Acids and Bases General properties ACIDS Taste sour

Acids and Bases

General properties ACIDS • Taste sour • Turn litmus • React with active metals – Fe, Zn • • BASES Taste bitter Turn litmus Feel soapy or slippery (react with fats to make soap) React with acids • React with bases blue to red to blue

More properties • • ACIDS Feel and look like water Conduct electricity (electrolytes) when dissolved in water Burn or sting Usually formula starts w/ H BASES • do not react with metals • Can be strong or weak electrolytes in water • Do not sting or burn

Definitions Arrehenius only in water • Acids – produce H+ • Bases - produce OH- • Acids – donate H+ Bronsted-Lowry • Bases – accept H+ any solvent
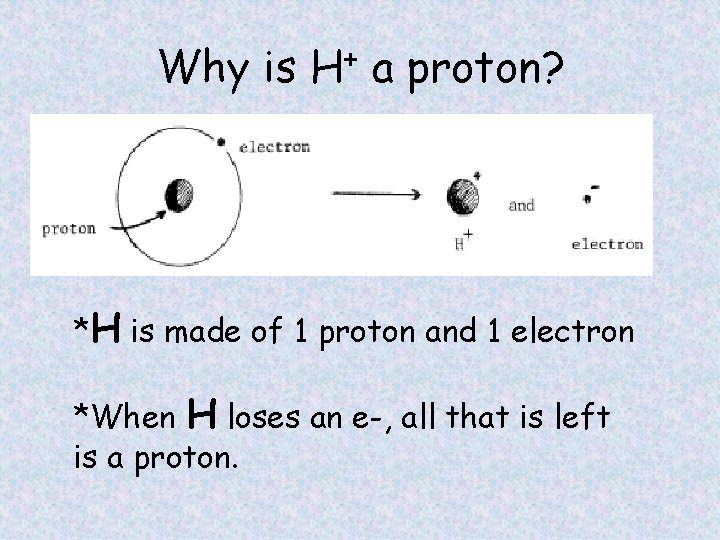
Why is H+ a proton? *H is made of 1 proton and 1 electron *When H loses an e-, all that is left is a proton.
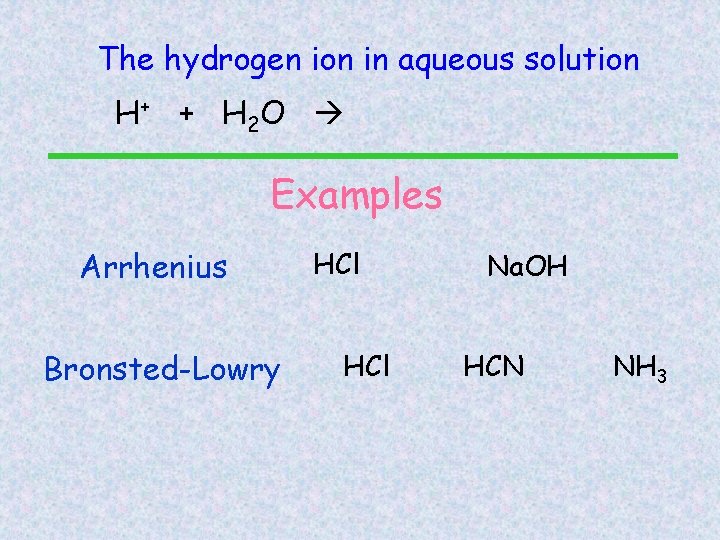
The hydrogen ion in aqueous solution H+ + H 2 O H 3 O+ (hydronium ion) Examples Arrhenius Bronsted-Lowry HCl Na. OH HCN NH 3

Conjugate acid/base pairs • Conjugate acid –acid that forms after the original base accepts an H+ • Conjugate base –base that forms after the original acid donates an H+ NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq)

