Acids and Bases Arrhenius BrnstedLowry Definitions The Arrhenius

Acids and Bases Arrhenius & Brønsted-Lowry Definitions
![The Arrhenius Definition of Acids Increase [H+] above normal when dissolved in H 2 The Arrhenius Definition of Acids Increase [H+] above normal when dissolved in H 2](http://slidetodoc.com/presentation_image_h2/98fb84400130a4b9518b51de20cc1752/image-2.jpg)
The Arrhenius Definition of Acids Increase [H+] above normal when dissolved in H 2 O(L) Examples: Hydrochloric Acid (HCl) Acetic Acid (CH 3 COOH)
![The Arrhenius Definition of Bases Increase [OH-] above normal when dissolved in H 2 The Arrhenius Definition of Bases Increase [OH-] above normal when dissolved in H 2](http://slidetodoc.com/presentation_image_h2/98fb84400130a4b9518b51de20cc1752/image-3.jpg)
The Arrhenius Definition of Bases Increase [OH-] above normal when dissolved in H 2 O(L) Examples: Sodium Chloride (Na. OH) Ammonia (NH 3)

The Brønsted-Lowry Definition + Acids Substances that donate protons Bases Substances that accept protons

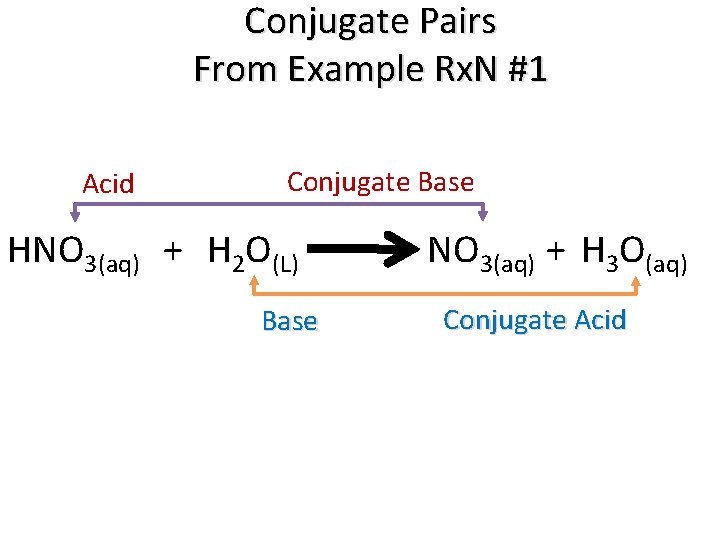
Example Rx. N #1 Dissociation of an Acid HNO 3(aq) + H 2 O(L) + NO 3(aq) + H 3 O(aq)
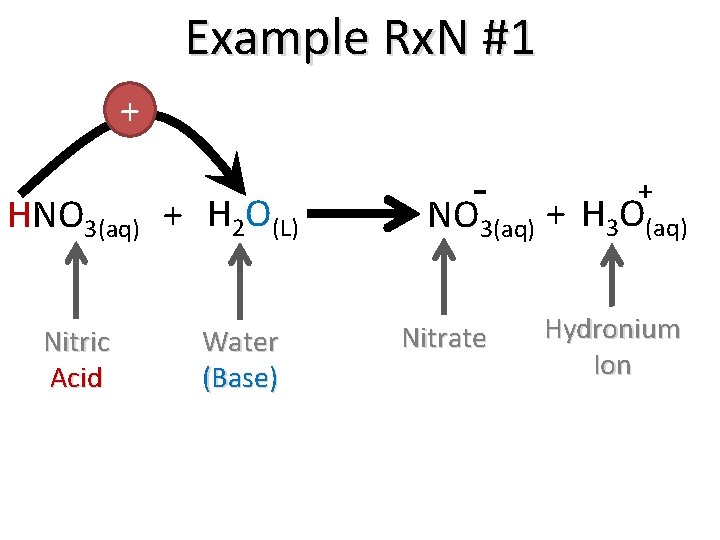
Example Rx. N #1 + HNO 3(aq) + H 2 O(L) Nitric Acid Water (Base) + NO 3(aq) + H 3 O(aq) Nitrate Hydronium Ion
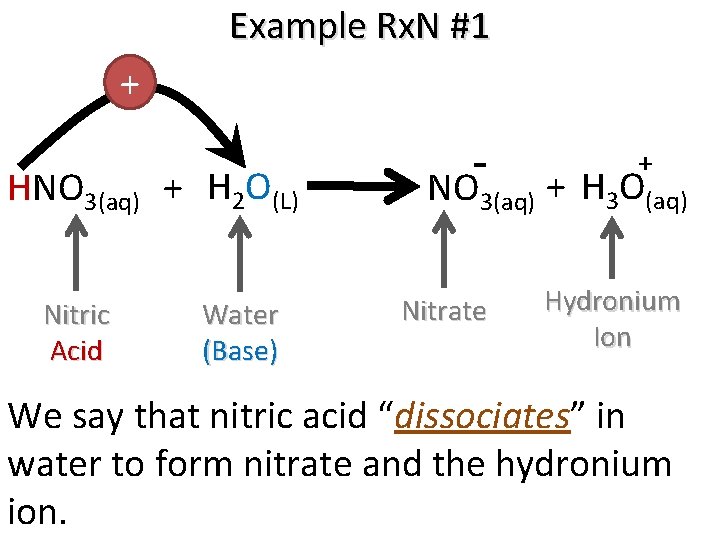
Example Rx. N #1 + HNO 3(aq) + H 2 O(L) Nitric Acid Water (Base) + NO 3(aq) + H 3 O(aq) Nitrate Hydronium Ion We say that nitric acid “dissociates” in water to form nitrate and the hydronium ion.
