Acids and Bases Chapter 14 DE Chemistry Dr

Acids and Bases Chapter 14 DE Chemistry Dr. Walker

Acid – Base Terminology • Arrhenius – Acids produce hydrogen ions in aqueous solutions – Based produce hydroxide ions in aqueous solutions • Bronsted-Lowry – Acids are proton donors – Bases are proton acceptors • Lewis – Acids are electron pair acceptors – Bases are electron pair donors

Conjugate Acids and Bases HCl + H 2 O Acid Base Cl+ H 3 O+ Conjugate Base Acid Conjugate Base – Formed when acid loses a proton Conjugate Acid – Formed when base gains a proton
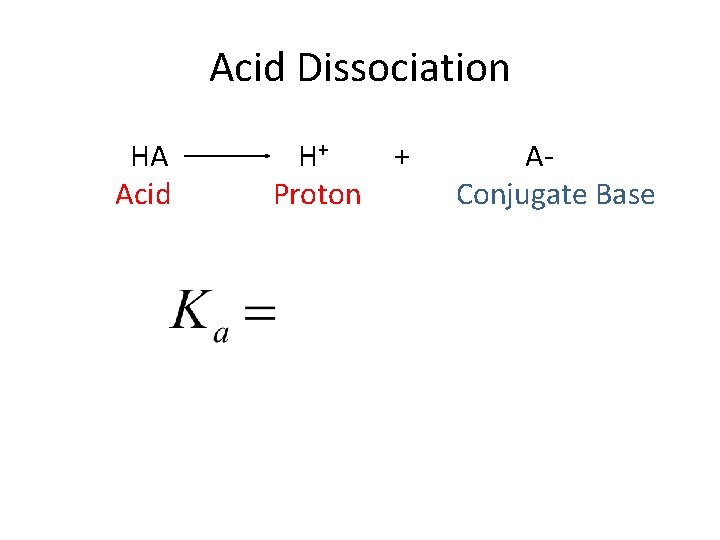
Acid Dissociation HA Acid H+ Proton + AConjugate Base

Acid Dissociation HA Acid H+ Proton + AConjugate Base -Based on equilbirium constants from last chapter -Ka is very high for strong acids, lower for weak acids

