Acids and Bases Chapters 14 and 15 AcidBase

Acids and Bases Chapters 14 and 15

Acid/Base Definitions l Arrhenius Model l Bronsted-Lowry Model l Acids produce hydrogen ions (H+ or H 3 O+) in aqueous solutions Bases produce hydroxide ions (OH-) in aqueous solutions Acids are proton (H+) donors Bases are proton (H+) acceptors Lewis Model l l Acids are electron pair acceptors Bases are electron pair donors

Example 1 l Water acting as a base by accepting a proton, HCl acitng as an acid by donating a proton. HCl acting as an acid by producing H 3 O+ ions in solution. H+ ions provided by the HCl act as an acid by accepting a pair of electrons from the oxygen atom in the water (a base) to form a bond in H 3 O+. HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl-(aq) acid base conjugate acid base

Example 2 l Water acting as an acid by donating a proton, NH 3 acting as a base by accepting a proton. NH 3 acting as a base by producing OH- ions in solution. NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq) base acid conjugate acid base

Example 3 l Electron rich ammonia acting as a base by donating a pair of electrons to electron deficient Boron, that in turn acts as an acid by accepting the pair of electrons. NH 3 + BF 3 H 3 N-BF 3

Conjugate Acid/Base Pairs l Related by a hydrogen ion on either side of the equation l Example 2 previously l NH 3 and NH 4+

Strength of Acids and Bases l Strong acids and bases are assumed to completely ionize in solution. l For a strong acid (HA) and a strong base (B), the following reactions go to completion (no reverse reaction occurs) HA(aq) + H 2 O(l) H 3 O+ + A-(aq) l B(aq) + H 2 O(l) BH+(aq) + OH-(aq) l

Strength of Acids and Bases l Weak acids and bases have very little ionization Equilibria are set up (reverse reactions are possible) l The equilibrium positions for each lie heavily to the left hand side of the equation l HA(aq) + H 2 O(l) H 3 O+ + A-(aq) l B(aq) + H 2 O(l) BH+(aq) + OH-(aq) l
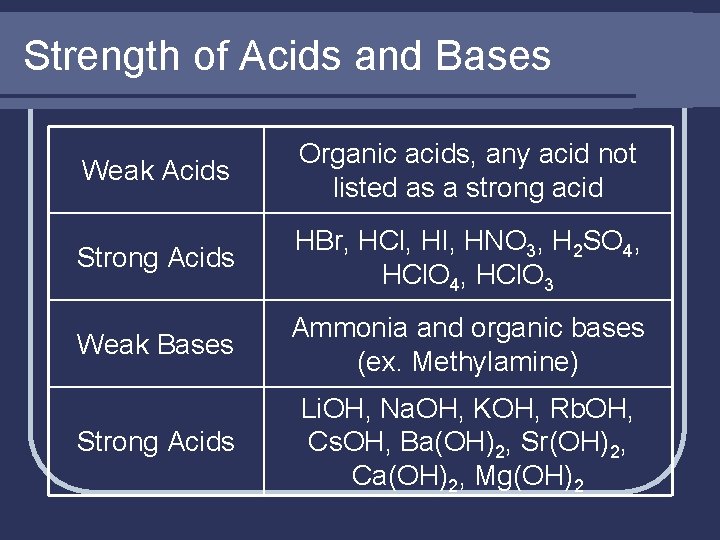
Strength of Acids and Bases Weak Acids Organic acids, any acid not listed as a strong acid Strong Acids HBr, HCl, HI, HNO 3, H 2 SO 4, HCl. O 3 Weak Bases Ammonia and organic bases (ex. Methylamine) Strong Acids Li. OH, Na. OH, KOH, Rb. OH, Cs. OH, Ba(OH)2, Sr(OH)2, Ca(OH)2, Mg(OH)2

Some Extra Terms Strong – completely dissociated into ions l Concentrated – a high number of moles of solute per liter of solution l Corrosive – chemically reactive l Weak – only slightly dissociated into ions l Dilute – a low number of moles of solute per liter of solution l

Self Ionization of Water l H 2 O + H 2 O H 3 O+ + OHl At 25 , [H 3 O+] = [OH-] = 1 x 10 -7 l Kw is a constant at 25 C: l Kw = [H 3 O+][OH-] l Kw = (1 x 10 -7) = 1 x 10 -14

Amphoterism l Water can accept H+ or donate H+ l This is called amphoterism l Pure water does it by itself, although not much l Water will make both protons and hydroxides without any outside work

Amphoterism l Reaction l 2 H 2 O(l) H 3 O+(aq) + OH-(aq) l We use the typical chemist’s shortcut to write this equation l 2 H 2 O(l) H+(aq) + OH-(aq) l Kw = [H+][OH-] l The value of Kw is known at room temp

Amphoterism l If you increase either protons or hydroxides (by adding acid or base) you MUST decrease the other mathematically in order to maintain the constant value of Kw l Example – calculate [H+] for a solution with 1. 0 x 10 -5 M OH- at 25 o. C. Is it neutral, acidic or basic?

