Acids and Bases Chapter 15 Acids and Bases

Acids and Bases

Chapter 15 Acids and Bases 15. 2 the Acids and Bases properties of water 15. 3 PH- a measure of acidity

Acids and Bases • Acid: Substance that produces hydrogen ions in water solution. • Base: Substance that produces hydroxide ions in water solution. • An acid neutralizes a base + → H 2 O(ℓ)

15. 2 the Acids and Bases properties of water • water is a unique solvent , it can act as acid or base. • In pure water, a few molecules act as bases and a few act as acids. acid(1) + Hydronium ion base (2) ⇄ acid(2) + base (1) • This is referred to as autoionization of water ( )ﺍﻟﺘﺄﻴﻦ ﺍﻟﺬﺍﺗﻲ • The equilibrium expression for this process is • This special equilibrium constant is referred to as the ion-product constant for water, Kw At 25 °C, Kw = 1. 0 10− 14 ﺛﺎﺑﺖ ﺍﻟﺤﺎﺻﻞ ﺍﻻﻳﻮﻧﻲ ﻟﻠﻤﺎﺀ

The Ion-Product of Water q In pure water, q In pure water [H 3 O+] = [OH−] (neutral) q In q acidic solution: In basic solution
![Example 15. 2 a: Calculate the [H+] ions in ammonia, [OH-] =0. 0025 M? Example 15. 2 a: Calculate the [H+] ions in ammonia, [OH-] =0. 0025 M?](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-6.jpg)
Example 15. 2 a: Calculate the [H+] ions in ammonia, [OH-] =0. 0025 M? Solution: [H+] = 4 x 10 -12; [OH-] =2. 5 x 10 -3 [H+] < [OH-] the solution is basic
![Example 15. 2 b: Calculate the [OH-] ions in 1. 3 M HCl ? Example 15. 2 b: Calculate the [OH-] ions in 1. 3 M HCl ?](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-7.jpg)
Example 15. 2 b: Calculate the [OH-] ions in 1. 3 M HCl ? Solution: [H+] = 1. 3; [OH-] =7. 7 x 10 -15 [H+] > [OH-] the solution is Acidic

15. 3 p. H-A Measure of Acidity • p. H of a solution: is the negative logarithm (base- 10) of the hydrogen ion (hydronium ion) concentration. • In the same manner

15. 3 p. H-A Measure of Acidity • In pure water,
![Example 15. 3 a: The [H+] = 3. 2 x 10– 4 M. The Example 15. 3 a: The [H+] = 3. 2 x 10– 4 M. The](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-10.jpg)
Example 15. 3 a: The [H+] = 3. 2 x 10– 4 M. The [H+]=1. 0 x 10– 3 M. What is the p. H in the two occasions. Solution: Thus: p. H is inversely proportional to [H+] increase, p. H decrease >>> more acidic
![Example 15. 3 b: The [H+] = 0. 76 M, in nitric acid solutions Example 15. 3 b: The [H+] = 0. 76 M, in nitric acid solutions](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-11.jpg)
Example 15. 3 b: The [H+] = 0. 76 M, in nitric acid solutions What is the p. H? Solution: Thus: p. H is inversely proportional to [H+] increase, p. H decrease >>> more acidic
![p. H Range p. H is inversely proportional to [H+] 0 1 2 3 p. H Range p. H is inversely proportional to [H+] 0 1 2 3](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-12.jpg)
p. H Range p. H is inversely proportional to [H+] 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Acidic Increase the acidity [H+] > [OH-] p. H < 7 Basic Increase the basicity Neutral [H+] = [OH-] p. H = p. OH = 7 [OH-]>[H+] p. H > 7

Example 15. 4 a: The p. H = 4. 82 , What is the [H+] of the rain water. Solution: In the calculator: Press SHEFT → log button → (-ve ) value (4. 82)=
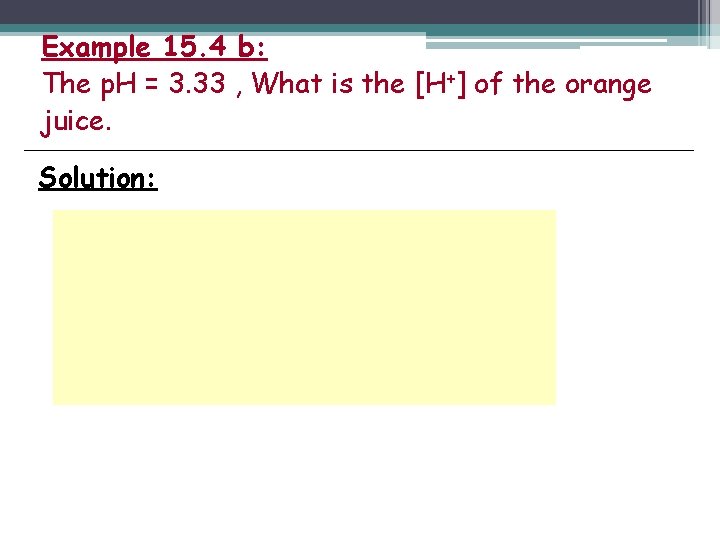
Example 15. 4 b: The p. H = 3. 33 , What is the [H+] of the orange juice. Solution:
![Example 15. 5 a: The [OH-] = 2. 9 x 10– 4 M. What Example 15. 5 a: The [OH-] = 2. 9 x 10– 4 M. What](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-15.jpg)
Example 15. 5 a: The [OH-] = 2. 9 x 10– 4 M. What is the p. H of the Na. OH solution. Solution: OR p. H = 10. 46 > 7 BASIC (Na. OH)
![Example 15. 5 b: The [OH-] = 2. 5 x 10– 7 M. What Example 15. 5 b: The [OH-] = 2. 5 x 10– 7 M. What](http://slidetodoc.com/presentation_image/349a130465607d77fb1efad39b46f578/image-16.jpg)
Example 15. 5 b: The [OH-] = 2. 5 x 10– 7 M. What is the p. H of the blood solution. Solution: OR p. H = 7. 4 ~ 7 neutral (blood)
- Slides: 16