Acids and Bases Ch 15 Slides Acids and

Acids and Bases Ch 15: Slides (Acids and Bases) 1

Acid/Base Definitions Lewis • Arrhenius Acids and Bases Bronsted. Lowry • Acid: Released H+ in water (H 3 O+) • Base: Releases OH- in water • Bronsted-Lowry • Acid: Proton (H+) Donor • Base: Proton (H+) Acceptor Arrhenius • Lewis • Acid: Electron pair Acceptor • Base: Electron pair Acceptor Ch 15: Slides (Acids and Bases) 2
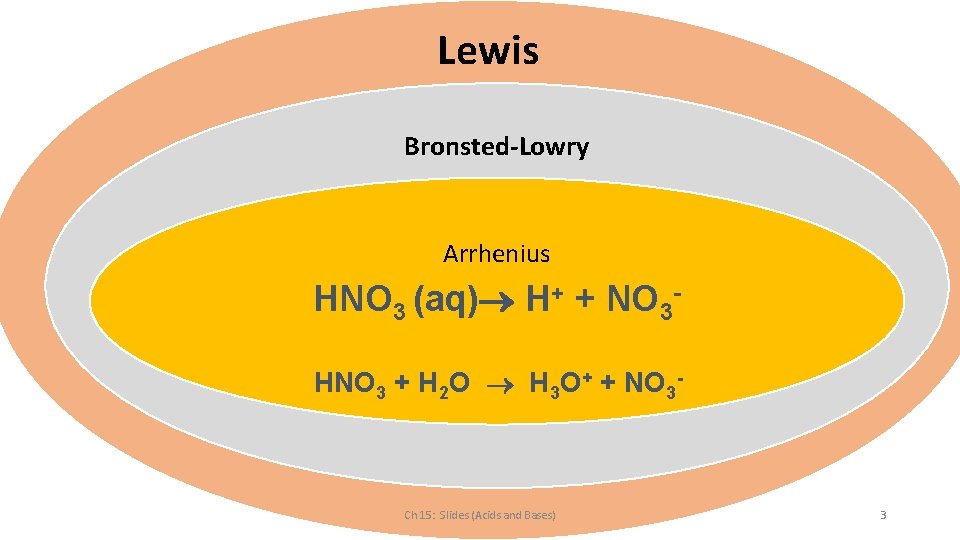
Lewis Bronsted-Lowry Arrhenius HNO 3 (aq) H+ + NO 3 HNO 3 + H 2 O H 3 O+ + NO 3 - Ch 15: Slides (Acids and Bases) 3
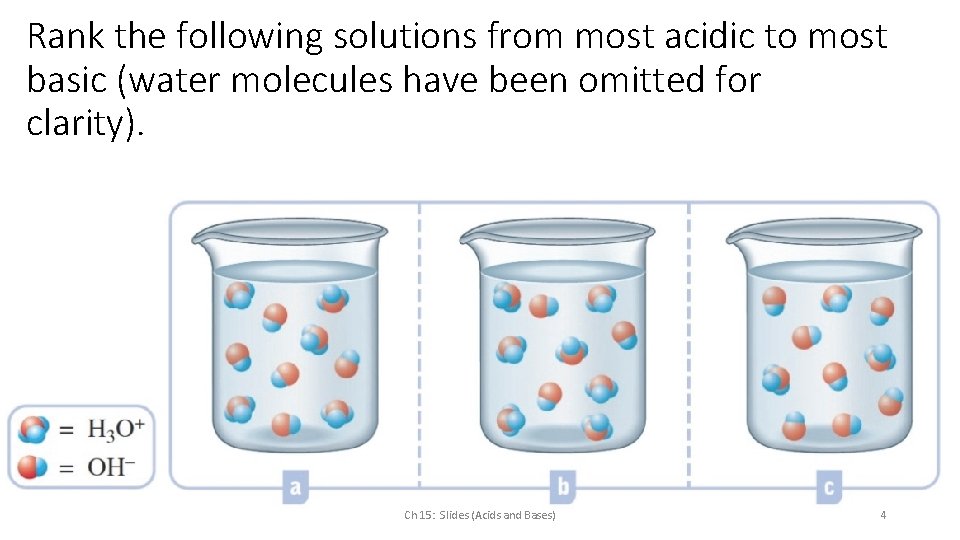
Rank the following solutions from most acidic to most basic (water molecules have been omitted for clarity). Ch 15: Slides (Acids and Bases) 4
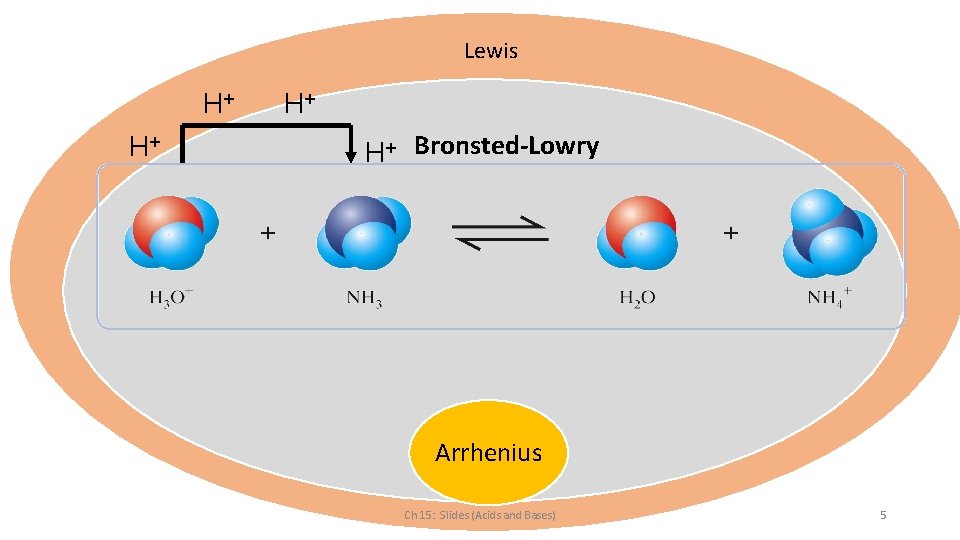
Lewis H+ H+ H+ Bronsted-Lowry + H Arrhenius Ch 15: Slides (Acids and Bases) 5
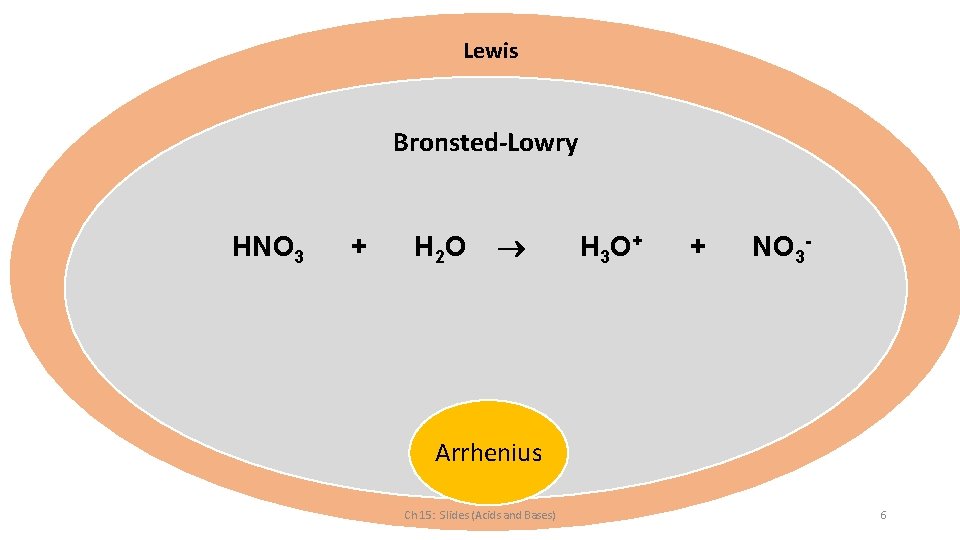
Lewis Bronsted-Lowry HNO 3 + H 2 O H 3 O + + NO 3 - Arrhenius Ch 15: Slides (Acids and Bases) 6