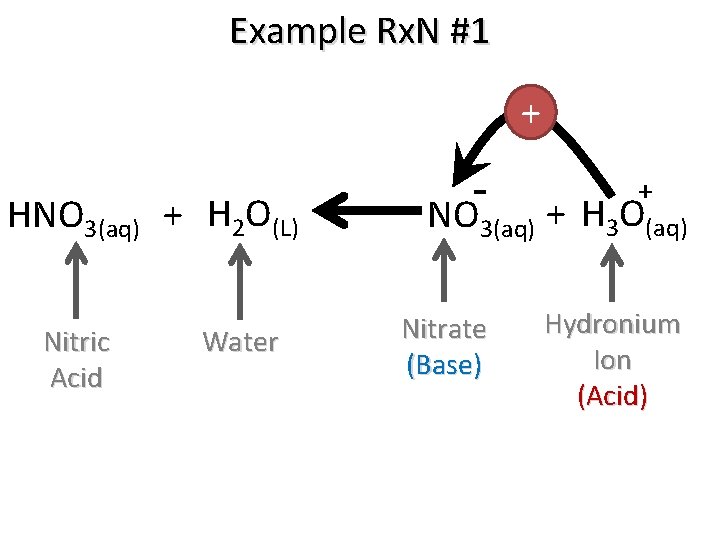
Example Rx. N #1 + HNO 3(aq) + H 2 O(L) Nitric Acid Water + NO 3(aq) + H 3 O(aq) Nitrate (Base) Hydronium Ion (Acid)
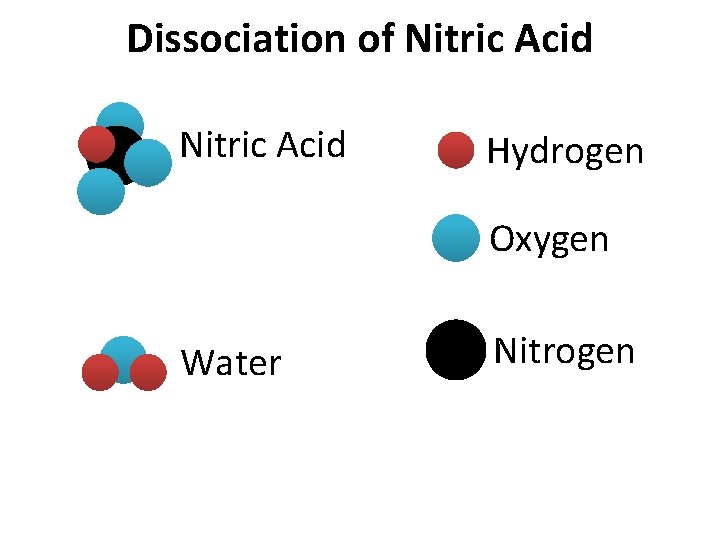
Dissociation of Nitric Acid Hydrogen Oxygen Water Nitrogen
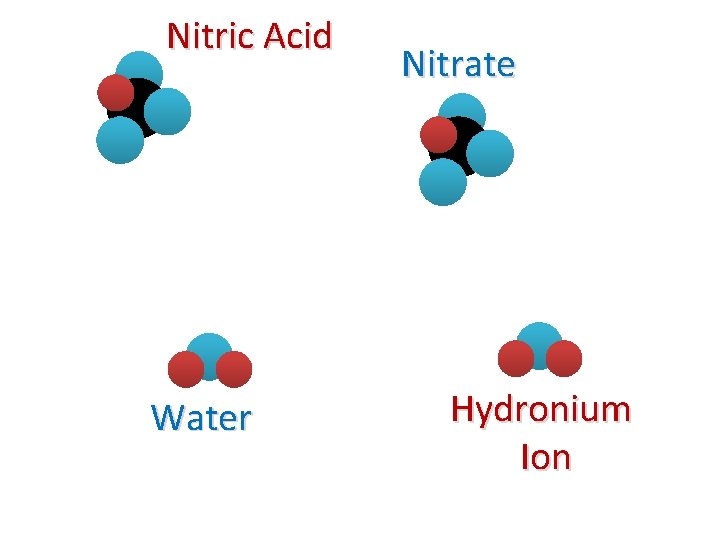
Nitric Acid Water Nitrate Hydronium Ion
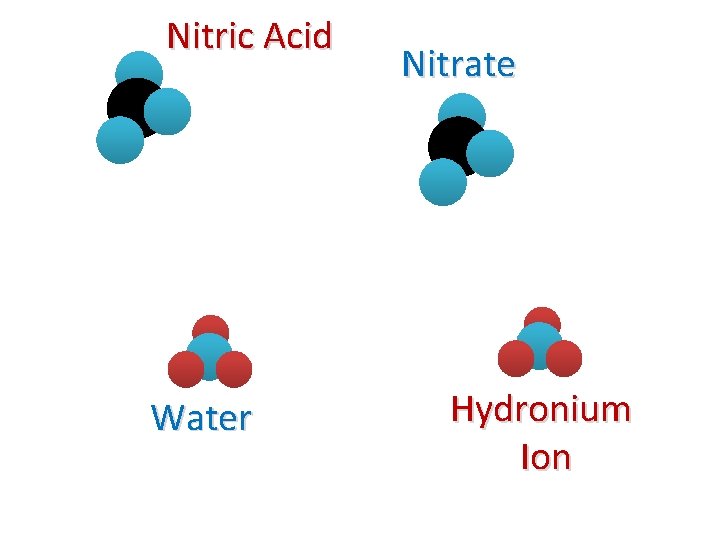