Conjugate acid/base pairs **Notice how water is an ACID in the first reaction and BASE in the other reaction *water is called AMPHOTERIC
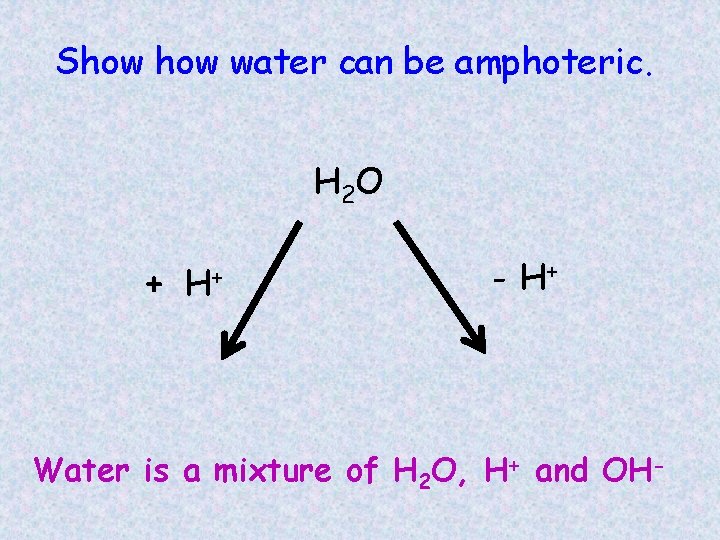
Show water can be amphoteric. H 2 O + H+ - H+ Water is a mixture of H 2 O, H+ and OH-
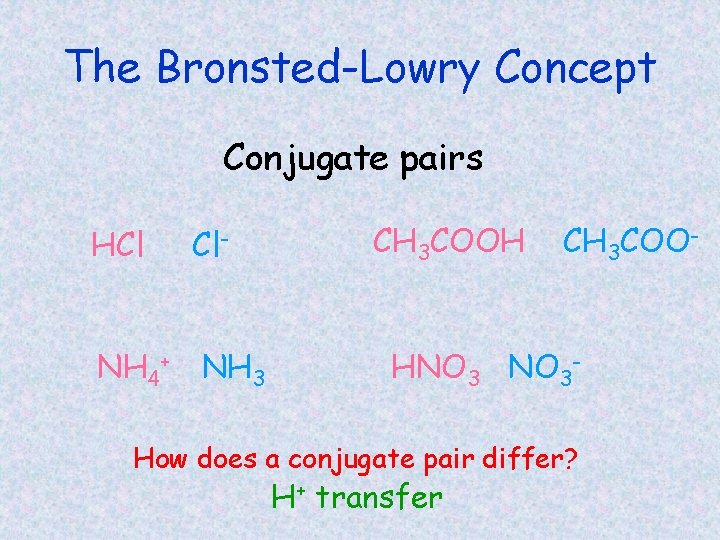
The Bronsted-Lowry Concept Conjugate pairs HCl Cl- NH 4+ NH 3 CH 3 COOH CH 3 COO- HNO 3 - How does a conjugate pair differ? H+ transfer
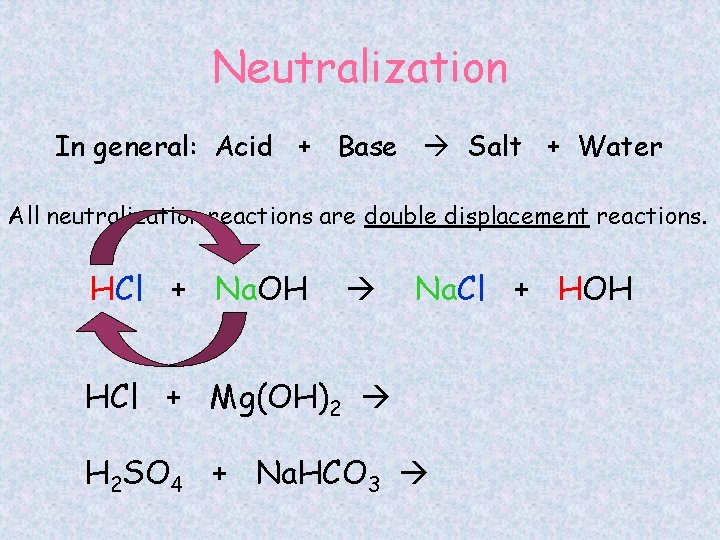
Neutralization In general: Acid + Base Salt + Water All neutralization reactions are double displacement reactions. HCl + Na. OH Na. Cl + HOH HCl + Mg(OH)2 H 2 SO 4 + Na. HCO 3
Let’s examine the behavior of an acid, HA, in aqueous solution. HA What happens to the HA molecules in solution?
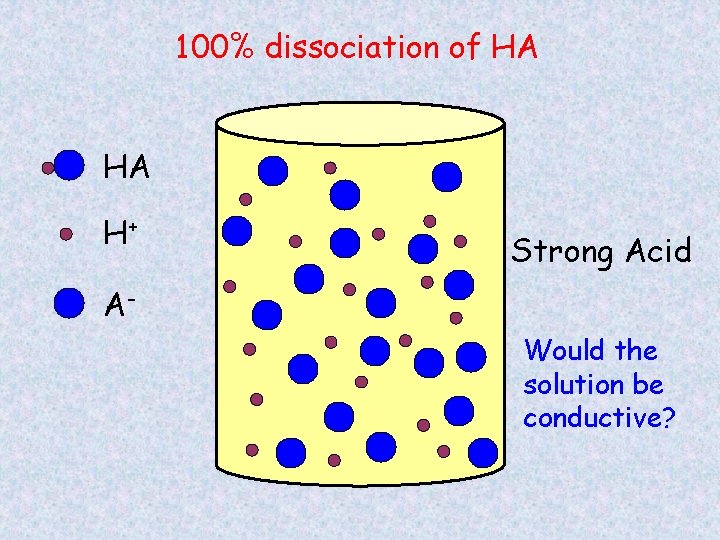