Dissociation of Strong Acids Strong acids are assumed to dissociate completely in solution. Large Ka or small Ka? Reactant favored or product favored?
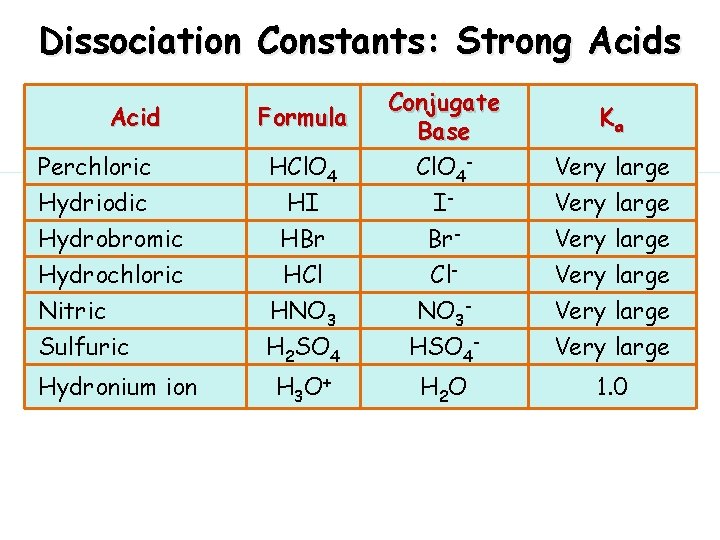
Dissociation Constants: Strong Acids Acid Formula Perchloric Hydriodic HCl. O 4 HI Conjugate Base Cl. O 4 - I- Hydrobromic Hydrochloric Nitric Sulfuric HBr HCl HNO 3 H 2 SO 4 Br- Cl- NO 3 - HSO 4 - Very large Hydronium ion H 3 O+ H 2 O 1. 0 Ka Very large
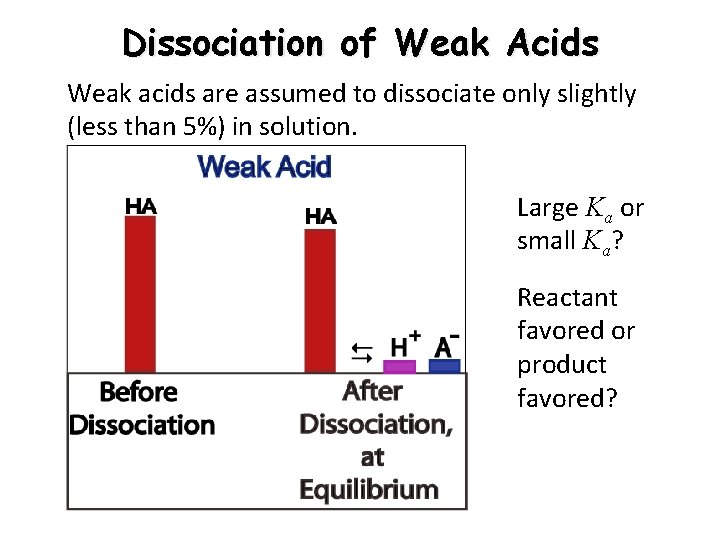
Dissociation of Weak Acids Weak acids are assumed to dissociate only slightly (less than 5%) in solution. Large Ka or small Ka? Reactant favored or product favored?
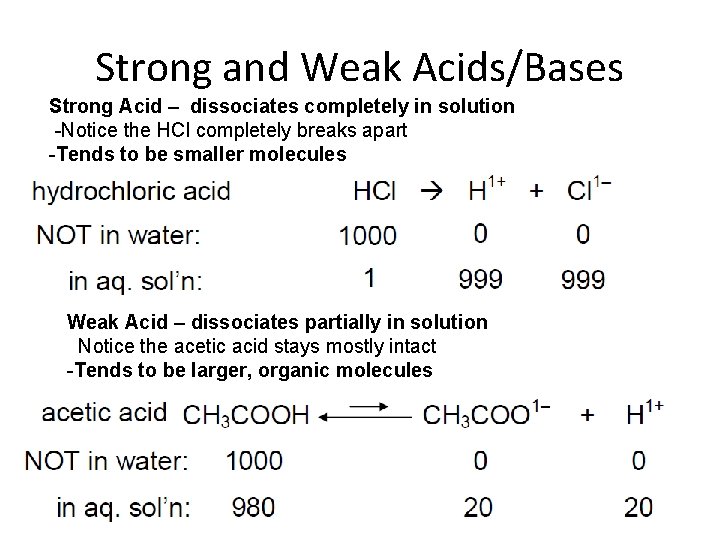
Strong and Weak Acids/Bases Strong Acid – dissociates completely in solution -Notice the HCl completely breaks apart -Tends to be smaller molecules Weak Acid – dissociates partially in solution Notice the acetic acid stays mostly intact -Tends to be larger, organic molecules
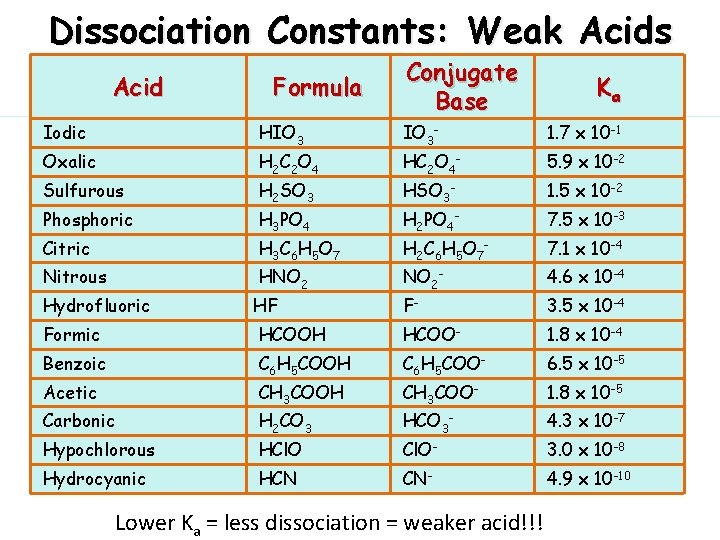
Dissociation Constants: Weak Acids Acid Formula Conjugate Base Ka Iodic HIO 3 IO 3 - 1. 7 x 10 -1 Oxalic H 2 C 2 O 4 HC 2 O 4 - 5. 9 x 10 -2 Sulfurous H 2 SO 3 HSO 3 - 1. 5 x 10 -2 Phosphoric H 3 PO 4 H 2 PO 4 - 7. 5 x 10 -3 Citric H 3 C 6 H 5 O 7 H 2 C 6 H 5 O 7 - 7. 1 x 10 -4 Nitrous HNO 2 NO 2 - 4. 6 x 10 -4 Hydrofluoric HF F- 3. 5 x 10 -4 Formic HCOOH HCOO- 1. 8 x 10 -4 Benzoic C 6 H 5 COOH C 6 H 5 COO- 6. 5 x 10 -5 Acetic CH 3 COOH CH 3 COO- 1. 8 x 10 -5 Carbonic H 2 CO 3 HCO 3 - 4. 3 x 10 -7 Hypochlorous HCl. O Cl. O- 3. 0 x 10 -8 Hydrocyanic HCN CN- 4. 9 x 10 -10 Lower Ka = less dissociation = weaker acid!!!

