ACIDS AND BASES GENERAL PROPERTIES ACIDS Taste sour

ACIDS AND BASES

GENERAL PROPERTIES ACIDS Taste sour Turn litmus React with active metals above hydrogen in the activity series React with bases blue to red BASES Taste bitter Turn litmus Feel soapy or slippery (react with fats to make soap) React with acids red to blue

DEFINITIONS Arrehenius only in water Bronsted-Lowry any solvent Acids – produce H 3 O + Bases - produce OH - Acids – donate H + Bases – accept H + Lewis used in organic chemistry, wider range of substances Acids – accept e - pair Bases – donate e - pair
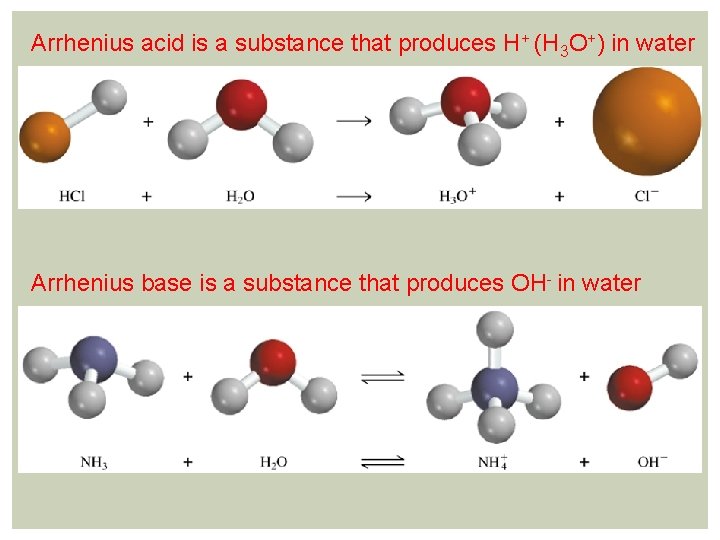
