Acids and Bases Johannes N Bronsted Thomas M

Acids and Bases Johannes N. Bronsted Thomas M. Lowry 1879 -1947. 1874 -1936. Both independently developed Bronsted-Lowry theory of acids and bases. 1

Acids and Bases: A Brief Review Classical Acids: Taste sour Donate H+ (called “H-plus” or “proton”) (Arrhenius theory) Turn litmus red Generally form from H-Z, where Z = nonmetal Classical Bases: Taste bitter and feel soapy/slippery Donate OH- (called “O-H-minus” or “hydroxide”) (Arrhenius theory) Turn litmus blue Generally form from MOH, where M = metal Neutralization: Acid + Base Salt + water H-Z + MOH MZ + HOH H+ in water is actually in the form of H 3 O+, “hydronium” 2

Acids & Bases Acids and bases are special kinds of electrolytes. Like all electrolytes they break up into charged particles. What sets them apart from each other, and other electrolytes is the way that they break up. If a particle originally made up of ions (therefore is charged) breaks up to form ions in water. . . it is called dissociation Na. OH → Na+ + OH If a particle originally not charged (molecular) breaks up to form ions in water …it is called ionization HCl→ H+ + Cl-

Practice Identify each of the following as acids/bases/salts: 1. 2. 3. 4. 5. HC 2 H 3 O 2 K 2 SO 4 KOH Li. OH HNO 3 Acid HC 2 H 3 O 2 H+ + C 2 H 3 O 2 Salt K 2 SO 4 2 K+ + SO 42 Base KOH K+ + OHBase Li. OH Li+ + OHAcid HNO 3 H+ + NO 3 -

Strong/Weak Acids can be either strong electrolytes or weak electrolytes. Strong acids (such as HCl) completely break up into their ions: HCl (aq) H+(aq) + Cl-(aq) Weak acids (such as HC 2 H 3 O 2) only partially break up into their ions: HC 2 H 3 O 2 H+ (aq) + C 2 H 3 O 2 -(aq) Weak acids don’t completely break up because they go to equilibrium!
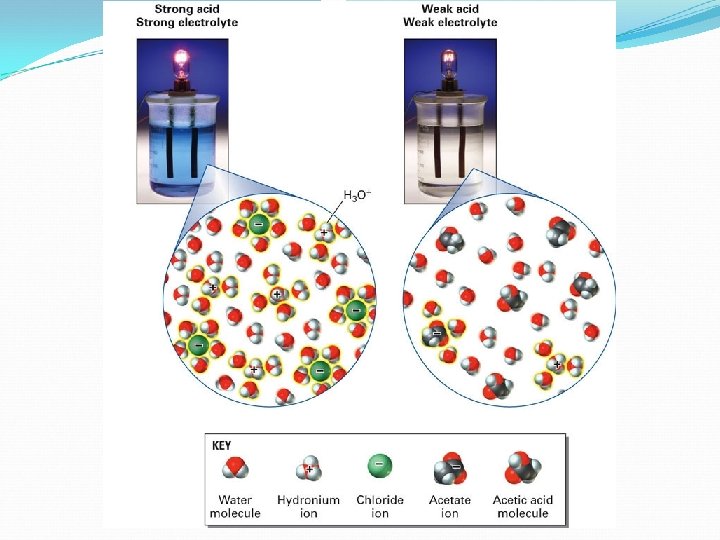

Strong/Weak Bases can be either strong electrolytes or weak electrolytes. Strong bases (such as Na. OH) completely break up into their ions: Na. OH (aq) Na+(aq) + OH-(aq) Weak bases (such as NH 3) only partially break up into their ions: NH 3 (aq) + H 2 O NH 4+ (aq) + OH-(aq) Weak bases don’t completely break up because they go to equilibrium!

