Acids and Bases Arrhenius Arrhenius In aqueous solution

Acids and Bases
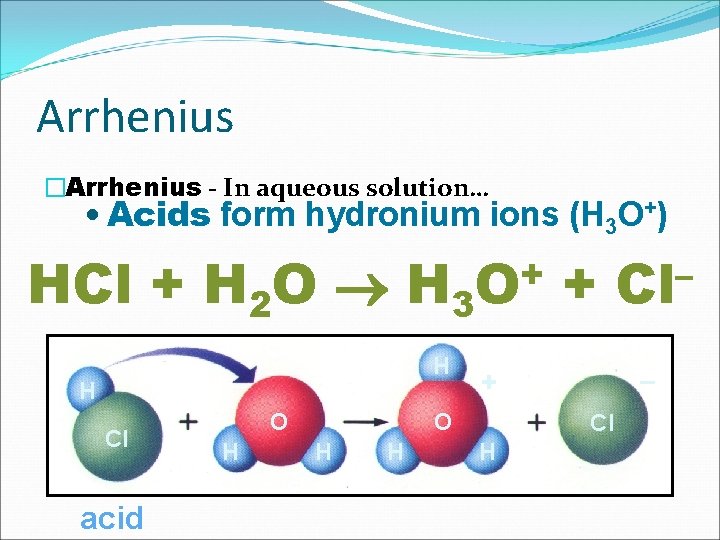
Arrhenius �Arrhenius - In aqueous solution… • Acids form hydronium ions (H 3 O+) HCl + H 2 O H 3 H H Cl acid O H + O H – + O H + Cl H – Cl
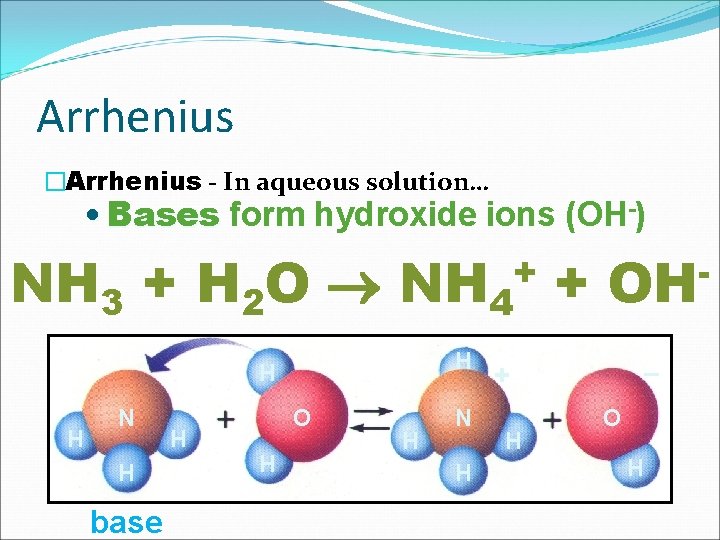
Arrhenius �Arrhenius - In aqueous solution… • Bases form hydroxide ions (OH-) NH 3 + H 2 O NH 4 + + H H H N H base H O H H N H OH – + H O H