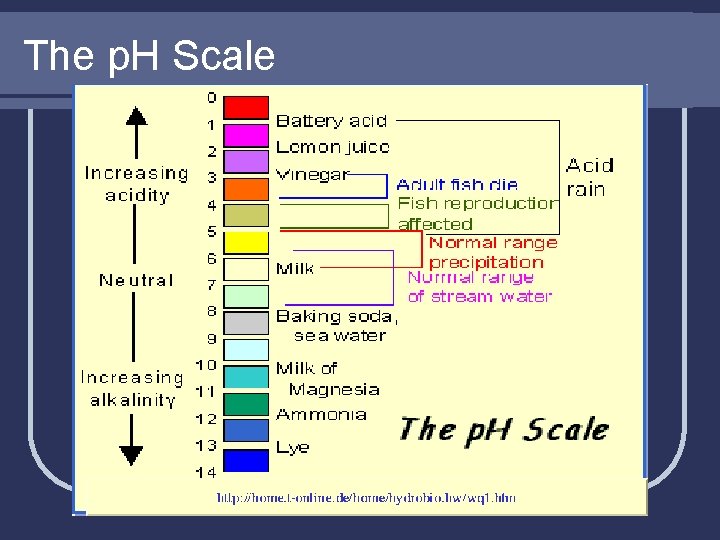
The p. H Scale l Used to indicate the strength of an acid or base l Ranges from 0 – 14 l Acids are less than 7 l Bases are greater than 7 l 7 is considered neutral
The p. H Scale

The p. H Scale l Calculating p. H, p. OH p. H = -log 10(H 3 O+) l p. OH = -log 10(OH-) l l Relationship l between p. H and p. OH p. H + p. OH = 14 l Finding [H 3 O+], [OH-] from p. H, p. OH [H 3 O+] = 10 -p. H l [OH-] = 10 -p. OH l
![The p. H Scale [OH-] = 1 x 10 -14 [H+] p. H [H+] The p. H Scale [OH-] = 1 x 10 -14 [H+] p. H [H+]](http://slidetodoc.com/presentation_image/d8180c66e7481226ff273a2e9db0f131/image-18.jpg)
The p. H Scale [OH-] = 1 x 10 -14 [H+] p. H [H+] = 1 x 10 -14 [OH-] p. OH = 14 - p. OH = -log[OH-] OH[OH-] = 10 -p. OH p. H = -log[H+] = 10 -p. H H+ p. OH

The p. H Scale l Examples Calculate the p. H of a solution of 0. 030 M hydrochloric acid l Calculate the p. H of a 0. 010 M solution of calcium hydroxide l Calculate the H 3 O+ concentration in a solution with a p. H of 4. 32 l Calculate the p. H of a solution made by dissolving 2. 00 g of potassium hydroxide in distilled water to a total volume of 250. m. L l

Ka – The Equilibrium Constant for Weak Acids For a weak acid (HA) dissociation is incomplete so an equilibrium must be set up HA(aq) + H 2 O(l) H 3 O+ + A-(aq) Initial X 0 0 Change -x +x +x Equilibrium X-x 0+x l Can’t go directly to the p. H equation because we don’t know how much acid is dissociated l Derive the following equilibrium expression to help solve these problems Ka = [H 3 O+][A-] [HA] l

Weak Acid Equilibrium Problem l What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? l Step #1: Write the dissociation equation l HC 2 H 3 O 2 - + H+

Weak Acid Equilibrium Problem l What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? l Step 2: ICE it! l HC 2 H 3 O 2 - + H+ I 0. 50 0 0 C -x +x +x E 0. 50 – x x x

Weak Acid Equilibrium Problem l What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? l Step 3: Set up the law of mass action l HC 2 H 3 O 2 - + H+ 1. 8 x 10 -5 = (x)(x) = x 2 (0. 50 -x) 0. 50