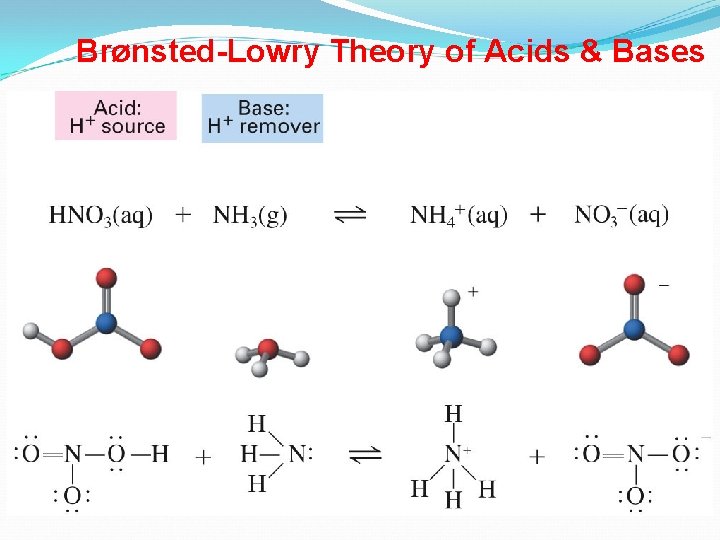
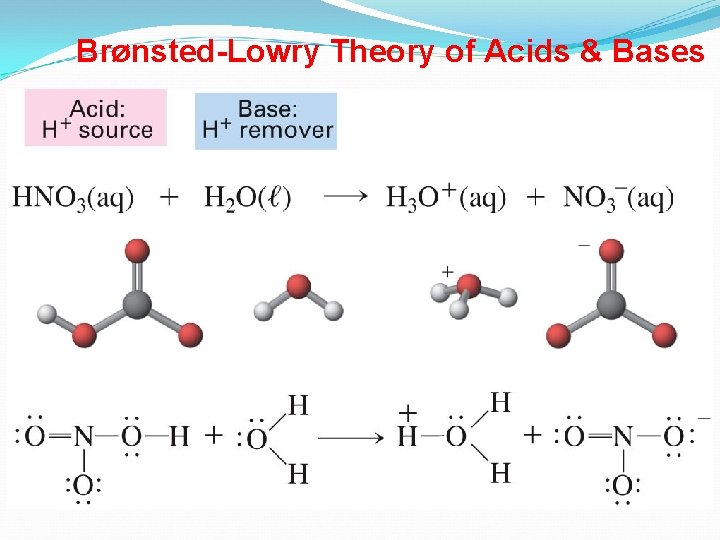
Bronsted-Lowry Definitions Bronsted and Lowry felt that this was too limiting, since there are many non-aqueous systems (no water is present). They came up with the following definitions for acids and bases. An acid is a proton (H+ ion) donor A base is a proton acceptor

Brønsted-Lowry Acids and Bases Proton Transfer Reactions • Brønsted-Lowry acid/base definition: acid donates H+ base accepts H+. • Brønsted-Lowry base does not need to contain OH-. • Consider HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl-(aq): – HCl donates a proton to H 2 O. Therefore, HCl is an acid. – H 2 O accepts a proton from HCl. Therefore, H 2 O is a base. • Water can behave as either an acid or a base. • Amphoteric substances can behave as acids and bases. 9

Relationship between the two models Arrhenius Acids/Bases only exist in water solutions. Bronsted. Lowry Acids/Bases can exist when no water is present All Arrhenius acids and bases can also be classified as Bronsted. Lowry acids and bases. Bronsted/Lowry theory is better at predicting

An example In the reaction below there are no Arrhenius acids or bases present (because no hydronium ions or hydroxide ions are formed). However, the HCl is acting as a Bronsted. Lowry acid because it is giving a H+ ion to the NH 3 (which is acting as a H+ ion acceptor - a base)

HCl as a proton donor Consider the following reaction: Since the HCl gives up a H+ ion to the water it is acting as a Bronsted-Lowry acid. In the process of donating the proton it also forms a hydronium ion, and that makes it an Arrhenius acid as well.

Water as a base But what does that make the water molecule? Since the water molecule is accepting the H+ ion, it is acting as a Bronsted-Lowry base. Since there is no hydroxide ion (OH-) formed, the water is not acting as an Arrhenius base in this reaction.

Ammonia as a base Let’s look at another example: Here the ammonia molecule is accepting a H+ ion and therefore is acting as an Bronsted-Lowry base. However, in the process of reacting with the water it is also forming a hydroxide ion. That makes the ammonia an Arrhenius base as well.

But what about the water? Since the water is giving up a H+ ion, it is acting as a Bronsted-Lowry acid. Since it does not form hydronium ions, it is NOT acting an Arrhenius acid.

Amphiprotic Sometimes a molecule can donate a proton (act as an acid) and sometimes it can accept a proton (act as a base). Molecules that have this ability to act as both an acid and a base are called amphoteric or amphiprotic. Water is the most common example of an amphoteric substance.