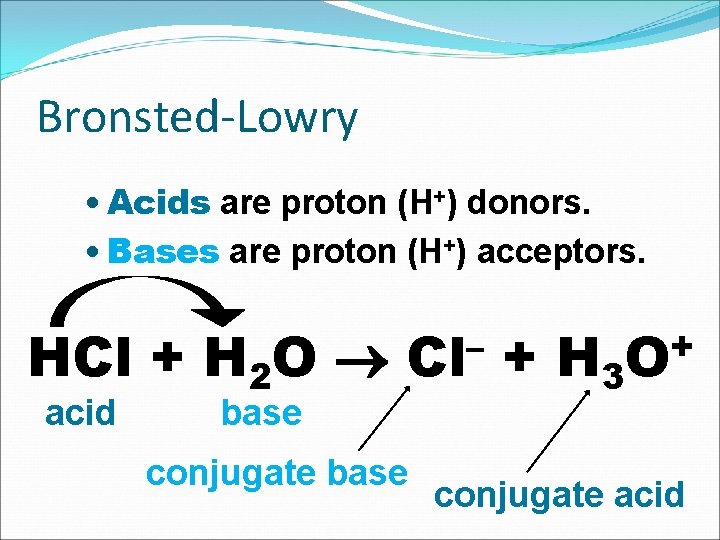
Bronsted-Lowry • Acids are proton (H+) donors. • Bases are proton (H+) acceptors. HCl + H 2 O acid – Cl base conjugate base + H 3 + O conjugate acid
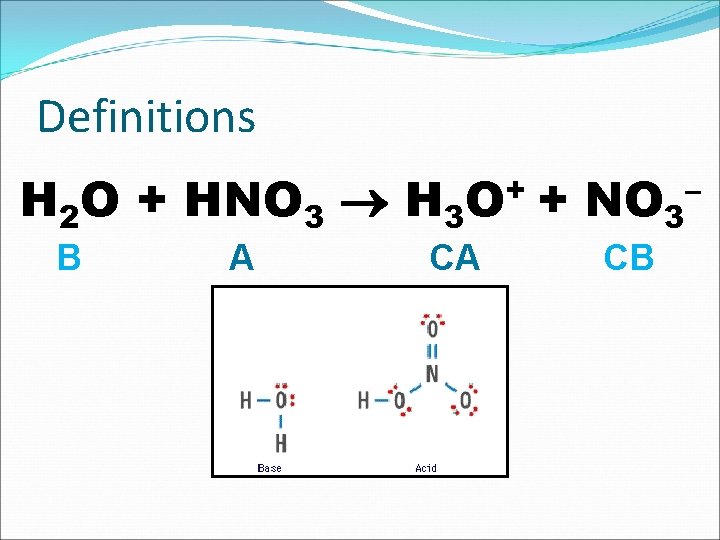
Definitions H 2 O + HNO 3 H 3 O+ + NO 3– B A CA CB
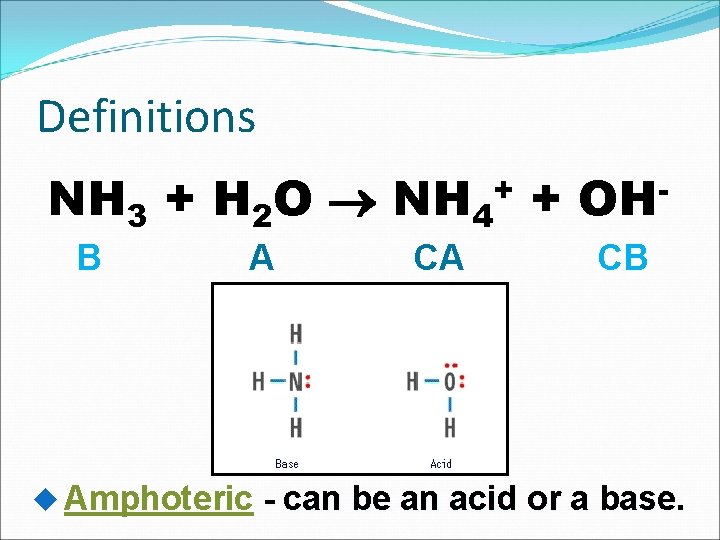
Definitions NH 3 + H 2 O NH 4+ + OHB A Amphoteric CA CB - can be an acid or a base.