Codeine, C 18 H 21 O 3 N is a BL base… What is the conjugate acid of codeine? Write a net ionic equation to show codeine behaving as a BL base in water. Identify the acid/base pairs Ch 15: Slides (Acids and Bases) 7

Water is amphoteric • Give an example of water acting as an ACID (BL) • Give an example of water acting as a BASE (BL) Ch 15: Slides (Acids and Bases) 8
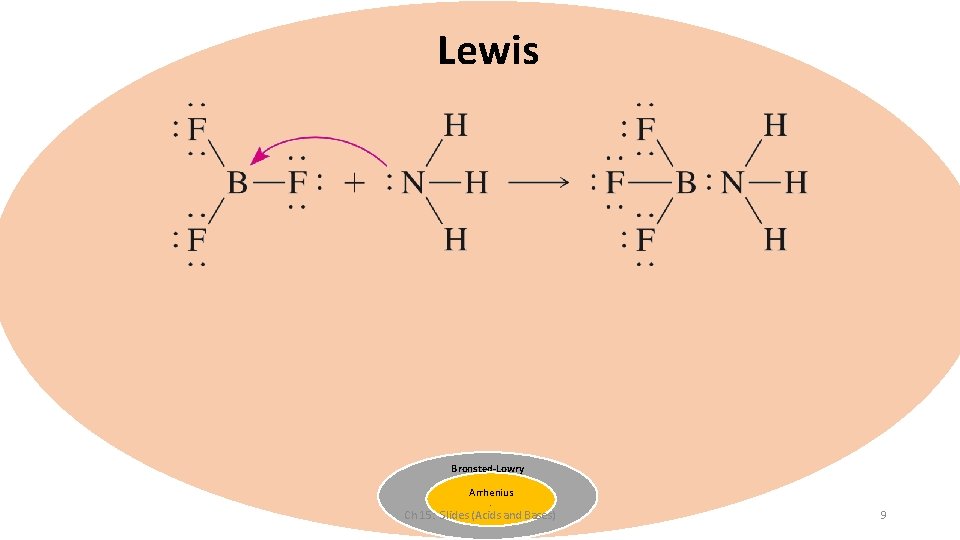
Lewis Bronsted-Lowry Arrhenius - Ch 15: Slides (Acids and Bases) 9