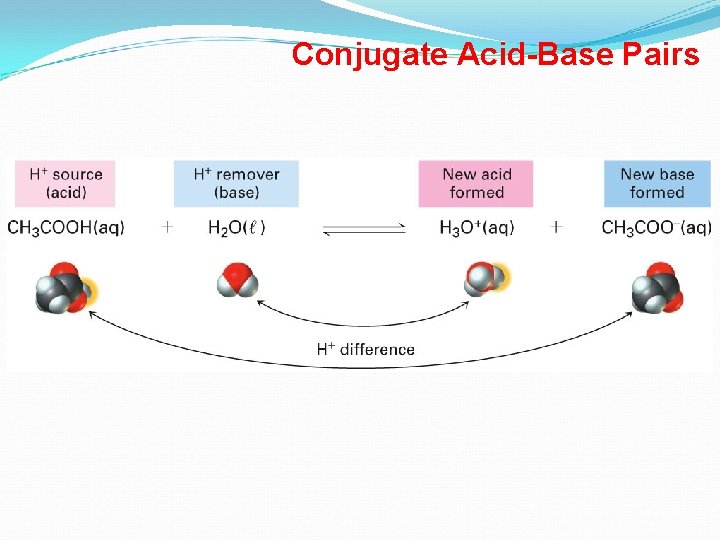
Brønsted-Lowry Acids and Bases Conjugate Acid-Base Pairs • Whatever is left of the acid after the proton is donated is called its conjugate base. • Similarly, whatever remains of the base after it accepts a proton is called a conjugate acid. • Consider –After H 2 O (base) gains a proton it is converted into H 3 O+ (acid). Therefore, H 2 O and H 3 O+ are conjugate acid-base pairs. –After HCl (acid) loses its proton it is converted into Cl- (base). Therefore HCl and Cl- are conjugate acid-base pairs. • Conjugate acid-base pairs differ by only one proton. 17
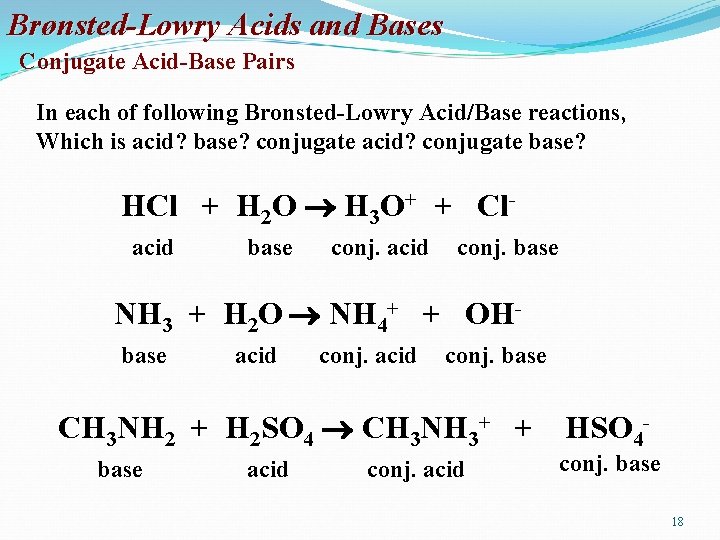
Brønsted-Lowry Acids and Bases Conjugate Acid-Base Pairs In each of following Bronsted-Lowry Acid/Base reactions, Which is acid? base? conjugate acid? conjugate base? HCl + H 2 O H 3 O+ + Clacid base conj. acid conj. base NH 3 + H 2 O NH 4+ + OHbase acid conj. base CH 3 NH 2 + H 2 SO 4 CH 3 NH 3+ + base acid conj. acid HSO 4 - conj. base 18