Weak Acid Equilibrium Problem l What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? l Step 4: Solve for x, which is also [H+] l [H+] = 3. 0 x 10 -3 M

Weak Acid Equilibrium Problem l What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? l Step 5: Convert [H+] to p. H = -log(3. 0 x 10 -3) = 2. 52

Kb – The Equilibrium Constant for Weak Bases For a weak base (B) dissociation is incomplete so an equilibrium must be set up B(aq) + H 2 O(l) BH+ + OH-(aq) Initial X 0 0 Change -x +x +x Equilibrium X-x 0+x l Can’t go directly to the p. OH equation because we don’t know how much base is dissociated l Derive the following equilibrium expression to help solve these problems Kb = [BH+][OH-] [B] l

Weak Base Equilibrium Problem l What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? l Step #1: Write the equation for the reaction NH 3 + H 2 O NH 4+ + OH-

Weak Base Equilibrium Problem l What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? l Step 2: ICE it! NH 3 + H 2 O NH 4+ + OHI 0. 50 0 0 C -x +x +x E 0. 50 – x x x

Weak Base Equilibrium Problem l What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? l Step 3: Set up the law of mass action NH 3 + H 2 O NH 4+ + OH- 1. 8 x 10 -5 = (x)(x) = x 2 (0. 50 -x) (0. 50)

Weak Base Equilibrium Problem l What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? l Step 4: Solve for x, which is also [OH-] = 3. 0 x 10 -3 M

Weak Base Equilibrium Problem l What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? l Step 5: Convert [OH-] to p. H p. OH = -log(3. 0 x 10 -3 M) = 2. 52 p. H = 14. 00 – p. OH = 11. 48

Percent Dissociation l% dissociation = amount dissociated(mol/L) x 100% initial concentration(mol/L) l Specifies the amount of weak acid that has dissociated to achieve equilibrium

Example l 0. 500 M uric acid (HC 5 H 3 N 4 O 4) is 1. 6% dissociated. Find Ka and p. H. l What is the percent dissociation of 0. 500 M HC 2 H 3 O 2, Ka = 1. 8 x 10 -5?

Indicators l. A substance that changes color according to the p. H of the solution l Often are weak acids where the ionized and unionized forms are different colors HIn(aq) H+(aq) + In-(aq) color 1 color 2