100% dissociation of HA HA H+ Strong Acid AWould the solution be conductive?
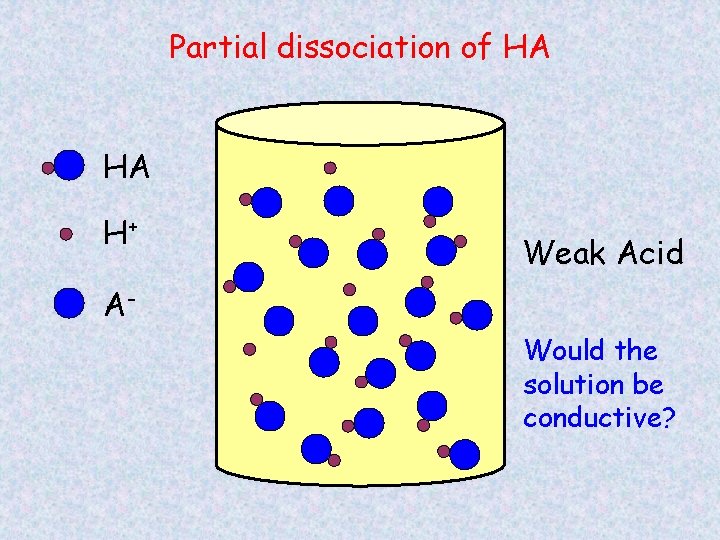
Partial dissociation of HA HA H+ Weak Acid AWould the solution be conductive?
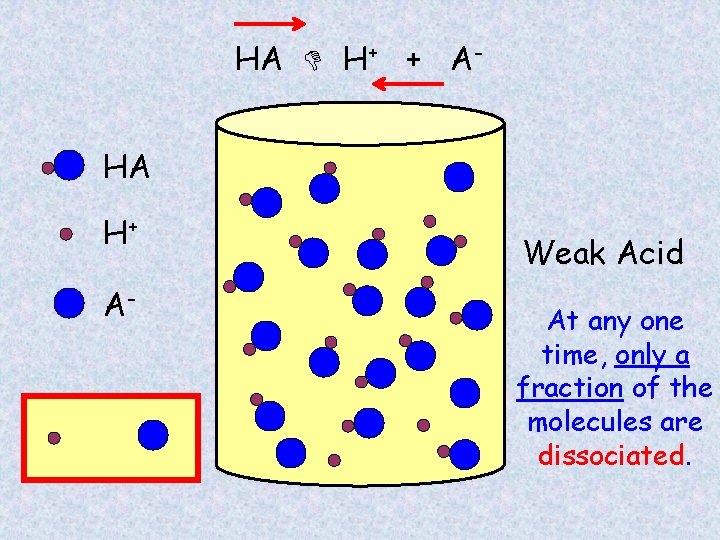
HA H+ + AHA H+ A- Weak Acid At any one time, only a fraction of the molecules are dissociated.
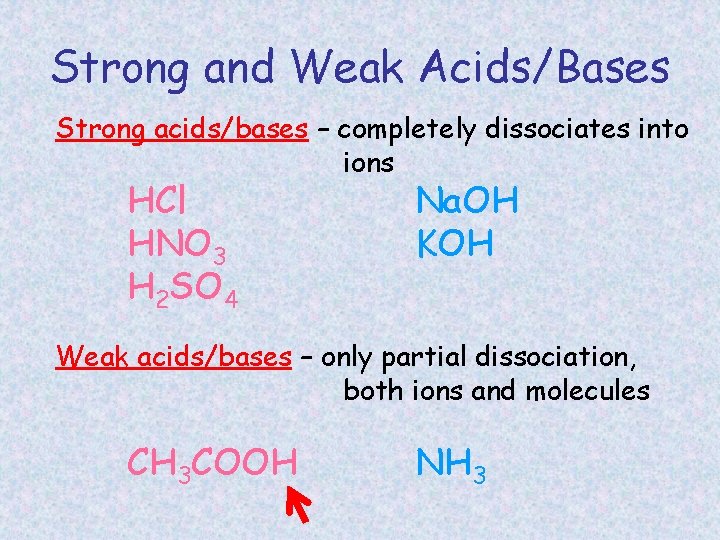
Strong and Weak Acids/Bases Strong acids/bases – completely dissociates into ions HCl HNO 3 H 2 SO 4 Na. OH KOH Weak acids/bases – only partial dissociation, both ions and molecules CH 3 COOH NH 3
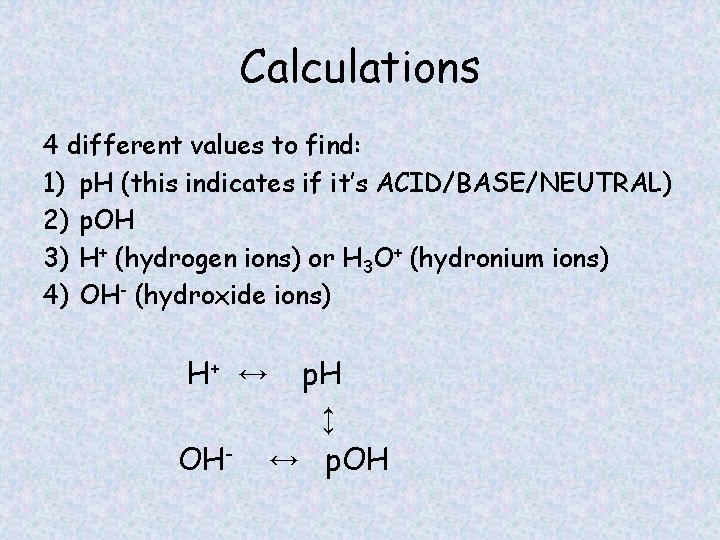
Calculations 4 different values to find: 1) p. H (this indicates if it’s ACID/BASE/NEUTRAL) 2) p. OH 3) H+ (hydrogen ions) or H 3 O+ (hydronium ions) 4) OH- (hydroxide ions) H+ ↔ OH- p. H ↕ ↔ p. OH