Acid Terminology • Polyprotic – Acids with more than one ionizable proton • Monoprotic - one acidic proton (HCl) • Diprotic - two acidic protons (H 2 SO 4) • Triprotic - three acidic protons (H 3 PO 4) • Oxyacids - acids in which the acidic proton is attached to an oxygen atom (HCl. O 3) Ionizable Hydrogen http: //withfriendship. com/images/j/45366/of-perchloric-acid. png

Polyprotic Acids • When multiple acidic protons are present, the removal of each one gives a different equilibrium constant • The first one will always have the highest (strongest) equilibrium constant
Acid Terminology • Organic acids - acids containing the mildly acidic carboxyl group – Generally weak acids – Equilibrium lies far to the left (weak acids) http: //boomeria. org/chemlectures/organic/acids. jpg

Self-Ionization of Water • H 2 O + H 2 O H 3 O+ + OH- – This happens in every glass of water – This occurs in low enough concentration that it is noticeable – [H 3 O+] = [OH-] = 1. 0 x 10 -7 M – [H 3 O+] [OH-] = 1. 0 x 10 -14 M

Dissociation of Strong Bases MOH(s) M+(aq) + OH-(aq) § Strong bases are metallic hydroxides § Group I hydroxides (Na. OH, KOH) are very soluble § Group II hydroxides (Ca, Ba, Mg, Sr) are less soluble § p. H of strong bases is calculated directly from the concentration of the base in solution
![Calculating p. H, p. OH p. H = -log[H 3 O+] p. OH = Calculating p. H, p. OH p. H = -log[H 3 O+] p. OH =](http://slidetodoc.com/presentation_image/0eae8e5d2e593ae5e4a6398ece7fa6d0/image-16.jpg)
Calculating p. H, p. OH p. H = -log[H 3 O+] p. OH = -log[OH-] Relationship between p. H and p. OH p. H + p. OH = 14 Finding [H 3 O+], [OH-] from p. H, p. OH [H 3 O+] = 10 -p. H [OH-] = 10 -p. OH
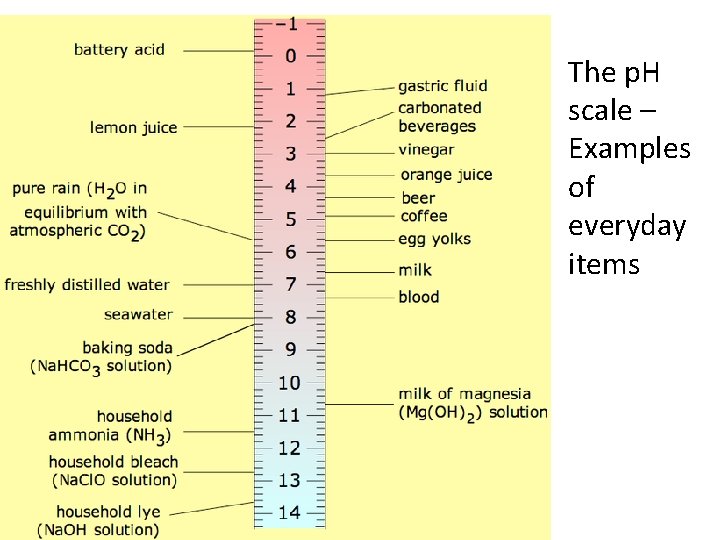
• The p. H scale – Examples of everyday items

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #1: Write the dissociation equation HC 2 H 3 O 2 C 2 H 3 O 2 - + H+