Ammonia is A Base? By All three definitions Write an example showing each definition: Ch 15: Slides (Acids and Bases) 10
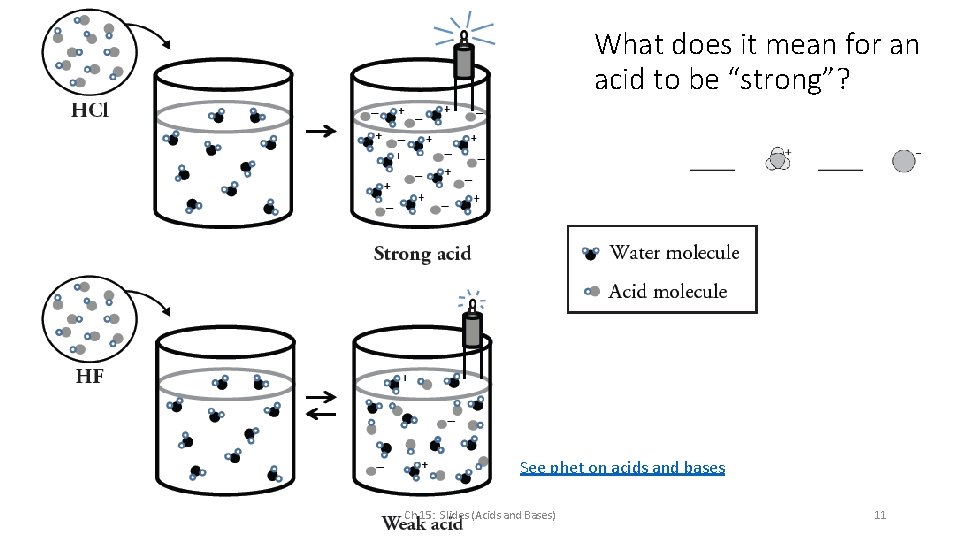
What does it mean for an acid to be “strong”? See phet on acids and bases Ch 15: Slides (Acids and Bases) 11

What does it mean for an acid to be “strong”? HCl is a ________ because: Write the reaction (BL) Ch 15: Slides (Acids and Bases) 12

What does it mean for an acid to be “weak”? HF is a ________ because: Write the reaction (BL) Ch 15: Slides (Acids and Bases) 13
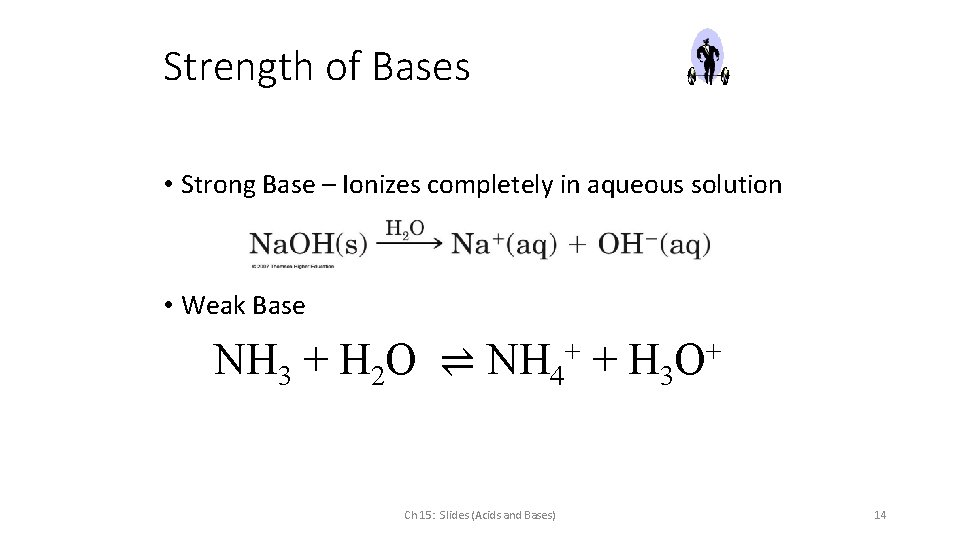
Strength of Bases • Strong Base – Ionizes completely in aqueous solution • Weak Base NH 3 + H 2 O ⇌ NH 4+ + H 3 O+ Ch 15: Slides (Acids and Bases) 14

The Strong Acids/Bases These should be Memorized! • Strong acids • HCl. O 4 • H 2 SO 4 • HNO 3 • HCl • HBr • HI • Strong bases • Li. OH • Na. OH • KOH • Ca(OH)2 • Sr(OH)2 • Ba(OH)2 If it does not appear on this list it is WEAK! Ch 15: Slides (Acids and Bases) 15

Relative Strengths of Acids and Bases The stronger an acid, the weaker its conjugate base. The weaker an acid, the stronger its conjugate base. Ch 15: Slides (Acids and Bases) 16
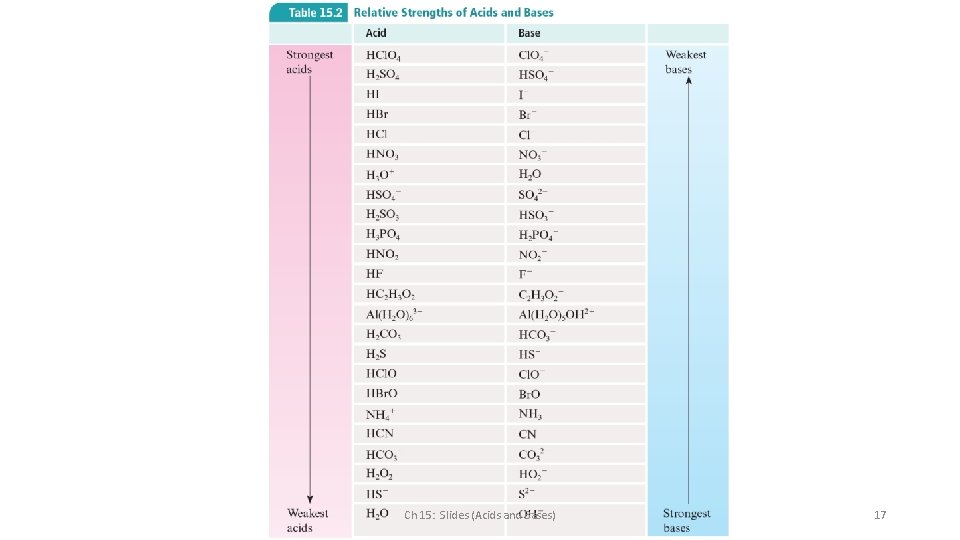
Ch 15: Slides (Acids and Bases) 17