Indicators l Common indicators for acids and bases l Litmus paper: red for acids, blue for bases l Phenophthalein: colorless for acids, pink for bases l Methyl orange: red for acids, yellow for bases
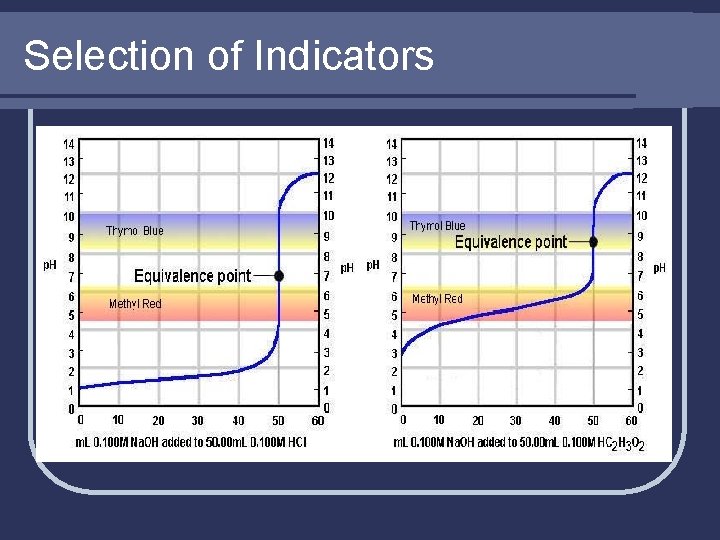
Selection of Indicators
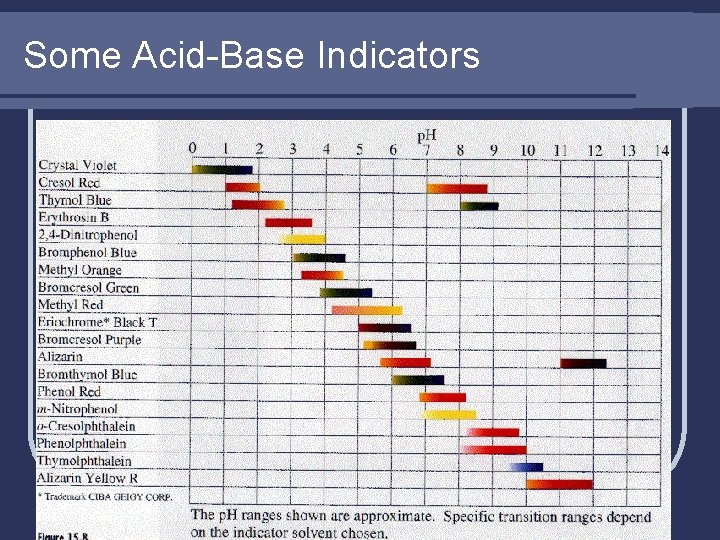
Some Acid-Base Indicators
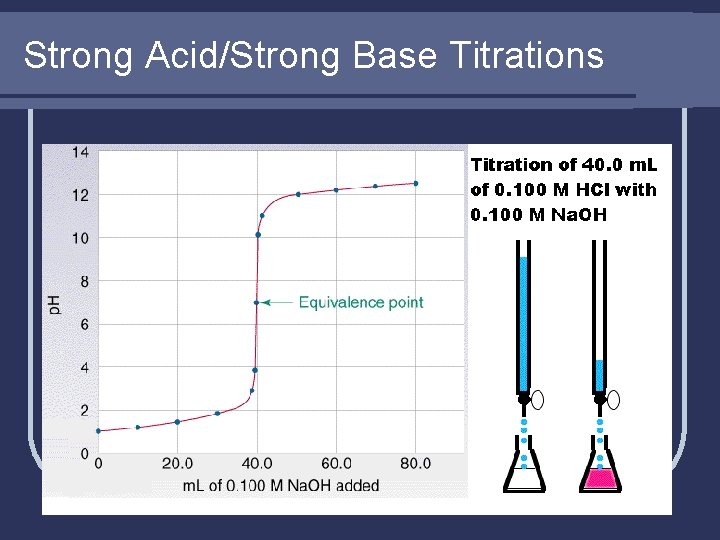
Strong Acid/Strong Base Titrations

Strong Acid/Strong Base Example l Calculate the p. H after these volumes of 0. 2500 M HCl are added to 50. 00 m. L of 0. 1500 M Na. OH. a) 0. 00 m. L b) 4. 00 m. L c) 29. 50 m. L d) 30. 00 m. L e) 30. 50 m. L f) 40. 00 m. L

Strong Acid/Strong Base Example l Notice the RAPID drop in p. H. l 1. 00 m. L between points c and e moves us from p. H 11. 20 to 2. 81 (9 orders of magnitude; a billion times difference!)
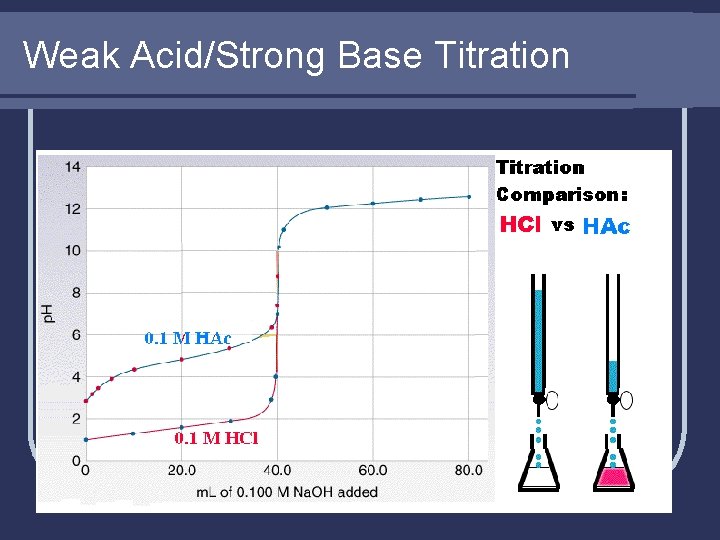
Weak Acid/Strong Base Titration
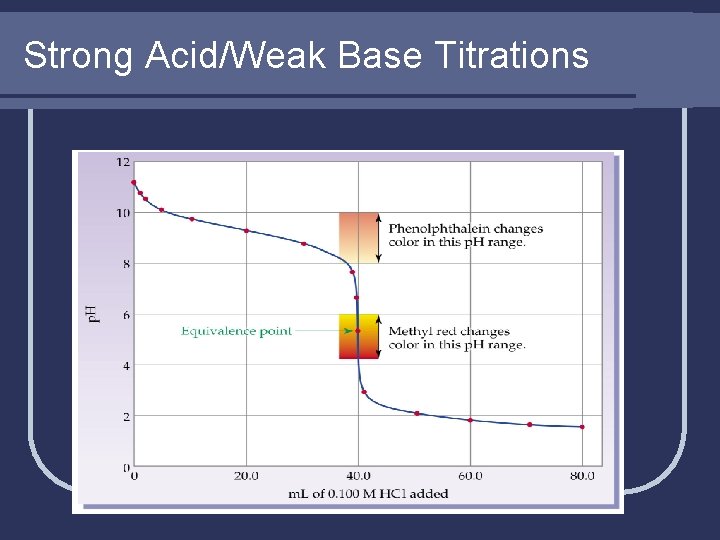
Strong Acid/Weak Base Titrations

