Acids and Bases Jeopardy Acid Calculations Base Calculations
Acids and Bases Jeopardy Acid Calculations Base Calculations Concentration of species Titration Curves Pot Luck 100 100 100 200 200 200 300 300 300 400 400 400 500 500 500 Final Jeopardy

Help (1) Save a duplicate of this template. (2) Enter all answers and questions in the normal view. (view/normal) (3) Change the category headings in the normal view (view/normal) (4) View as a slideshow. (5) Use the home red button after each question. ©Norman Herr, 2003

Acid Calculations-100 • QUESTION: Calculate the p. H of a solution containing 0. 134 mol. L-1 hydronium ions • ANSWER: 0. 873 Answer Question

Acid Calculations-200 • QUESTION: Calculate the p. H of 1. 35 x 10 -9 mol. L-1 HCl • ANSWER: 6. 99 Answer Question

Acid Calculations-300 • QUESTION: Calculate the p. H of 3. 0 x 104 mol. L-1 H SO 2 4 • ANSWER: 3. 22 Answer Question

Acid Calculations-400 • QUESTION: Calculate the p. H of an acid with a hydroxide ion concentration of 1. 38 x 10 -10 mol. L-1 • ANSWER: 4. 14 Answer Question

Acid Calculations-500 • QUESTION: Calculate the p. H of 0. 0243 mo. L-1 ethanoic acid, given Ka = 1. 74 x 10 -5 • ANSWER: 3. 19 Answer Question

Base Calculations-100 • QUESTION: Calcuate the p. H of 0. 1 mol. L -1 hydroxide ions • ANSWER: 13 Answer Question

Base Calculations-200 • QUESTION: Calculate the p. H of 0. 153 mol. L-1 KOH • ANSWER: 13. 2 Answer Question

Base calculations-300 • QUESTION: Calculate the hydroxide ion concentration of a 3. 02 x 10 -4 mol. L-1 HCl solution • ANSWER: 3. 31 x 10 -11 mol. L-1 Answer Question

Base Calculations-400 • QUESTION: If the p. H is 8. 65, what is the concentration of hydronium ions? • ANSWER: 4. 47 x 10 -6 mol. L-1 Answer Question

Base calculations-500 • QUESTION: Calculate the p. H of a 0. 107 mol. L-1 solution of NH 3, given the p. Ka of the ammonium ion is 9. 75 • ANSWER: 11. 4 Answer Question

Concentration of species-100 • QUESTION: From highest to lowest concentration, what are the species present in a 1 mol. L-1 solution of H 2 SO 4? • ANSWER: H 2 O>>H 3 O+>SO 42 ->OH- Answer Question

Concentration of species-200 • QUESTION: From highest to lowest concentration, what are the species present in a 1 mol. L-1 solution of CH 3 COOH? • ANSWER: H 2 O>> CH 3 COOH>H 3 O+ =CH 3 COO->OH- Answer Question

Concentration of species-300 • QUESTION: From highest to lowest concentration, what are the species present in a 1 mol. L-1 solution of NH 3? • ANSWER: H 2 O >> NH 3 > OH- = NH 4+ > H 3 O+ Answer Question

Concentration of species-400 • QUESTION: From highest to lowest concentration, what are the species present in a 1 mol. L-1 solution of NH 4 NO 3? • ANSWER: H 2 O >> NO 3 - = NH 4+ >H 3 O+ = NH 3 > OH- Answer Question

Concentration of species-500 • QUESTION: From highest to lowest concentration, what are the species present in a 1 mol. L-1 solution of HCOONa? • ANSWER: H 2 O >> HCOO- = Na+ > OH= HCOOH > H 3 O+ Answer Question