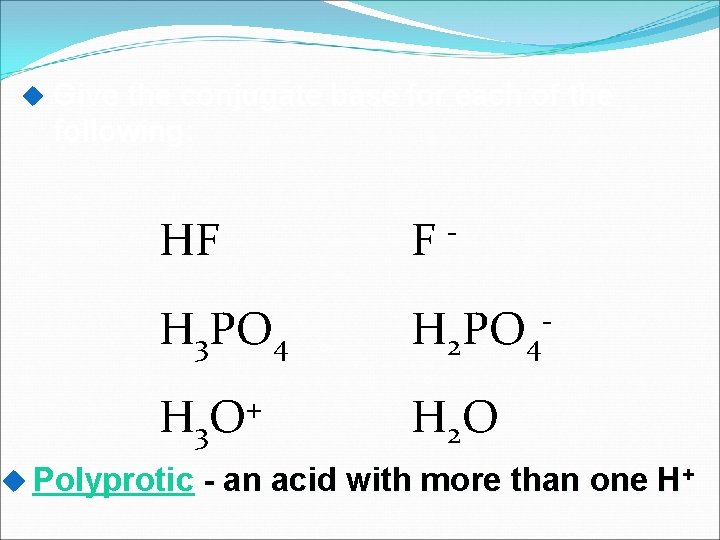
Give the conjugate base for each of the following: HF F- H 3 PO 4 H 2 PO 4 - H 3 O + H 2 O Polyprotic - an acid with more than one H+
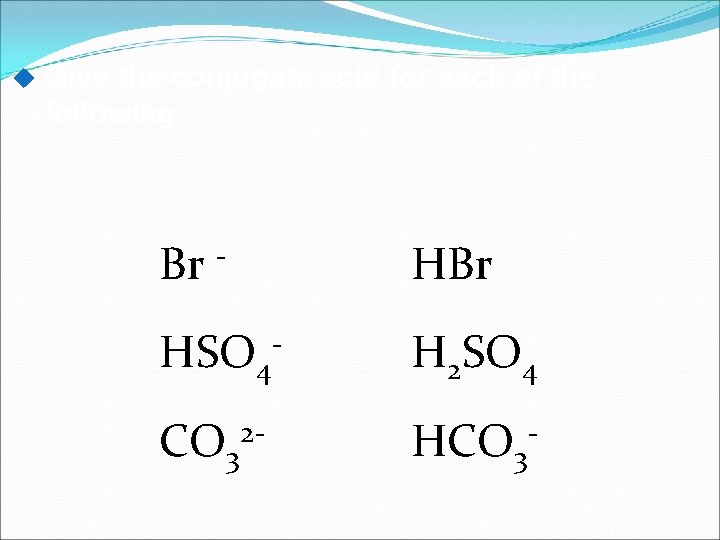
Give the conjugate acid for each of the following: Br - HBr HSO 4 - H 2 SO 4 CO 32 - HCO 3 -
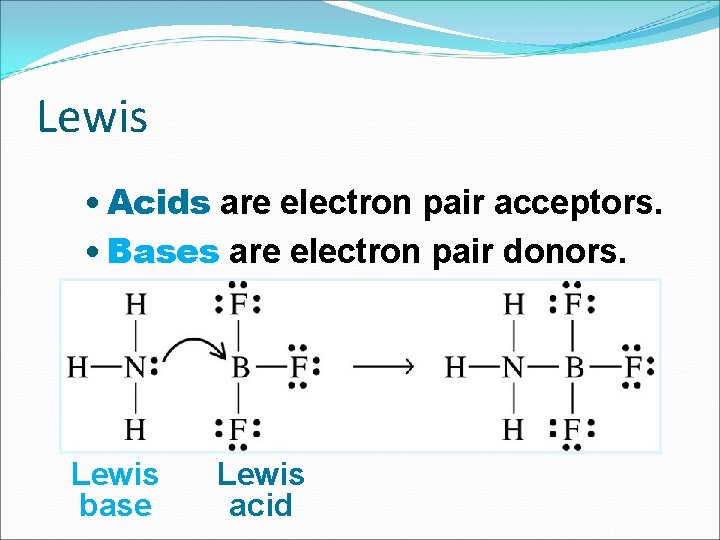
Lewis • Acids are electron pair acceptors. • Bases are electron pair donors. Lewis base Lewis acid