Calculations • What is p. H? The power of hydrogen (used to determine if solution is and acid, base or neutral) • What is p. OH? The power of hydroxide • What is [H+]? The molarity of acid ions in a solution • What is [OH-]? The molarity of base ions in a solution
![p. H = -log[H 3 O+]* *or [H +] p. OH = -log[OH ] p. H = -log[H 3 O+]* *or [H +] p. OH = -log[OH ]](http://slidetodoc.com/presentation_image/acb2062d1e26687fd5e717d5a337f55c/image-19.jpg)
p. H = -log[H 3 O+]* *or [H +] p. OH = -log[OH ] p. H + p. OH = 14
![[H+]= 10^(-p. H) [OH-] = 10^(-p. OH) How to check your work: [H 3 [H+]= 10^(-p. H) [OH-] = 10^(-p. OH) How to check your work: [H 3](http://slidetodoc.com/presentation_image/acb2062d1e26687fd5e717d5a337f55c/image-20.jpg)
[H+]= 10^(-p. H) [OH-] = 10^(-p. OH) How to check your work: [H 3 O+][OH-] = 1. 0 10 -14 p. H + p. OH = 14

p. H of Common Substances
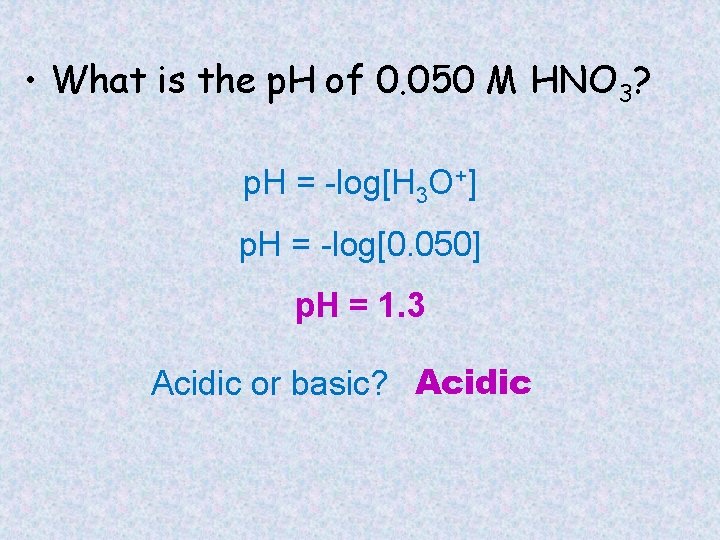
• What is the p. H of 0. 050 M HNO 3? p. H = -log[H 3 O+] p. H = -log[0. 050] p. H = 1. 3 Acidic or basic? Acidic
![Example Find the p. H, p. OH, & [OH- ] if [H+]= 3. 25 Example Find the p. H, p. OH, & [OH- ] if [H+]= 3. 25](http://slidetodoc.com/presentation_image/acb2062d1e26687fd5e717d5a337f55c/image-23.jpg)
Example Find the p. H, p. OH, & [OH- ] if [H+]= 3. 25 x 10 -4 M