Write the BL reaction equation for: Generic weak acid in water. Generic anion (base) in water. BL reaction of formic acid with water. BL reaction of Acetic acid in water. BL reaction equation of formate ion in water. Write the reaction of acetate ion in water. Formic acid, HCHO 2, is a stronger acid than acetic acid, HC 2 H 3 O 2. Which is the stronger base: formate ion, CHO 2−, or acetate ion, C 2 H 3 O 2−? The acetate ion is a stronger base than the formate ion. Ch 15: Slides (Acids and Bases) 18

Structure and Acid Strength : The issue HOW EASILY IS THE PROTON REMOVED Easily removed = _______ Tightly held = _________ Ch 15: Slides (Acids and Bases) 19

#1 Strength of bond depends on size of atom How does bond strength correspond to acid strength? • H-X; large X = strong acid • Within a column (group), size is the main issue Compare acid strength HF, HCl, HBr, HI Ch 15: Slides (Acids and Bases) 20
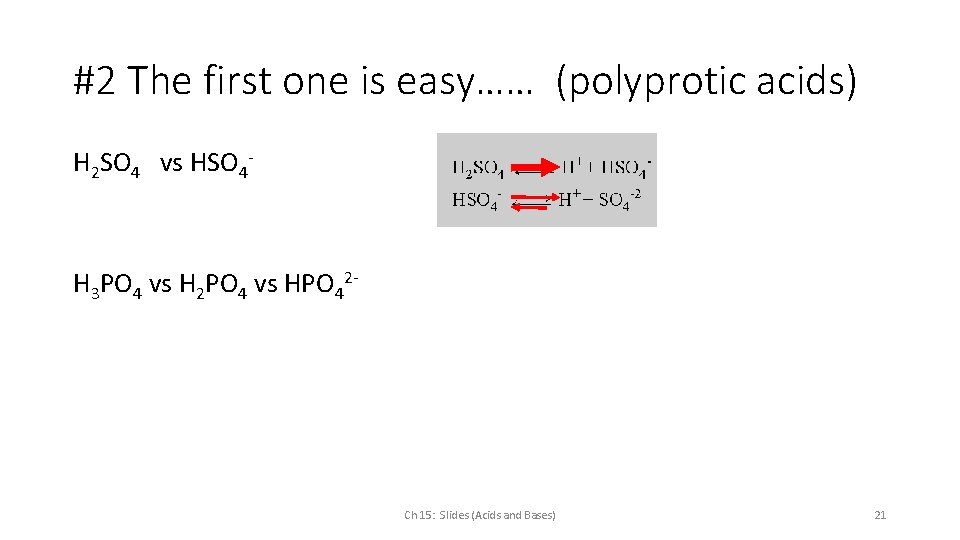
#2 The first one is easy…… (polyprotic acids) H 2 SO 4 vs HSO 4 - H 3 PO 4 vs H 2 PO 4 vs HPO 42 - Ch 15: Slides (Acids and Bases) 21
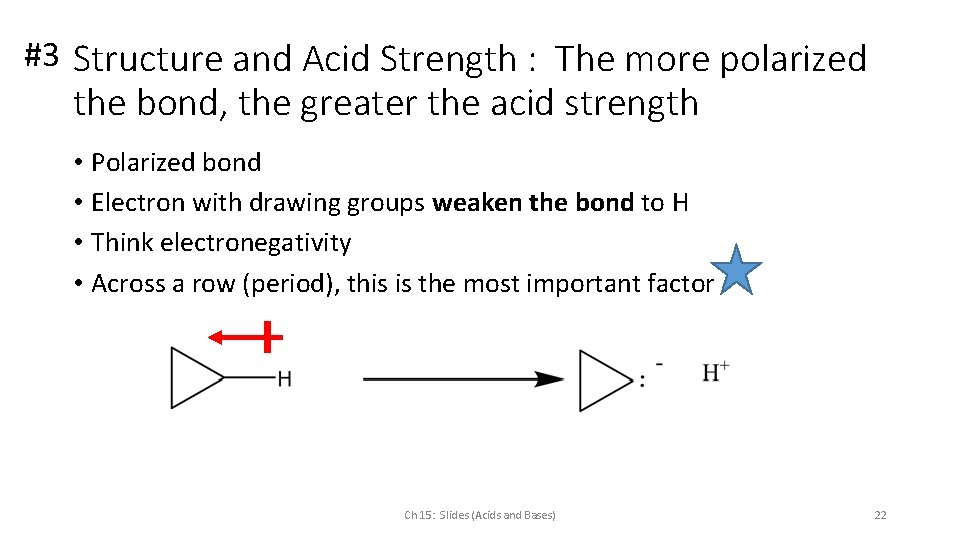
#3 Structure and Acid Strength : The more polarized the bond, the greater the acid strength • Polarized bond • Electron with drawing groups weaken the bond to H • Think electronegativity • Across a row (period), this is the most important factor Ch 15: Slides (Acids and Bases) 22
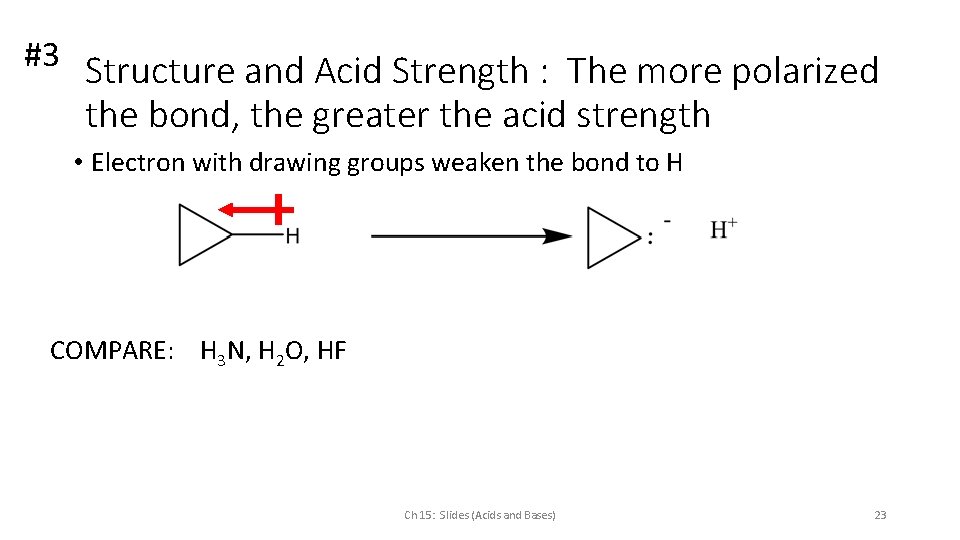
#3 Structure and Acid Strength : The more polarized the bond, the greater the acid strength • Electron with drawing groups weaken the bond to H COMPARE: H 3 N, H 2 O, HF Ch 15: Slides (Acids and Bases) 23
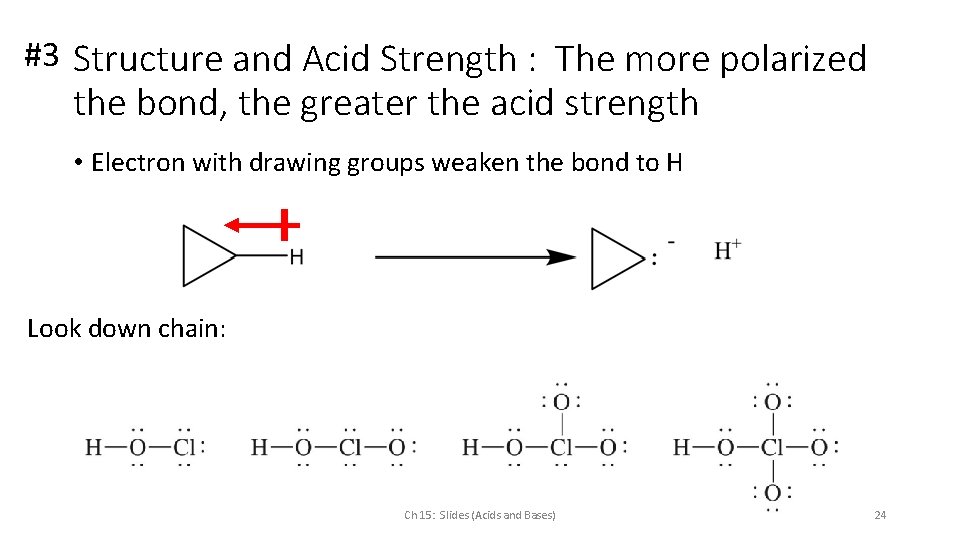
#3 Structure and Acid Strength : The more polarized the bond, the greater the acid strength • Electron with drawing groups weaken the bond to H Look down chain: Ch 15: Slides (Acids and Bases) 24