Weak Acid/Strong Base Example l Find p. H after these volumes of 0. 400 M Na. OH are added to 50. 00 m. L of 0. 200 M HCOOH (Ka = 1. 8 x 10 -4) a) 0. 00 m. L e) 25. 00 m. L b) 5. 00 m. L f) 25. 50 m. L c) 12. 50 m. L g) 40. 00 m. L d) 24. 50 m. L

Weak Acid/Strong Base Example l Notice weak acid/strong base has endpoint (equivalence point) in the BASIC region, not at p. H of 7. 00 like in strong acid/strong base. l The exact same logic applies (and the same math is used) for strong ACID/weak BASE

Monoprotic and Polyprotic Acids l Monoprotic acids contain only one “ionizable” proton l Polyprotic acids contain more than one l Usually only the first H+ comes off easily as a strong ion l Each comes H+ off in a stepwise manner, each with its own Ka
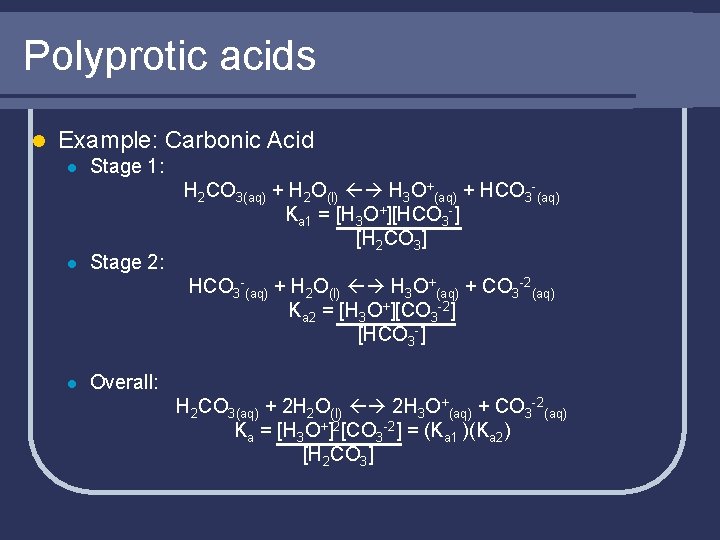
Polyprotic acids l Example: Carbonic Acid l l Stage 1: Stage 2: H 2 CO 3(aq) + H 2 O(l) H 3 O+(aq) + HCO 3 -(aq) Ka 1 = [H 3 O+][HCO 3 -] [H 2 CO 3] HCO 3 -(aq) + H 2 O(l) H 3 O+(aq) + CO 3 -2(aq) Ka 2 = [H 3 O+][CO 3 -2] [HCO 3 -] l Overall: H 2 CO 3(aq) + 2 H 2 O(l) 2 H 3 O+(aq) + CO 3 -2(aq) Ka = [H 3 O+]2[CO 3 -2] = (Ka 1 )(Ka 2) [H 2 CO 3]
![Example l Find [PO 4 -3], p. H and [OH-] in 6. 0 M Example l Find [PO 4 -3], p. H and [OH-] in 6. 0 M](http://slidetodoc.com/presentation_image/d8180c66e7481226ff273a2e9db0f131/image-47.jpg)
Example l Find [PO 4 -3], p. H and [OH-] in 6. 0 M H 3 PO 4 l Ka 1 = 7. 5 x 10 -3 Ka 2 = 6. 2 x 10 -8 Ka 3 = 4. 8 x 10 -13 l Since Ka 1 >> Ka 2 >> Ka 3 we can ignore the H+ in reactions 2 and 3 l To find [PO 4 -3] we must work our way all the way to the last equation