• What is the molarity (concentration) of HBr in a solution that has a p. OH of 9. 6? p. H + p. OH = 14 p. H = -log[H 3 O+] p. H + 9. 6 = 14 4. 4 = -log[H 3 O+] p. H = 4. 4 -4. 4 = log[H 3 O+] Acidic [H 3 O+] = 4. 0 10 -5 M HBr
![p. H p. OH [H+] [OH―] 5. 87 2. 51 x 10 -2 M p. H p. OH [H+] [OH―] 5. 87 2. 51 x 10 -2 M](http://slidetodoc.com/presentation_image/acb2062d1e26687fd5e717d5a337f55c/image-25.jpg)
p. H p. OH [H+] [OH―] 5. 87 2. 51 x 10 -2 M Acid/Base /Neutral 3. 21 . 00875 M

Testing p. H in the Lab • Litmus paper • Indicators • p. H meter

Indicators • dyes that will change color in the presence of an acid or base. • Color changes occur when the molecules are ionized (acting as an acid) or unionized (acting as a base). • Indicators change color during specific p. H ranges

Some of the many p. H Indicators and their p. H range

Titration • A laboratory method for determining the concentration of an unknown acid or base using a neutralization reaction. • A standard solution, (a solution of known concentration), is used.

Titration Terminology • Equivalence point - The point at which there are equal concentrations of acid and base. [H+] = [OH-] • End point – the point at which the chosen indicator changes color to mark a change in p. H (also called indication point)
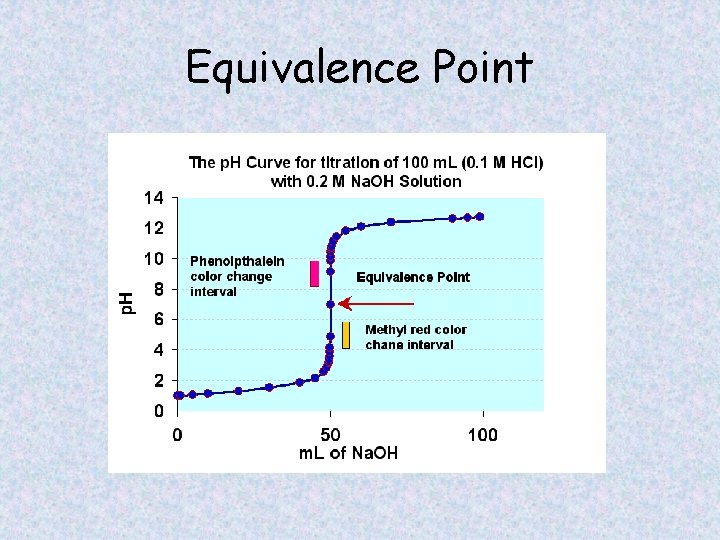
Equivalence Point

Solving Titration Problems • Use the titration formula M a V a = M b. V b What is the concentration of HCl if 30. 0 m. L of 0. 10 M Na. OH neutralizes 50. 0 m. L HCl?
- Slides: 32