Nitric Acid Water Nitrate Hydronium Ion
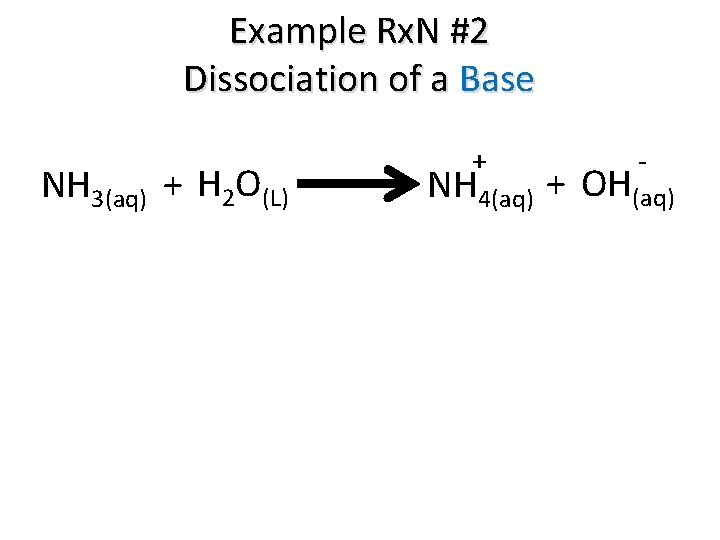
Example Rx. N #2 Dissociation of a Base NH 3(aq) + H 2 O(L) + - NH 4(aq) + OH(aq)
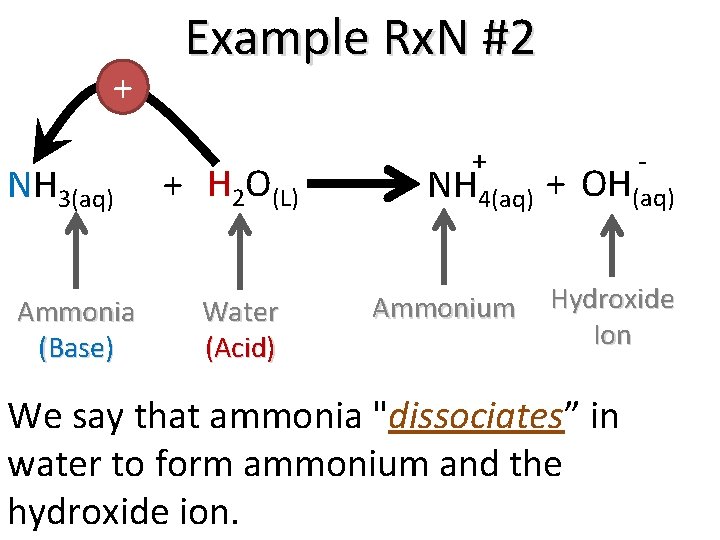
+ NH 3(aq) Ammonia (Base) Example Rx. N #2 + H 2 O(L) Water (Acid) + - NH 4(aq) + OH(aq) Ammonium Hydroxide Ion We say that ammonia "dissociates” in water to form ammonium and the hydroxide ion.