#3 Structure and Acid Strength : The more polarized the bond, the greater the acid strength • Electron with drawing groups weaken the bond to H Look down chain: Ch 15: Slides (Acids and Bases) 25
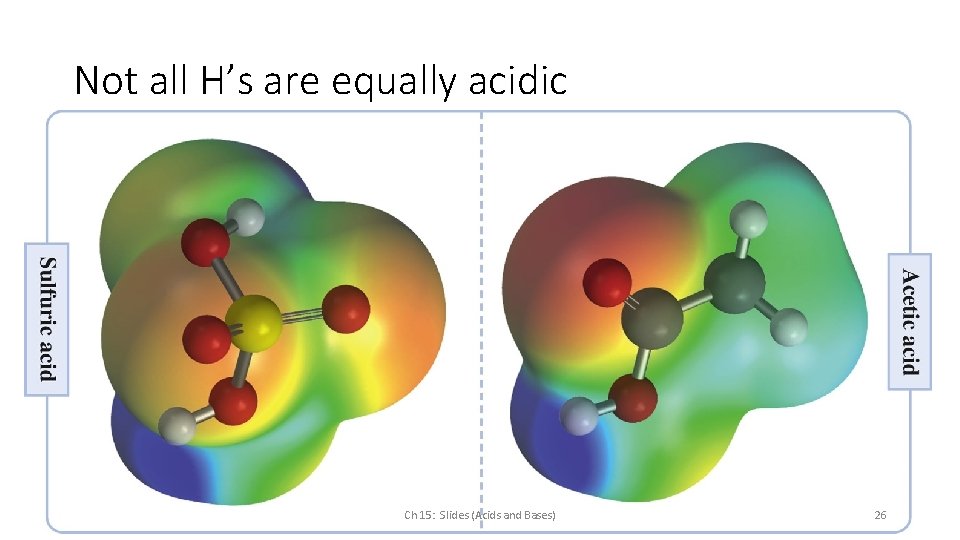
Not all H’s are equally acidic Ch 15: Slides (Acids and Bases) 26

