Acids and Bases Ch 19 General Properties ACIDS

Acids and Bases Ch. 19 General Properties ACIDS Taste sour

Acid • • • A substance that donates (gives up) H+ ions Ex: HCl + H 2 O H+ + Cl- + H 2 O High concentration of H+ ions On the p. H table: 0 ≈ strong acid 6. 9 ≈ weak acid A note about hydronium ion (H 3 O+): in water H+ H 3 O+ so they are the same thing!!!

Base • • • A substance that accepts (bonds with) H+ ions Ex: Na. OH + H+ Na+ + H 2 O Low concentration of H+ ions (they all bonded) On the p. H table: 14 ≈ strong base 7. 1 ≈ weak base Amphoteric – a substance that can act as both an acid and base- as water shows

Acids + bases come in pairs • General equation is: HX(aq) + H 2 O(l) ↔ H 3 O+(aq) + X-(aq) Acid + Base ↔ Conjugate + Conjugate acid base • Ex: HCl + Na. OH ↔ H 2 O

Acids and bases come in pairs • A “conjugate base” is the remainder of the original acid, after it donates it’s hydrogen ion • A “conjugate acid” is the particle formed when the original base gains a hydrogen ion
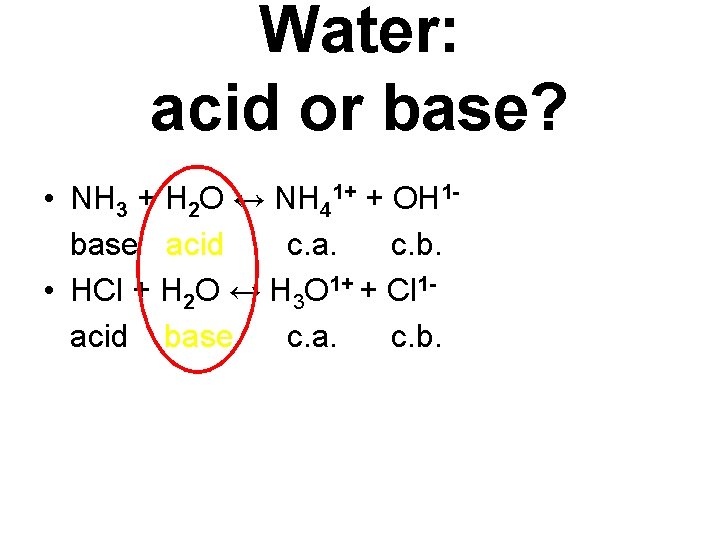
Water: acid or base? • NH 3 + H 2 O ↔ NH 41+ + OH 1 base acid c. a. c. b. • HCl + H 2 O ↔ H 3 O 1+ + Cl 1 acid base c. a. c. b.

Measuring strength • Ionization is reversible: HA + H 2 O ↔ H+ + Athat the arrow • This makes an equilibrium (Note goes both directions. ) • Acid dissociation constant = Ka (Note that water is NOT shown, • Ka = [H+ ][A- ] because its concentration is [HA] constant, and built into Ka) • Stronger acid = more products (ions), thus a larger Ka

What about bases? • Strong bases dissociate completely. • MOH + H 2 O ↔ M+ + OH- (M = a metal) • Base dissociation constant = Kb • Kb = [M+ ][OH-] [MOH] • Stronger base = more dissociated ions are produced, thus a larger Kb.
- Slides: 8