Properties electrolytes sour taste bitter taste turn litmus red turn litmus blue react with metals to form H 2 gas slippery feel vinegar, milk, soda, apples, citrus fruits ammonia, lye, antacid, baking soda Chem. ASAP

Properties of Acids q Acids taste sour q Acids effect indicators q Blue litmus turns red q Phenolphthalein turns clear/colorless q Acids have a p. H lower than 7 q Acids are proton (hydrogen ion, H+) donors q Acids react with active metals, produce H 2 q Acids react with carbonates q Acids neutralize bases

Acids to know: Strong Acids Weak Acids Sulfuric acid, H 2 SO 4 Phosphoric acid, H 3 PO 4 Hydrochloric acid, HCl Acetic acid, HC 2 H 3 O 2 Nitric acid, HNO 3

Nitric Acid �Used in the production of fertilizers �Used in the production of explosives �Nitric acid is a volatile acid – its reactive components evaporate easily �Stains proteins (including skin!)

Acetic Acid v. Used in the manufacture of plastics v. Used in making pharmaceuticals v. Acetic acid is the acid present in vinegar

Acids are Proton Donors Monoprotic acids Diprotic acids HCl H 2 SO 4 HC 2 H 3 O 2 H 2 CO 3 HNO 3 Triprotic acids H 3 PO 4
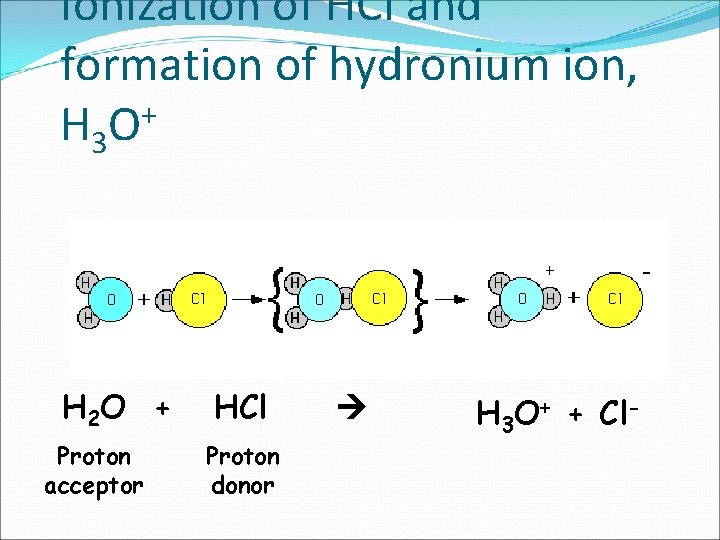
Ionization of HCl and formation of hydronium ion, + H 3 O H 2 O + Proton acceptor HCl Proton donor H 3 O+ + Cl-

Strong vs Weak & Concentrated vs Dilute �From last unit �Concentrated: A lot of solute has been dissolved �Dilute: A small amount of solute has been dissolved �THESE HAVE TO DO WITH AMOUNT DISSOLVED �From this unit �Strong: substance breaks up completely into ions �Dilute: substance does not break up completey into ions �THIS HAS NOTHING TO DO WITH HOW MUCH! � If you had only 1 molecule, but it was broken into ions, it would be strong!
STRONG ACID WEAK ACID STRONG BASE WEAK BASE

Strong Acids vs. Weak Acids Strong acids are assumed to be 100% ionized in solution (good proton donors). HCl H 2 SO 4 HNO 3 Weak acids are usually less than 5% ionized in solution (poor proton donors). H 3 PO 4 HC 2 H 3 O 2 Organic acids

Strong Acid Dissociation
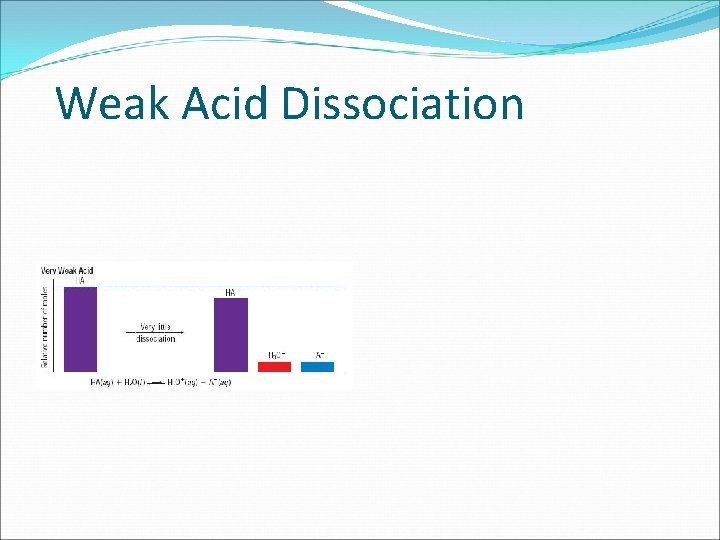
Weak Acid Dissociation

Concep. Test Which of the following "molecular" pictures best represents a concentrated solution of the weak acid HA? A B

Acids Effect Indicators Blue litmus paper turns red in contact with an acid. Phenolphthalein turns clear/colorless in an acid.

