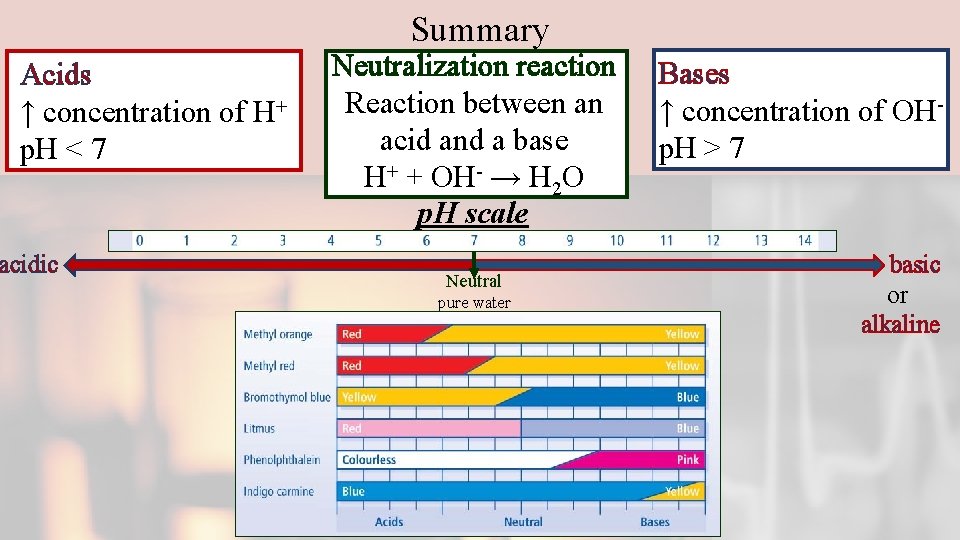
Summary Acids ↑ concentration of H+ p. H < 7 acidic Neutralization reaction Reaction between an acid and a base H+ + OH- → H 2 O p. H scale Neutral pure water Bases ↑ concentration of OHp. H > 7 basic or alkaline
- Slides: 9