The ISSUES SUMMARIZED 1. In a column/group SIZE matters (as X gets bigger, acid gets stronger) 2. In a row/period ELECTRON withdrawing matters (more electronegativity = stronger acid) Don’t forget to look down chain 3. In a polyprotic acid, the first one is easy (more H’s= stronger acid) Ch 15: Slides (Acids and Bases) 27
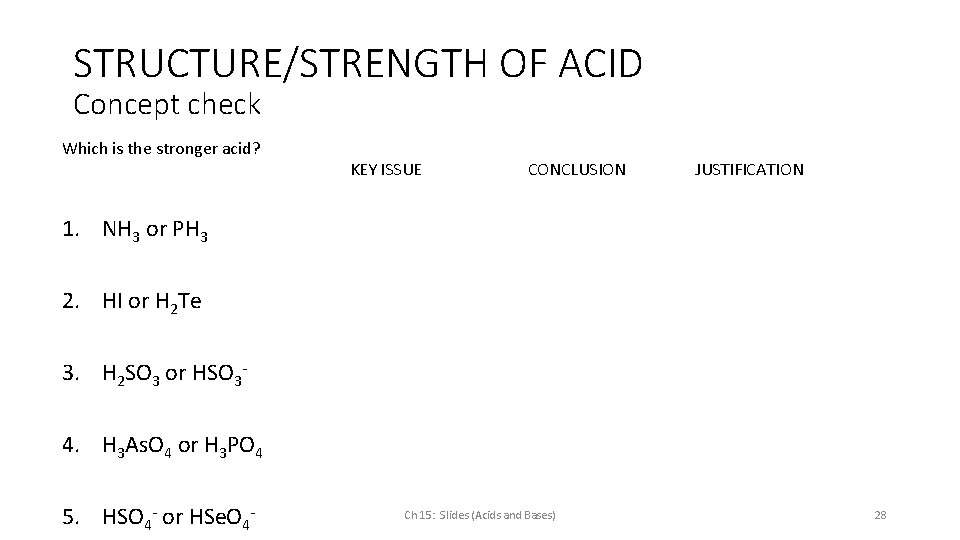
STRUCTURE/STRENGTH OF ACID Concept check Which is the stronger acid? KEY ISSUE CONCLUSION JUSTIFICATION 1. NH 3 or PH 3 2. HI or H 2 Te 3. H 2 SO 3 or HSO 34. H 3 As. O 4 or H 3 PO 4 5. HSO 4 - or HSe. O 4 - Ch 15: Slides (Acids and Bases) 28

IF the conjugate base is resonance stabilized THEN the acid is strong(er). . Info: Resonance structures that increase stability are those that delocalize electrons over the molecule and place negative charge on electronegative atoms. For example, HNO 3 is a strong acid and HNO 2 is a weak acid because HNO 3 has more resonance structures that place the negative charge on difference oxygen atoms. Ch 15: Slides (Acids and Bases) 29
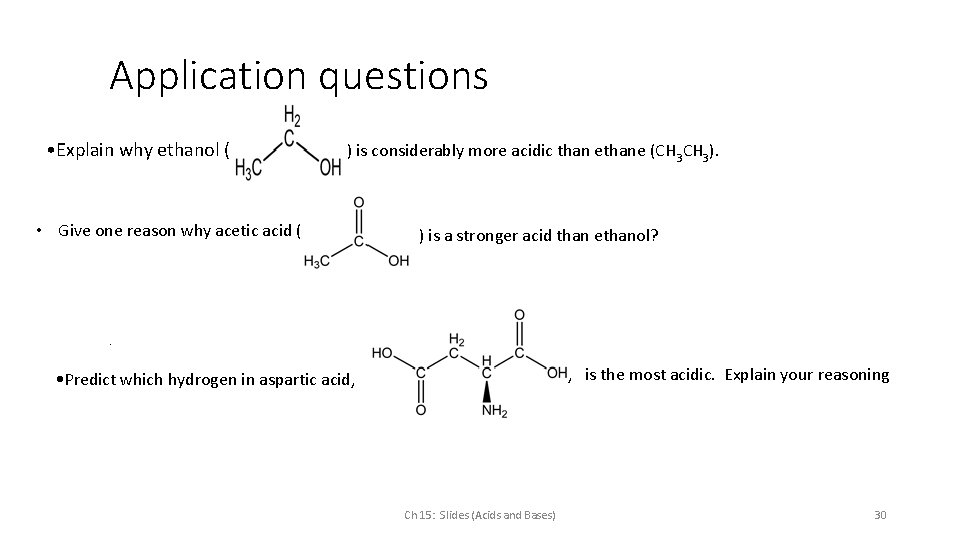
Application questions • Explain why ethanol ( ) is considerably more acidic than ethane (CH 3). • Give one reason why acetic acid ( ) is a stronger acid than ethanol? . , is the most acidic. Explain your reasoning • Predict which hydrogen in aspartic acid, Ch 15: Slides (Acids and Bases) 30

Determining the position of Equilibrium: The crabapple analogy • Think of the “crab apple” as the proton • Strength of acid refers to how quickly or aggressively those crab apples are thrown. • This is a “WAR” of the acids Ch 15: Slides (Acids and Bases) 31

Chapter 8: Acids and Bases 32

Boy Throws apples across the fence. Chapter 8: Acids and Bases 33

Old Man Fights Back. Boy is faster, but he has to run around gathering apples. Old man is a slow runner, but his apples are close at hand. Chapter 8: Acids and Bases 34