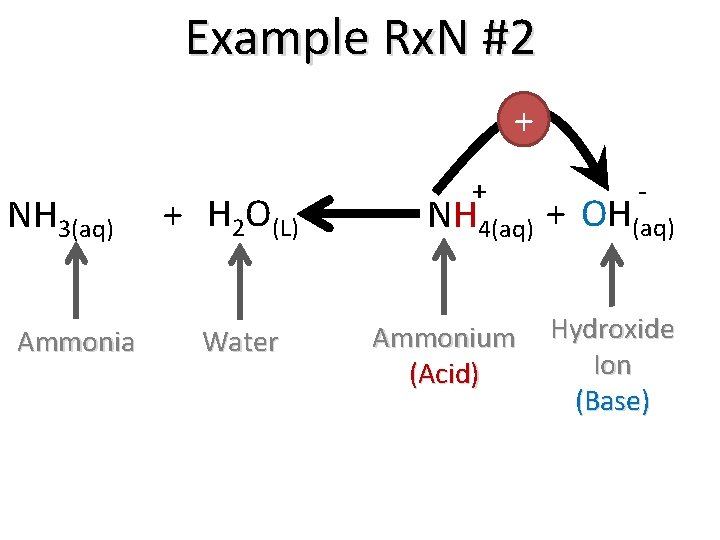
Example Rx. N #2 + NH 3(aq) Ammonia + H 2 O(L) Water + - NH 4(aq) + OH(aq) Ammonium (Acid) Hydroxide Ion (Base)
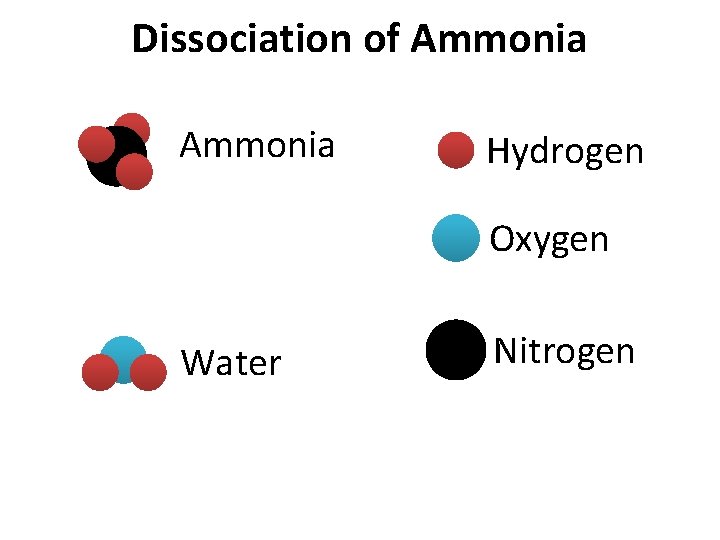
Dissociation of Ammonia Hydrogen Oxygen Water Nitrogen

Ammonia Water Ammonium Hydroxide Ion
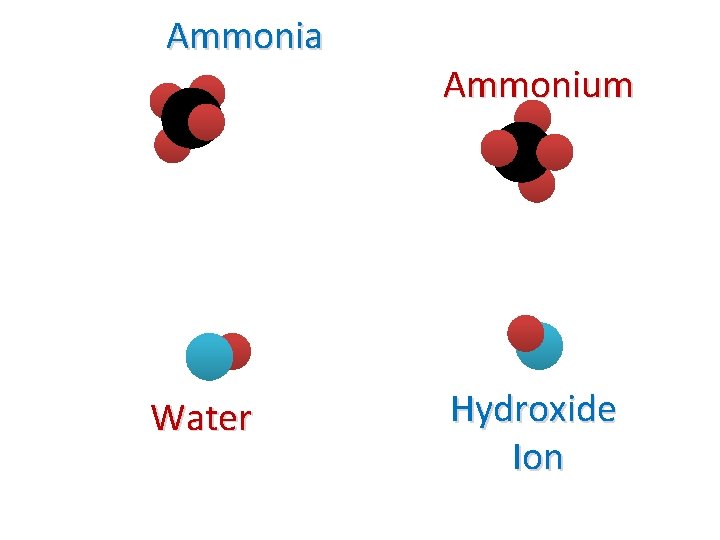
Ammonia Water Ammonium Hydroxide Ion
Conjugate Pairs From Example Rx. N #1 Acid Conjugate Base HNO 3(aq) + H 2 O(L) Base NO 3(aq) + H 3 O(aq) Conjugate Acid
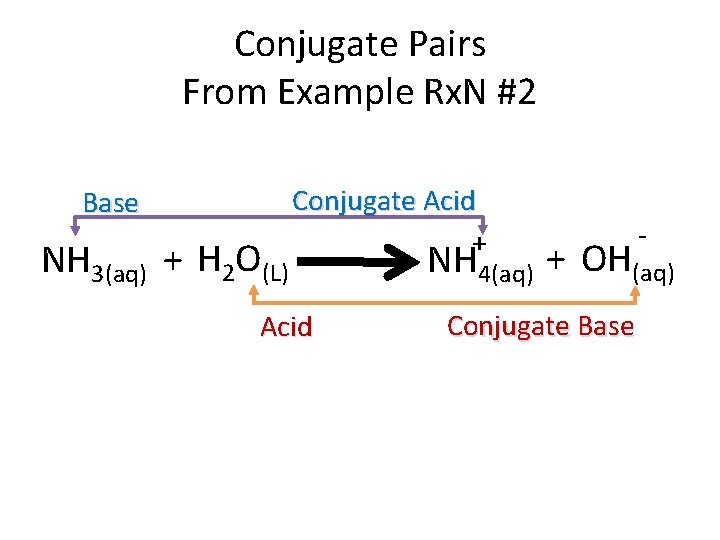