Titration curves-100 • QUESTION: The concentration of HPab (a weak acid) solution was determined by titration. A 20. 0 m. L sample of the HPab solution required 12. 0 m. L of 0. 0500 mol L– 1 Na. OH to reach the equivalence point. The equation for the reaction occurring is HPab + Na. OH → Na. Pab + H 2 O Calculate the concentration of the HPab solution. • ANSWER: 0. 03 mol. L-1 Answer Question
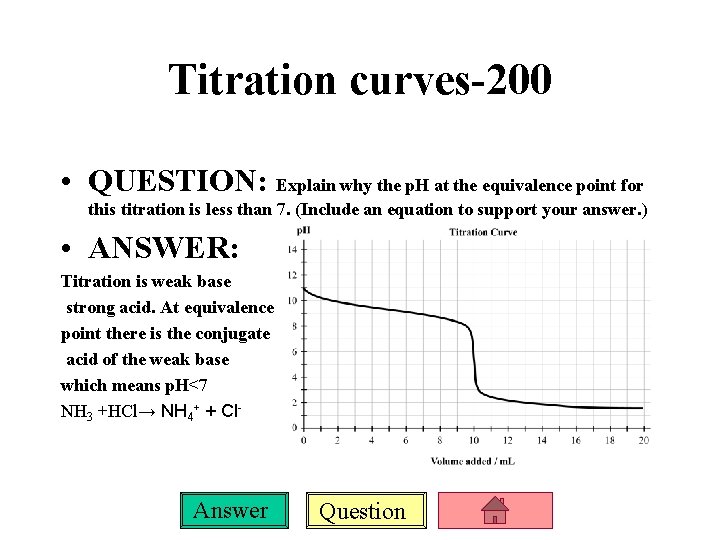
Titration curves-200 • QUESTION: Explain why the p. H at the equivalence point for this titration is less than 7. (Include an equation to support your answer. ) • ANSWER: Titration is weak base strong acid. At equivalence point there is the conjugate acid of the weak base which means p. H<7 NH 3 +HCl→ NH 4+ + Cl- Answer Question
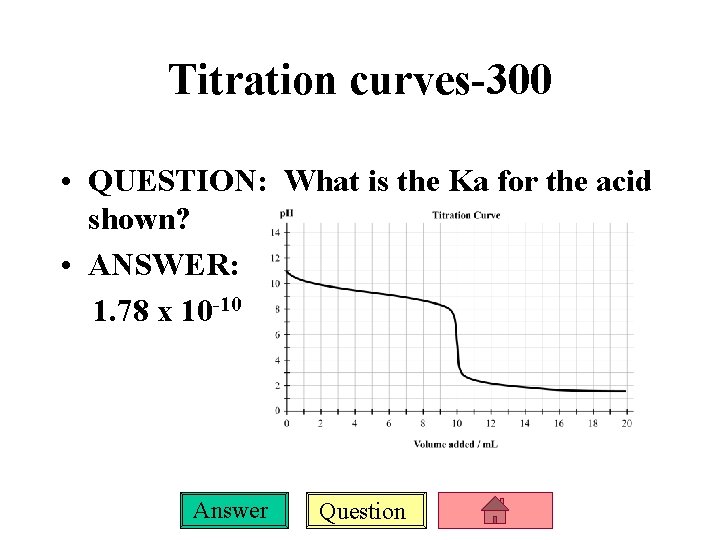
Titration curves-300 • QUESTION: What is the Ka for the acid shown? • ANSWER: 1. 78 x 10 -10 Answer Question

Titration curves-400 • QUESTION: 20. 0 m. L of the weak acid HPab (0. 03 mol. L-1) is titrated against 0. 05 mol. L-1 Na. OH. The Ka of (Pab-) is 1. 2 x 10 -5. Its initial p. H is 3. 22. Using the information above, sketch a curve showing the change in p. H against the volume of sodium hydroxide added to the solution in the flask. • ANSWER: Curve has: p. H 3. 22 at 0 m. L, 4. 92 at 6 m. L, and end point p. H 8 -10 at 12 m. L, correct shape Answer Question