Acid-Base Equilibrium • How do we determine equilibrium position? Example: CH 3 COOH + NH 3 ⇌ CH 3 COO- + NH 4+ 1. Identify the acids (one on each side) 2. Which acid is stronger: (See table) 3. Identify the stronger base (the stronger acid gives the weaker conjugate base) 4. Strongest acid and base react to give weaker acid and base Strength of acids 35
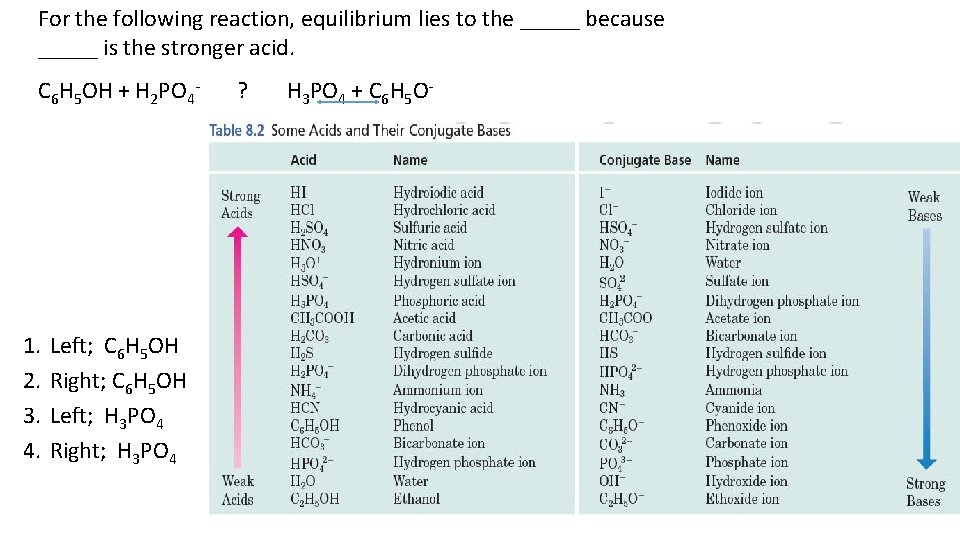
For the following reaction, equilibrium lies to the _____ because _____ is the stronger acid. C 6 H 5 OH + H 2 PO 4 - ? H 3 PO 4 + C 6 H 5 O- 1. 2. 3. 4. Left; C 6 H 5 OH Right; C 6 H 5 OH Left; H 3 PO 4 Right; H 3 PO 4
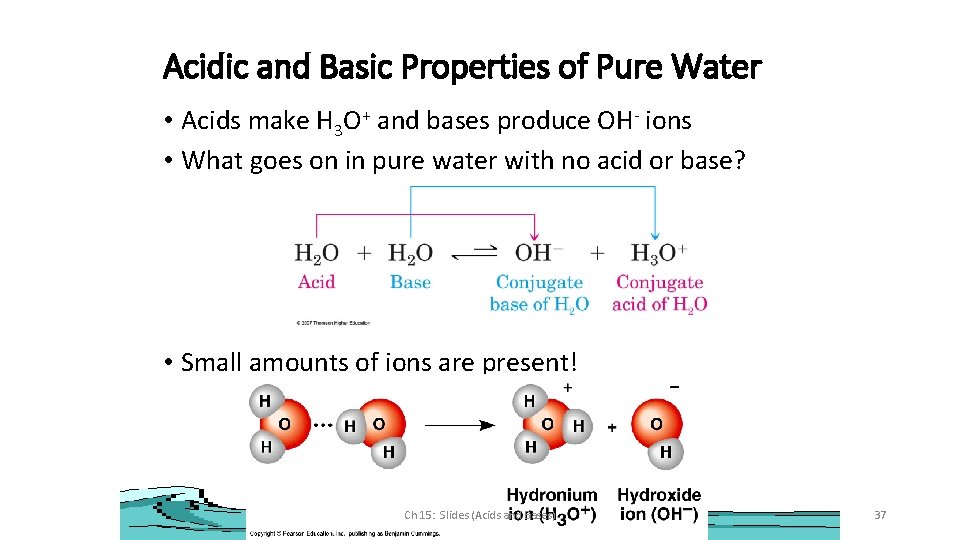
Acidic and Basic Properties of Pure Water • Acids make H 3 O+ and bases produce OH- ions • What goes on in pure water with no acid or base? • Small amounts of ions are present! Ch 15: Slides (Acids and Bases) 37

Amphoteric Water H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH-(aq) Soooo. . . K = Auto-ionization of water [H 3 O+] [OH-] [H 2 O] Kw = [H 3 O+] [OH-] Kw, the ion product of water Ch 15: Slides (Acids and Bases) 38

