ACIDS AND BASES KONICHEK III ACIDS BASES AND

ACIDS AND BASES KONICHEK

• III ACIDS BASES AND PH • A. acids- this is any substance which produces a hydronium ion when in dissolved in water. ( HCl) • 1. Hydronium ion is H 3 O or H+ ion • 2. characteristics of acids • a. taste sour • b. donate protons • c. turn blue litmus red • 3. strong acids ionize completely- Nitric acid. • 4. Weak acids only partially ionize the water molecule

• B. Bases- Any substance which produces OH- (hydroxide ion) ions when dissolved in water. ( Na. OH) • 1. characteristics of bases • a. taste bitter • b, proton acceptor • c, taste bitter • d. feel slippery •

• C. Measuring acidity-Measure the number of hydronium ions in solution • 1. p. H scale- This is a scale which measures the hydronium ion concentration. p. H = -log[H+] • a. a logarithmic scale as -Log {H+} goes from 100 to 1. 0 -14 • 1. 10 -4 has a p. H of 4, so p. H goes from 0 to 14, 0 very acidic, and • 14 basic
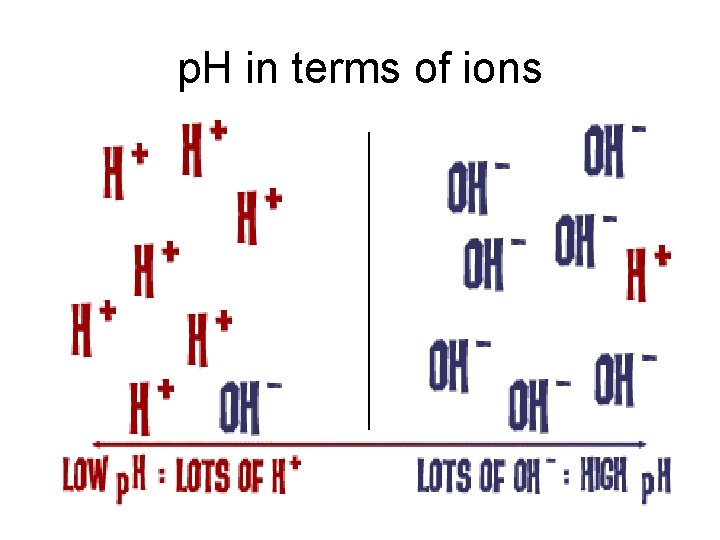
p. H in terms of ions

• Here a couple of definitions you should know: -Acid: A solution that has an excess of H+ ions. It comes from the Latin word acidus that means "sharp" or "sour". -Base: A solution that has an excess of OH- ions. Another word for base is alkali. -Aqueous: A solution that is mainly water. Think about the word aquarium. AQUA means water. -Strong Acid: An acid that has a very low p. H (0 -4). -Strong Base: A base that has a very high p. H (10 -14). -Weak Acid: An acid that only partially ionizes in an aqueous solution. That means not every molecule breaks apart. They usually have a p. H close to 7 (3 -6). -Weak Base: A base that only partially ionizes in an aqueous solution. That means not every molecule breaks apart. They usually have a p. H close to 7 (8 -10). -Neutral: A solution that has a p. H of 7. It is neither

• D, Neutralization reactions- a reaction which a hydronium ion and a hydroxide ion combine to form a water molecule. • 1. Strong acids and strong bases reaction to form salt water • a. HCl + Na. OH----> H 2 O+ Na. Cl • 1. double replacement reaction. • 2. Not all acid and base reactions neutralize each other, weak base and strong acids, so it takes more weak acid to neutralize a strong base.

• • • • • IV ACIDS AND BASES IN THE HOME A. Cleaning products 1. soap- a reaction between a fatty acid and a base a. fatty acid radical dissolves greases and the COO- allows it to dissolve in water. 2. detergents- use the sulfate radical rather than the carboxylate group a, chains come from petroleum distillates rather than animal fats 3. ammonia- ammonia gas is dissolved in water forming a basic solution a. hydroxide ions react with greasy dirt causing the dirt and grease to emulsify in water, so it can be drained away. B. Disinfectants-bleach( Na. Cl. O 2 - sodium chlorite, or sodium hyperchlorite(Na. Cl. O) 1. kills harmful bacteria C. antacids- Neutralizes the acid in the stomach 1. calcium hydroxide, magnesium hydroxide D. Acids and bases in the kitchen 1. Vinegar- acetic acid 2. baking soda- sodium hydrogen carbonate( base) 3. drain cleaners potassium hydroxide- KOH
- Slides: 8