Buffered Solutions l. A solution that resists a change in p. H when either hydroxide ions or protons are added. l Buffered solutions contain either: l. A weak acid and its salt l. A weak base and its salt

Acid/Salt Buffering Pairs Formula of the l. Weak The. Acid salt will contain the weak acid the. Example anionofofa salt theofacid, and acid the cation of a strong base (Na. OH, KOH) Hydrofluoric HF KF – Potassium fluoride Formic HCOOH KHCOO – Potassium formate Benzoic C 6 H 5 COOH Na. C 6 H 5 COO – Sodium benzoate Acetic CH 3 COOH Na. H 3 COO – Sodium acetate Carbonic H 2 CO 3 Na. HCO 3 – Sodium bicarbonate Propanoic HC 3 H 5 O 2 Na. C 3 H 5 O 2 – Sodium propanoate Hydrocyanic HCN KCN – Potassium cyanide

Base/Salt Buffering Pairs l The Base salt will contain cation the base, Formula of the Example of aof salt of the weak base the base and the anion of a strong acid (HCl, HNO 3) Ammonia NH 3 NH 4 Cl – ammonium chloride Methylamine CH 3 NH 2 Cl - Methylammonium chloride Ethylamine C 2 H 5 NH 2 C 2 H 5 NH 3 NO 3 - Ethylammonium nitrate Aniline C 6 H 5 NH 2 C 6 H 5 NH 3 Cl - Aniline hyrdrochloride Pyridine C 5 H 5 NHCl - Pyridine hydrochloride

Buffered Solutions - Example l. A 0. 100 M solution of ethanoic acid (Ka = 1. 8 x 10 -5) is mixed with a solution of 0. 100 M potassium ethanoate. Calculate the p. H of the resulting solution.
![Henderson-Hasselbalch Equation p. H = p. Ka + log [A-] = p. Ka + Henderson-Hasselbalch Equation p. H = p. Ka + log [A-] = p. Ka +](http://slidetodoc.com/presentation_image/d8180c66e7481226ff273a2e9db0f131/image-52.jpg)
Henderson-Hasselbalch Equation p. H = p. Ka + log [A-] = p. Ka + log [salt] [HA] [acid] p. OH = p. Kb + log [BH+] = p. Kb + log [salt] [B] [base]

Acid-Base Properties of Salts Type of Salt Examples Comment p. H of l These salts simply dissociate in water: solution Cation is from a strong base, anion from a strong acid l KCl, KNO 3, Na. Cl, Na. NO 3 KCl(s) K+(aq) + Cl-(aq) Both ions neutral are neutral

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is from Na. C 2 H 3 O 2 a strong base, KCN, anion from a Na. F weak acid l Cation is neutral, anion is basic p. H of solution basic The basic anion can accept a proton from water: C 2 H 3 O 2 - + H 2 O HC 2 H 3 O 2 + OH-

Acid-Base Properties of Salts Type of Salt Examples Comment p. H of solution acidic NH 4 Cl, Cation is the Cation is conjugate acid NH 4 NO 3 acidic, of a weak anion is base, anion is neutral from a strong acid l The acidic cation can act as a proton donor: NH 4+ NH 3 + H+

Acid-Base Properties of Salts Type of Salt Examples Comment p. H of solution Cation is the NH 4 C 2 H 3 O 2 Cation is See below conjugate acid of NH 4 CN acidic, anion a weak base, is basic anion is conjugate base of a weak acid IF Ka for the acidic ion is greater than Kb for the basic ion, the solution is acidic l IF Kb for the basic ion is greater than Ka for the acidic ion, the solution is basic l IF Kb for the basic ion is equal to Ka for the acidic ion, the solution is neutral l

Acid-Base Properties of Salts Type of Salt Examples Cation is a highly Al(NO 3)2; charged metal Fe. Cl 3 ion; anion is from a strong acid l Comment Hydrated cation acts as an acid; anion is neutral p. H of solution acidic Step #1: l Al. Cl 3(s) + 6 H 2 O Al(H 2 O)63+(aq) + Cl-(aq) Salt l Step #2: l water Complex ion anion Al(H 2 O)63+(aq) Al(OH)(H 2 O)52+(aq) + H+(aq) Acid Conjugate base Proton
- Slides: 57