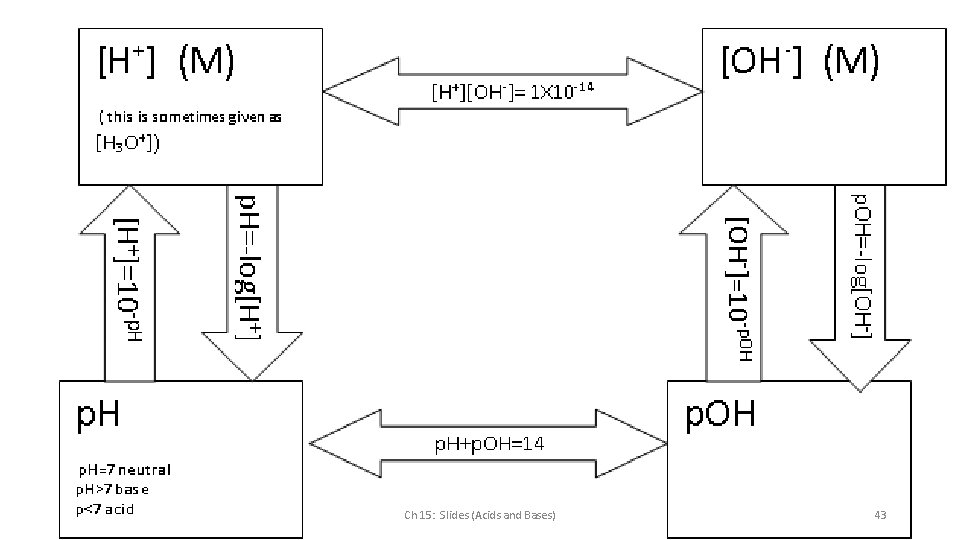
Meaning of “p” in p. H and p. OH Ch 15: Slides (Acids and Bases) 39

SIG FIG RULE of Logs: # of digits in the mantissa = # of sig figs in the original number log(3. 000 x 104) =4. 4771 log(3 x 104) = 4. 5 antilog(0. 301) = 2. 00 antilog(0. 30) = 2. 0 Ch 15: Slides (Acids and Bases) 40

Prove the relationships and/or practice (p. H/p. OH worksheet) Ch 15: Slides (Acids and Bases) 41
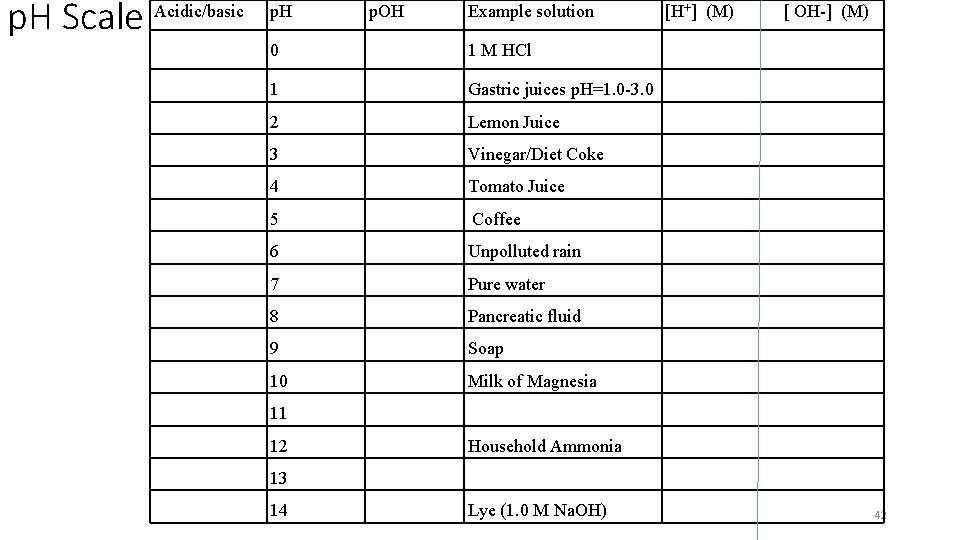
p. H Scale Acidic/basic p. H p. OH Example solution 0 1 M HCl 1 Gastric juices p. H=1. 0 -3. 0 2 Lemon Juice 3 Vinegar/Diet Coke 4 Tomato Juice 5 Coffee 6 Unpolluted rain 7 Pure water 8 Pancreatic fluid 9 Soap 10 Milk of Magnesia [H+] (M) [ OH-] (M) 11 12 Household Ammonia 13 14 Lye (1. 0 M Na. OH) 42
Ch 15: Slides (Acids and Bases) 43
![[H+] [OH-] p. H p. OH Acid/Base/or Neutral 1. 0 E-6 M 1. 0 [H+] [OH-] p. H p. OH Acid/Base/or Neutral 1. 0 E-6 M 1. 0](http://slidetodoc.com/presentation_image_h/b55439e1c3f1b32347871fff0b289e3f/image-44.jpg)
[H+] [OH-] p. H p. OH Acid/Base/or Neutral 1. 0 E-6 M 1. 0 E-8 M 9. 00 7. 00 2. 5 E-9 M 3. 3 E-5 M 3. 25 Ch 15: Slides (Acids and Bases) 9. 6 44
![[H+] [OH-] p. H p. OH Acid/Base/or Neutral 3. 8 E-6 M 9. 4 [H+] [OH-] p. H p. OH Acid/Base/or Neutral 3. 8 E-6 M 9. 4](http://slidetodoc.com/presentation_image_h/b55439e1c3f1b32347871fff0b289e3f/image-45.jpg)
[H+] [OH-] p. H p. OH Acid/Base/or Neutral 3. 8 E-6 M 9. 4 E-8 M 9. 55 6. 53 6. 8 E-9 M 1. 6 E-5 M 2. 25 Ch 15: Slides (Acids and Bases) 45

p. H strategy problems 1. You want to make up 3. 00 L of aqueous hydrochloric acid, HCl(aq), that has a p. H of 2. 00. How many grams of concentrated hydrochloric acid will you need? Concentrated hydrochloric acid contains 37. 2 % HCl (wt/wt). This problem is an application of the following concepts: p. H scale Degree of ionization of a strong acid Concentration terms, definitions and conversion between different terms (strict unit analysis). Dilution Ch 15: Slides (Acids and Bases) 46
- Slides: 46