Titration curves-500 • QUESTION: A titration is carried out – 40. 0 m. L of 0. 0500 mol L– 1 Na. OH solution is titrated against 0. 200 mol L– 1 CH 3 COOH solution. Calculate the p. H after 5 m. L CH 3 COOH added. • ANSWER: 12. 3 Answer Question
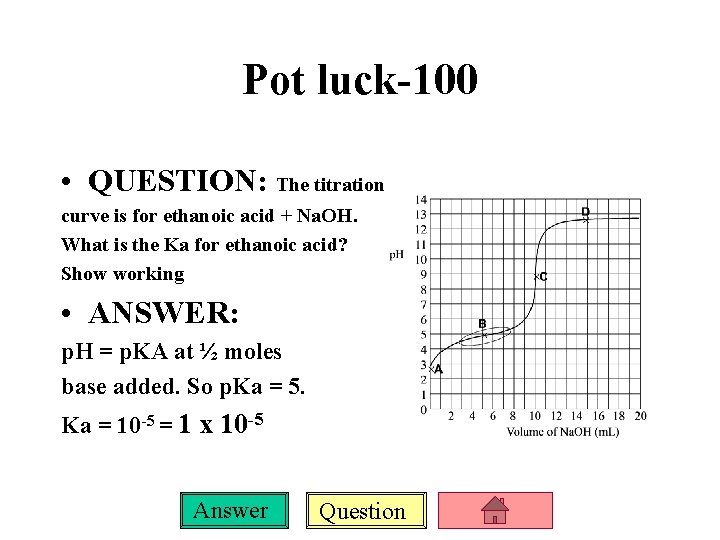
Pot luck-100 • QUESTION: The titration curve is for ethanoic acid + Na. OH. What is the Ka for ethanoic acid? Show working • ANSWER: p. H = p. KA at ½ moles base added. So p. Ka = 5. Ka = 10 -5 = 1 x 10 -5 Answer Question

Pot luck-200 • QUESTION: The titration curve is for ethanoic acid + Na. OH. List the species present in decreasing concentration at point B • ANSWER: H 2 O>>CH 3 COOH = CH 3 COO-=Na+>OH- Answer Question

Pot luck-300 • QUESTION: Indicator p. Ka Thymol blue 1. 7 Chlorophenol red 6. 1 Cresol red 8. 3 Choose the best indicator for the titration. Give a reason. • ANSWER: Cresol red – its p. Ka means it would change colour in the range of the equivalence pint of the titration Answer Question

Pot luck-400 • QUESTION: Discuss how a buffer solution of p. H = 3. 3 could be prepared from solutions of 0. 01 mol L– 1 HNO 2 and 0. 01 mol L– 1 KOH. Include relevant equations in your answer. p. Ka (HNO 2) = 3. 3 • ANSWER: Mix 10 m. L of HNO 2 with 5 m. L of KOH. (or equivalent) HNO 2 + OH– NO 2– + H 2 O At half neutralisation conc. HNO 2 = conc. NO 2 - and therefore Ka = H 3 O+ p. Ka = p. H Answer Question

Pot luck-500 • QUESTION: An acid base indicator has the formula HIn. It is yellow in the presence of any acid solution and blue in the presence of any base solution. p. Ka for HIn is 7. 0. Using chemical equations explain how such an indicator works. Explain under what conditions and p. H the indicator is green. • ANSWER: The indicator HIn is a weak acid and reacts with water as shown in the equation. HIn(aq) + H 2 O(aq) → In-(aq) + H 3 O+(aq) The HIn form of the indicator is yellow and the In- form is blue. If acid, i. e. hydronium ions are added to the indicator solution, the position of equilibrium is shifted to the left hand side, forming more HIn and therefore giving an yellow colour. If base is added to the indicaor solution, the hydronium ions will react with it, shifting the equilibrium position to the right, forming more In-, and therefore giving a blue colour. At ap. H of 7 the concentration of the HIn and the In- will be equal and the colour of the solution will be a mixture of blue and yellow, i. e. green. Answer Question

FINAL JEOPARDY • ANSWER: • QUESTION: Answer Question
- Slides: 28