Arrhenius acid is a substance that produces H+ (H 3 O+) in water Arrhenius base is a substance that produces OH- in water
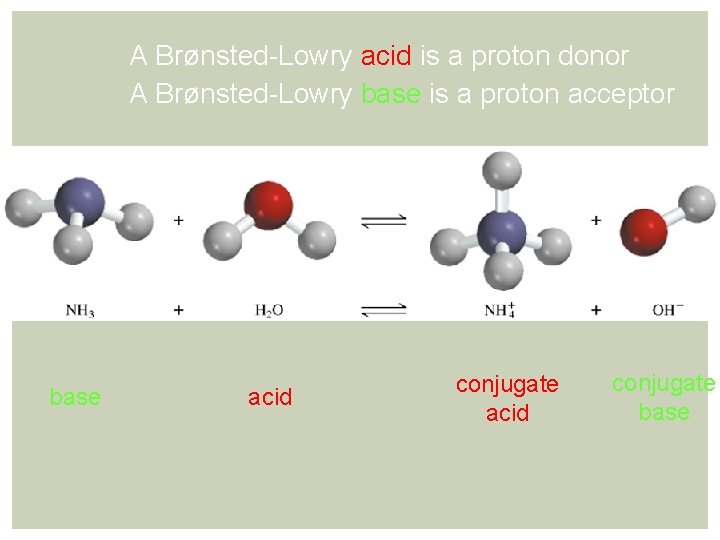
A Brønsted-Lowry acid is a proton donor A Brønsted-Lowry base is a proton acceptor base acid conjugate base
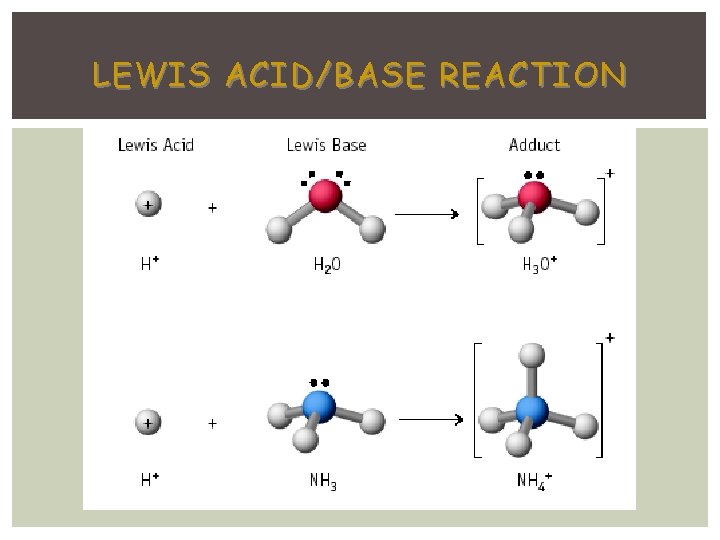
LEWIS ACID/BASE REACTION
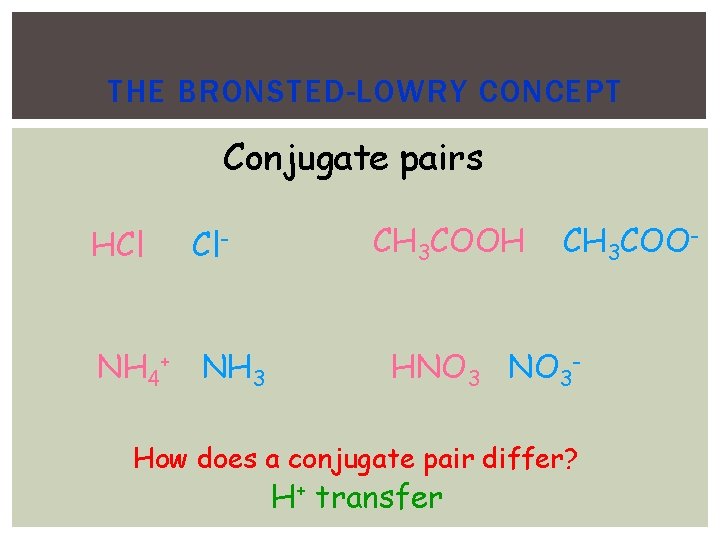
THE BRONSTED-LOWRY CONCEPT Conjugate pairs HCl Cl- NH 4+ NH 3 CH 3 COOH CH 3 COO- HNO 3 - How does a conjugate pair differ? H+ transfer
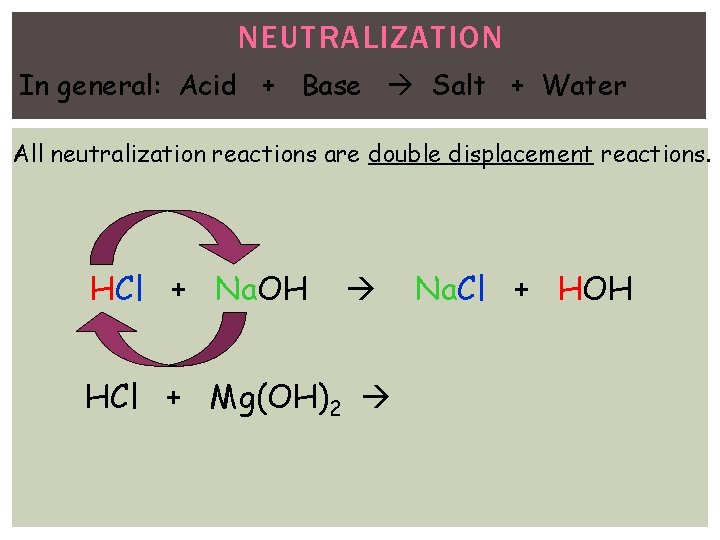
NEUTRALIZATION In general: Acid + Base Salt + Water All neutralization reactions are double displacement reactions. HCl + Na. OH HCl + Mg(OH)2 Na. Cl + HOH