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #2: ICE it! HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ I C E 0. 50 -x 0. 50 - x 0 0 +x +x x x

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #3: Set up the law of mass action HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ E 0. 50 - x x x For weak acids, dissociation is small enough that It is assumed that a weak acid (acetic acid, here), dissociates less than 5%

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #4: Solve for x, which is also [H+] E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ x x 0. 50 - x [H+] = 3. 0 x 10 -3 M

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #5: Convert [H+] to p. H E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ x x 0. 50 - x

Reaction of Weak Bases with Water The base reacts with water, producing its conjugate acid and hydroxide ion: CH 3 NH 2 + H 2 O CH 3 NH 3+ + OH- Kb = 4. 38 x 10 -4
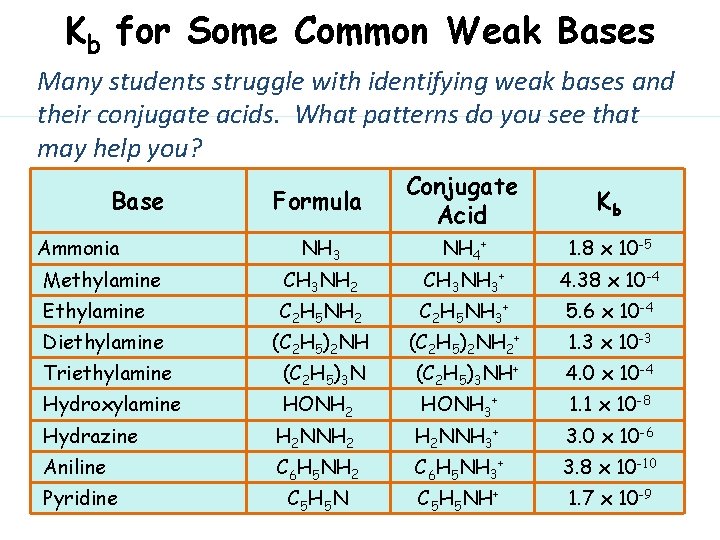
Kb for Some Common Weak Bases Many students struggle with identifying weak bases and their conjugate acids. What patterns do you see that may help you? Formula Conjugate Acid Kb NH 3 NH 4+ 1. 8 x 10 -5 Methylamine CH 3 NH 2 CH 3 NH 3+ 4. 38 x 10 -4 Ethylamine C 2 H 5 NH 2 C 2 H 5 NH 3+ 5. 6 x 10 -4 Diethylamine (C 2 H 5)2 NH 2+ 1. 3 x 10 -3 Triethylamine (C 2 H 5)3 NH+ 4. 0 x 10 -4 Hydroxylamine HONH 2 HONH 3+ 1. 1 x 10 -8 Base Ammonia Hydrazine H 2 NNH 2 H 2 NNH 3+ 3. 0 x 10 -6 Aniline C 6 H 5 NH 2 C 6 H 5 NH 3+ 3. 8 x 10 -10 Pyridine C 5 H 5 NH+ 1. 7 x 10 -9

A Weak Base Equilibrium Problem What is the p. H of a 0. 10 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #1: Write the equation for the reaction NH 3 + H 2 O NH 4+ + OH-

A Weak Base Equilibrium Problem What is the p. H of a 0. 10 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #2: ICE it! I C NH 3 + H 2 O NH 4+ + OH 0. 10 0 +x -x E 0. 10 - x x 0 +x x

A Weak Base Equilibrium Problem What is the p. H of a 0. 10 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #3: Set up the law of mass action NH 3 + H 2 O NH 4+ + OHE 0. 10 - x x x

A Weak Base Equilibrium Problem What is the p. H of a 0. 10 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #4: Solve for x, which is also [OH-] NH 3 + H 2 O NH 4+ + OHE 0. 10 - x x x [OH-] = 1. 3 x 10 -3 M

