Acids and Bases Goal of the class To

Acids and Bases

Goal of the class • To understand the properties of acids and bases • Question of the day: What are the chemical properties of acids and bases? • Previous Answer: Salt dissolves in water because the water is attracted to the salts ions

Acids and Bases Characteristics • Acids – Have a sour taste – Can burn skin when touched – Make hydrogen gas when mixed with metals • Bases – Have a bitter taste – They have a slippery feel – They can also be corrosive as well

Acid Chemistry • An acid is any substance that produces hydrogen ions. • All acids will break apart in water giving hydronium ion – Example: HCl + H 2 O → H+ + Cl- + H 2 O – Those hydrogen ions can react with metals to form H 2

Base Chemistry • Bases cause the formation of hydroxide ions – Example: Na. OH + H 2 O → Na+ + OH- + H 2 O • Some bases use the water in solution to form a base – Example: NH 3 + H 2 O → NH 4+ + OH-

Water Dissociation • Water will naturally split into both hydronium and hydroxide ions. • So it can behave as both an acid and a base.

Acid and Base Strength • Different substances give different amounts of hydronium or hydroxide ions. • Indicator solutions can be used to determine if a solution is an acid or base. • Acids and bases can be measured using litmus paper – Acids turn red – Bases turn blue
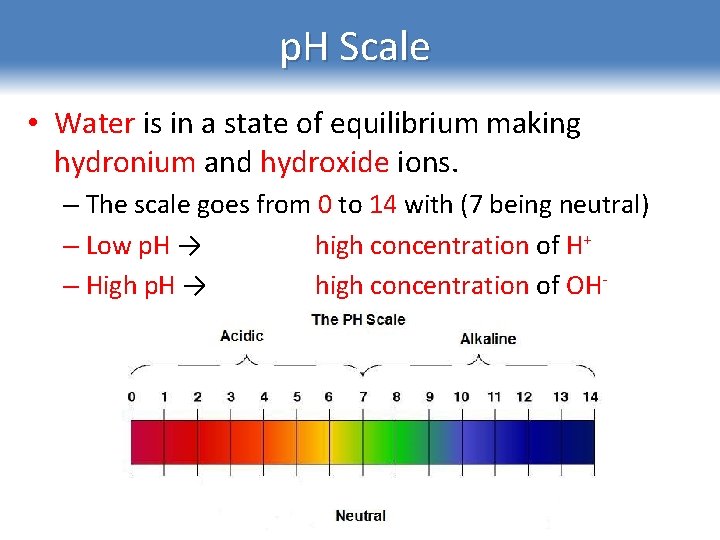
p. H Scale • Water is in a state of equilibrium making hydronium and hydroxide ions. – The scale goes from 0 to 14 with (7 being neutral) – Low p. H → high concentration of H+ – High p. H → high concentration of OH-

Acid Base Reactions • Acids and bases will neutralise when mixed – Water and a salt will remain • The hydronium and hydroxide ions will mix to form water • The remaining ions will form salts

Practice • Please think and answer the following questions: • Which would be a stronger acid, hydrochloric acid (p. H 1) or ascorbic acid (p. H 2. 5)? • Write the chemical equation and explain how sodium hydroxide (Na. OH) and hydrochloric acid (HCl) neutralize each other.

Vocabulary • Neutralize – a chemical reaction where an acid and base react to form a salt.

Homework • Please Read: Chemical interactions page: 104 109 • Pleases complete Chemical Interactions workbook pages 46 -48
- Slides: 12