Reality check For each of the following reactions identify any Bronsted. Lowry acids and bases. acid base HNO 3 + H 2 O H 3 O+ + NO 3 acid base HNO 3 + NH 3 NH 4+ + NO 3 base S 2+ Hacid O HS + OH 2 acidbase HS + OH S 2 - + H-OH base acid HS + HCl H 2 S + Cl. Are any of the substances above amphoteric?
![The Autoionization of Water In pure water at 25 o. C, [H 3 O+][OH-] The Autoionization of Water In pure water at 25 o. C, [H 3 O+][OH-]](http://slidetodoc.com/presentation_image_h2/463c2f5e5bf96a2918cf1cbc6e00d772/image-20.jpg)
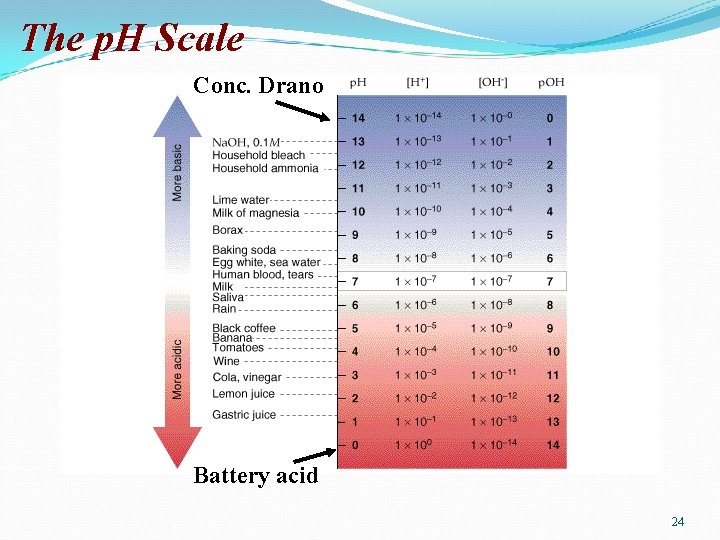
The Autoionization of Water In pure water at 25 o. C, [H 3 O+][OH-] = 1 x 10 -14 and also, [H 3 O+] = [OH-] = 1 x 10 -7 (From now on, for simplification, let’s use the abbreviation: [H+] = [H 3 O+] which means [H]+ = [OH-] = 1 x 10 -7 We define p. H = -log [H+] and p. OH = -log [OH-] In pure water at 25 o. C, p. H = p. OH = 7. 00 p. H + p. OH = 14 p. Kw = 14 Acidic solutions have p. H < 7. 00 Basic solutions have p. H > 7. 00 20
Brønsted-Lowry Theory of Acids & Bases
Brønsted-Lowry Theory of Acids & Bases
Conjugate Acid-Base Pairs
The p. H Scale Conc. Drano Battery acid 24
![The p. H Scale (a) For [H+] = 3. 4 x 10 -5 M, The p. H Scale (a) For [H+] = 3. 4 x 10 -5 M,](http://slidetodoc.com/presentation_image_h2/463c2f5e5bf96a2918cf1cbc6e00d772/image-25.jpg)
The p. H Scale (a) For [H+] = 3. 4 x 10 -5 M, calculate p. H, p. OH and [OH-] p. H = 4. 47, p. OH = 9. 53, [OH-] = 2. 95 x 10 -10 (b) For [OH-] = 4. 4 x 10 -3 M, calculate p. H, p. OH, and [H+] p. OH = 2. 36, p. H = 11. 64, [H+] = 2. 27 x 10 -12 (c) For p. H= 8. 9, calculate, p. OH, [H+], [OH-] p. OH = 5. 1, [H+] = 1. 26 x 10 -9, [OH-] = 7. 94 x 10 -6 (d) For p. OH= 3. 2, calculate p. H, [H+], [OH-] p. H = 10. 8, [H+] = 1. 58 x 10 -11, [OH-] = 6. 3 x 10 -4 The p. H meter is the most accurate way to measure p. H values of solutions. 25

2. Strong Acids Calculate the p. H of 0. 2 M HCl. p. H = -log[H+] = -log[0. 2] = 0. 70 3. Strong Bases Calculate the p. H of 0. 2 M Na. OH. p. OH = -log[OH-] = -log[0. 2] = 0. 70 p. H = 14 – 0. 70 = 13. 30 Calculate the p. H of 0. 2 M Ba(OH)2. p. OH = -log[OH-] = -log[0. 4] = 0. 40 p. H = 13. 60 26

p. H A measure of the hydronium ion The scale for measuring the hydronium ion concentration [H 3 O+] in any solution must be able to cover a large range. A logarithmic scale covers factors of 10. The “p” in p. H stands for log. A solution with a p. H of 1 has [H 3 O+] of 0. 1 mol/L or 10 -1 A solution with a p. H of 3 has [H 3 O+] of 0. 001 mol/L or 10 -3 A solution with a p. H of 7 has [H 3 O+] of 0. 0000001 mol/L or 10 -7 p. H = - log [H 3 O+]

The p. H scale ranges from 1 to 10 -14 mol/L or from 1 to 14. p. H = - log [H 3 O+] 1 2 3 4 5 6 7 8 9 10 11 12 13 14 acid neutral base
![Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+] Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+]](http://slidetodoc.com/presentation_image_h2/463c2f5e5bf96a2918cf1cbc6e00d772/image-29.jpg)
Manipulating p. H Algebraic manipulation of: p. H = - log [H 3 O+] allows for: [H 3 O+] = 10 -p. H If p. H is a measure of the hydronium ion concentration the same equations could be used to describe the hydroxide (base) concentration. [OH-] = 10 -p. OH = - log [OH-] thus: p. H + p. OH = 14 ; the entire p. H range!
- Slides: 29