A Weak Base Equilibrium Problem What is the p. H of a 0. 10 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #5: Convert [OH-] to p. H NH 3 + H 2 O NH 4+ + OHE 0. 10 - x x x
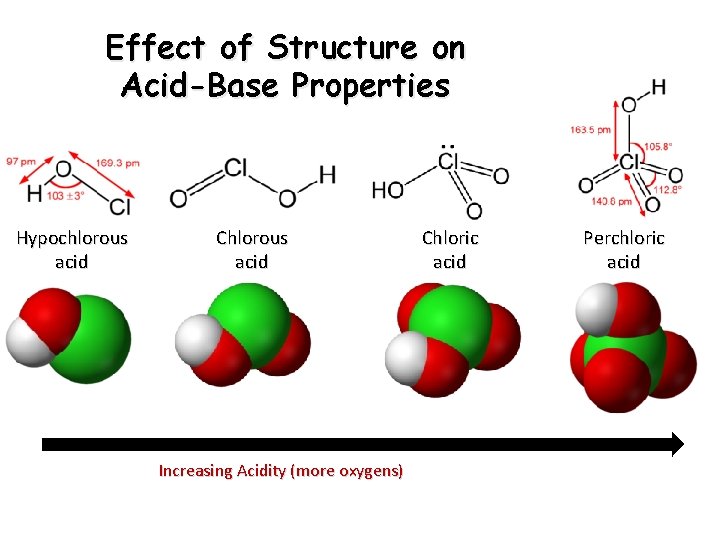
Effect of Structure on Acid-Base Properties Hypochlorous acid Chlorous acid Increasing Acidity (more oxygens) Chloric acid Perchloric acid

Acid-Base Properties of Oxides • Acidic Oxides (also called acid anhydrides) – Nonmetal oxides that react with water to form acidic solutions – SO 3(g) + H 2 O(l) H 2 SO 4(aq) – 2 NO 2(g) + H 2 O(l) HNO 3(aq) + HNO 2(aq)

Acid-Base Properties of Oxides • Basic Oxides (also called basic anhydrides) – Metallic oxides of Group 1 A and 2 A metals react with water to form basic solutions – K 2 O(s) + H 2 O(l) 2 KOH (aq) – Ca. O(s) + H 2 O(l) Ca(OH)2(aq)

Titrations • Type of quantitative analysis • Start with known concentration and volume of a solution, known as a titrant – Titrant can be acid or base • Add to a sample solution with unknown concentration, known as an analyte • Analyte will be the opposite of the titrant • Reaches the endpoint when the indicator turns color – Indicator: Dye that changes color with a change in p. H

A Titration Pictured • Left: student performing a titration. The titrant is in a long tube called a buret. The analyte is in the Erlenmeyer flask below. • Right: A close-up view of a buret, measuring in 0. 1 m. L increments.
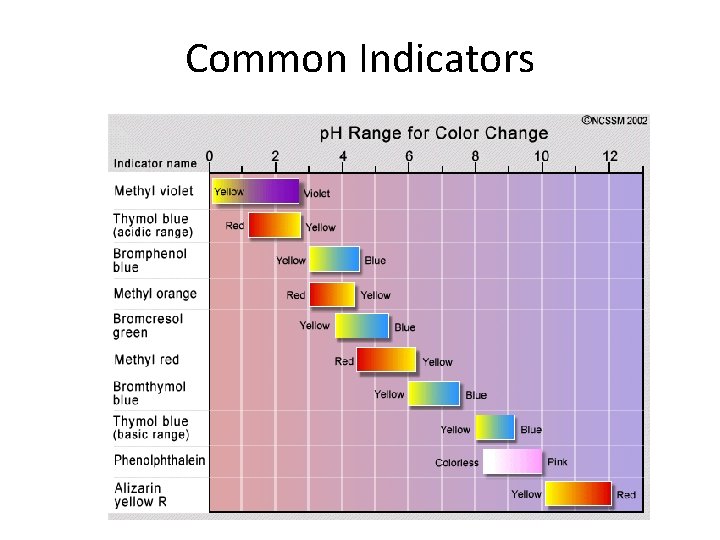
Common Indicators

Calculations • The most common calculation involves using dilutions • Example: 32. 5 m. L of 0. 1 M HCl solution is required to titrate 100 m. L of a base. What is the concentration of the base?