Conjugate Pairs From Example Rx. N #2 Base NH 3(aq) + H 2 O(L) Conjugate Acid + Acid - NH 4(aq) + OH(aq) Conjugate Base

The Amphoterism of Water Recall that in Rx. N #1, water behaved as a base HNO 3(aq) + H 2 O(L) + NO 3(aq) + H 3 O(aq) …but in Rx. N #2, it behaved as an acid NH 3(aq) + H 2 O(L) + NH 4(aq) + OH(aq)

The Amphoterism of Water When a substance is able to act as an acid or a base, it is called “amphoteric. ” Pronounced: “Am-fo-tear-ick”
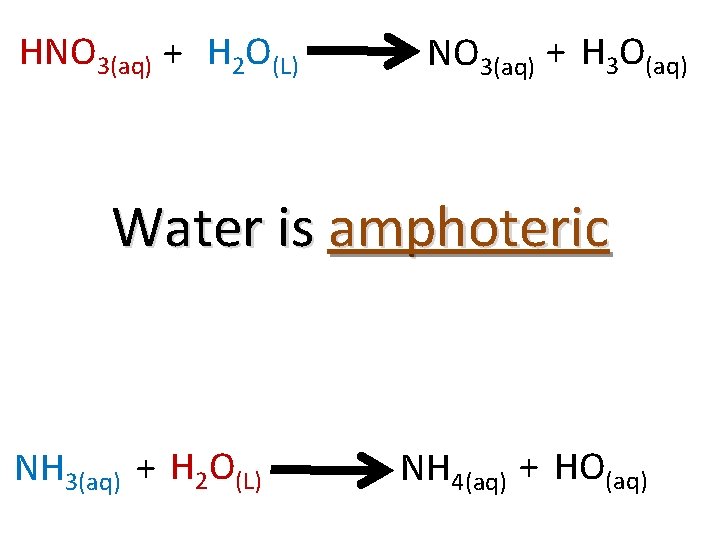
HNO 3(aq) + H 2 O(L) NO 3(aq) + H 3 O(aq) Water is amphoteric NH 3(aq) + H 2 O(L) NH 4(aq) + HO(aq)

- Slides: 23