ACID - BASE TITRATIONS • This is a neutralization reaction used in Chemistry lab to determine the concentration of an acid or a base; Øuse a base or an acid with a known concentration to neutralize the acid or base with the unknown concentration EQUATION TO USE FOR SOLVING TITRATION PROBLEMS: n. A * MA. *VA = n. B * MB * VB n. A = number of H’s at the beginning of the formula for the acid MA = molarity of the acid VA = volume of the acid n. B = number of OH’s in the formula for the base MB = molarity of the base VB = volume of the base EXAMPLE PROBLEM: What is the molarity of a H 2 SO 4 solution if 190 m. L of the acid is needed to exactly neutralize 150 m. L of a 2. 5 M Na. OH solution? 2. MA. 190 m. L = 1. 2. 5 M. 150 m. L MA= 0. 99 M

PH SCALE (NEGATIVE LOG FUNCTION) p. H = 0 p. H = 1 p. H = 2 p. H = 3 p. H = 4 p. H = 5 p. H = 6 p. H = 7 p. H = 8 p. H = 9 p. H = 10 p. H = 11 p. H = 12 p. H = 13 p. H = 14 [H+] = 1 x 100 M = 1 M [H+] = 1 x 10 -1 M = 0. 1 M [H+] = 1 x 10 -2 M = 0. 01 M [H+] = 1 x 10 -3 M = 0. 001 M [H+] = 1 x 10 -4 M = 0. 0001 M [H+] = 1 x 10 -5 M = 0. 00001 M [H+] = 1 x 10 -6 M = 0. 000001 M [H+] = 1 x 10 -7 M = 0. 0000001 M [H+] = 1 x 10 -8 M = 0. 00000001 M [H+] = 1 x 10 -9 M = 0. 00001 M [H+] = 1 x 10 -10 M = 0. 00001 M [H+] = 1 x 10 -11 M = 0. 000001 M [H+] = 1 x 10 -12 M = 0. 000001 M [H+] = 1 x 10 -13 M = 0. 0000001 M [H+] = 1 x 10 -14 M = 0. 0000001 M Note: Every time the p. H of a substance goes up by 1, the concentration of H + goes down by a factor of ten, which is why you take the negative log of [H +] to get p. H = -log[H+] =10 -p. H
![[H + ], [OH - ], PH AND POH [H+] = the molarity of [H + ], [OH - ], PH AND POH [H+] = the molarity of](http://slidetodoc.com/presentation_image/62c74bae5cb521a87580321b59396fba/image-11.jpg)
[H + ], [OH - ], PH AND POH [H+] = the molarity of H+ in the solution [OH-] = the molarity of OH- in the solution Ø When [H+] = [OH-], the solution is neutral [H+][OH-] = 1 x 10 -14 p. H = parts hydrogen Ø Friendly number between 1 and 14 used to describe the acidity or basicity of a substance relative to water Ø Derived from the exponent of the concentration of H+ p. OH = parts hydroxide Ø Similar to p. H, but derived from the concentration of OHØ Since acids and bases are opposites, p. H and p. OH are opposites! p. H + p. OH = 14
![PH [H + ] POH [OH - ] PH [H + ] POH [OH - ]](http://slidetodoc.com/presentation_image/62c74bae5cb521a87580321b59396fba/image-12.jpg)
PH [H + ] POH [OH - ]
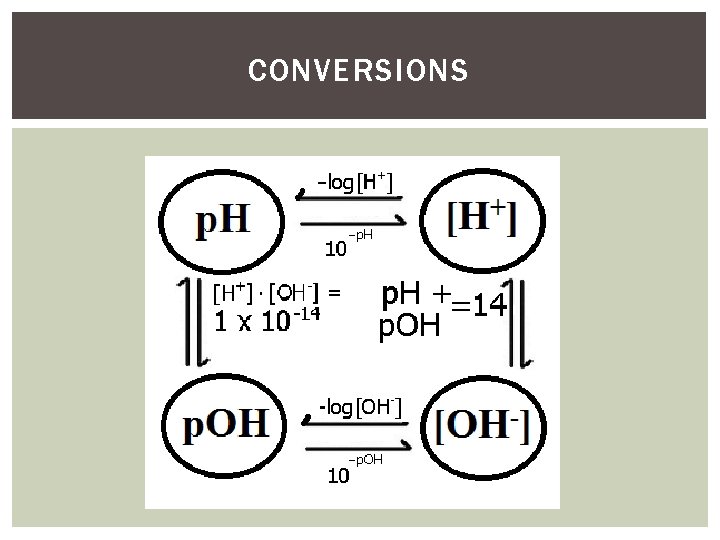
CONVERSIONS
- Slides: 13