Calculations • The most common calculation involves using dilutions • Example: 32. 5 m. L of 0. 1 M HCl solution is required to titrate 50 m. L of a base. What is the concentration of the base? – M 1 V 1 = M 2 V 2 – (0. 1 M)(32. 5 m. L) = M 2 (50 m. L) – M 2 = 0. 065 M
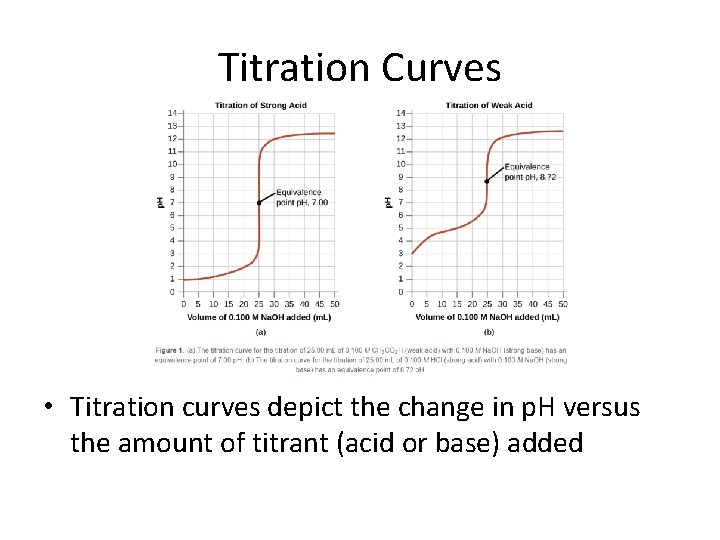
Titration Curves • Titration curves depict the change in p. H versus the amount of titrant (acid or base) added
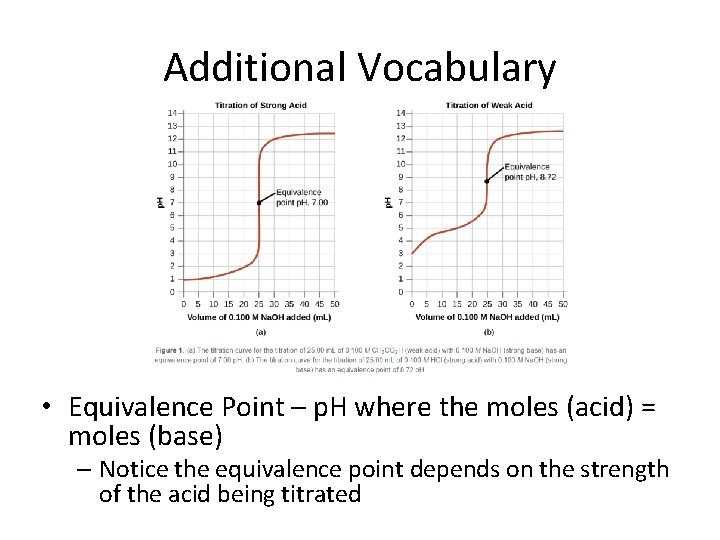
Additional Vocabulary • Equivalence Point – p. H where the moles (acid) = moles (base) – Notice the equivalence point depends on the strength of the acid being titrated
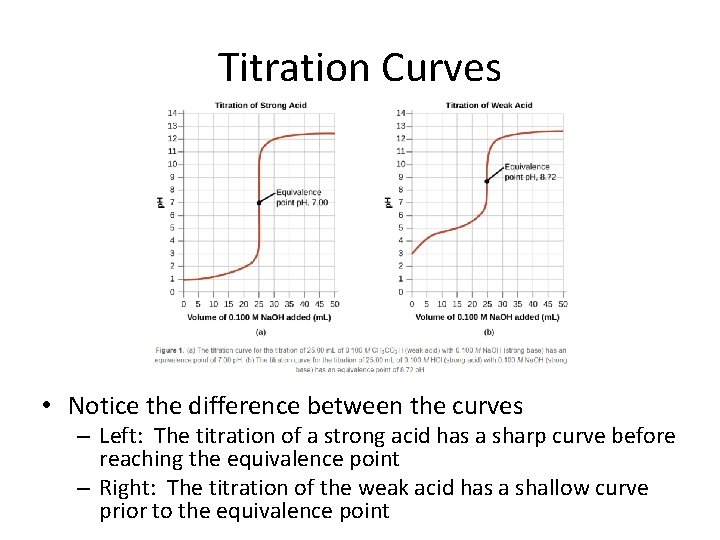
Titration Curves • Notice the difference between the curves – Left: The titration of a strong acid has a sharp curve before reaching the equivalence point – Right: The titration of the weak acid has a shallow curve prior to the equivalence point

Polyprotic Acids • Polyprotic acids have multiple Ka values – one for each ionizable proton • Each ionizable proton has a separate equivalence point at a different p. H
- Slides: 41