Acids have a p. H less than 7
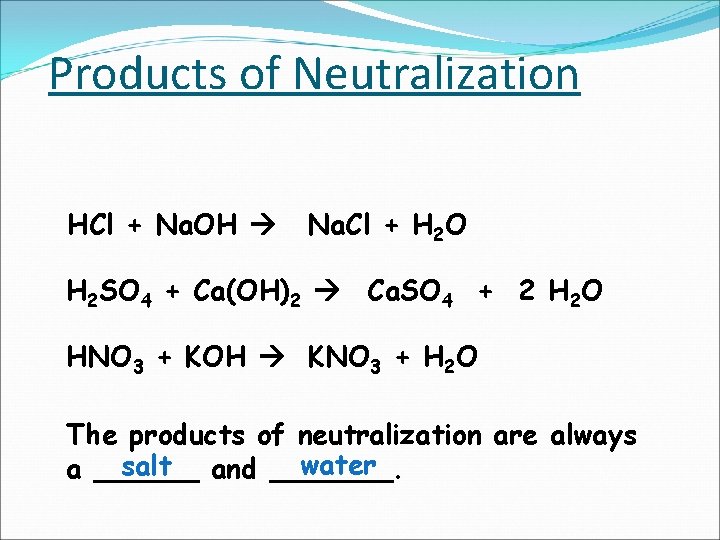
Products of Neutralization HCl + Na. OH Na. Cl + H 2 O H 2 SO 4 + Ca(OH)2 Ca. SO 4 + 2 H 2 O HNO 3 + KOH KNO 3 + H 2 O The products of neutralization are always water salt and _______. a ______

Properties of Bases q Bases taste bitter q Bases effect indicators q Red litmus turns blue q Phenolphthalein turns pinkish q Bases have a p. H greater than 7 q Bases are proton (hydrogen ion, H+) acceptors q Solutions of bases feel slippery q Bases neutralize acids

Examples of Bases Ø Sodium hydroxide (lye), Na. OH Ø Potassium hydroxide, KOH Ø Magnesium hydroxide, Mg(OH)2 Ø Calcium hydroxide (lime), Ca(OH)2

Bases Effect Indicators Red litmus paper turns blue in contact with a base. Phenolphthalein turns purple/pink in a base.

Bases have a p. H greater than 7

Bases Neutralize Acids Milk of Magnesia contains magnesium hydroxide, Mg(OH)2, which neutralizes stomach acid, HCl. 2 HCl + Mg(OH)2 Mg. Cl 2 + 2 H 2 O
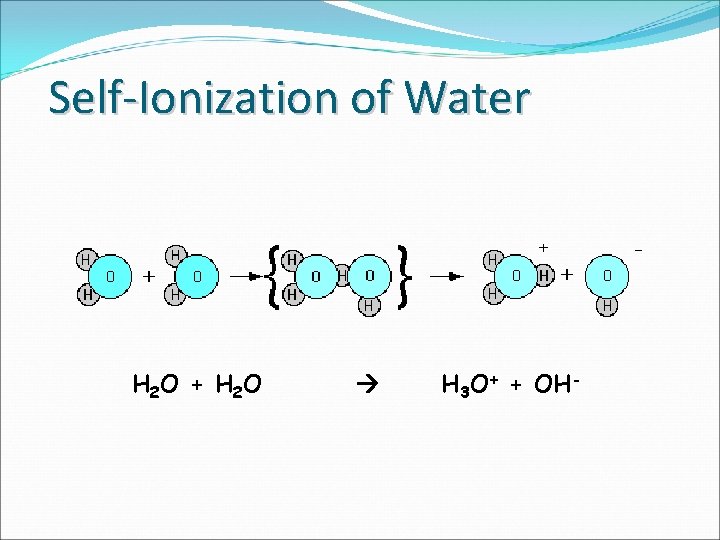
Self-Ionization of Water H 2 O + H 2 O H 3 O+ + OH-
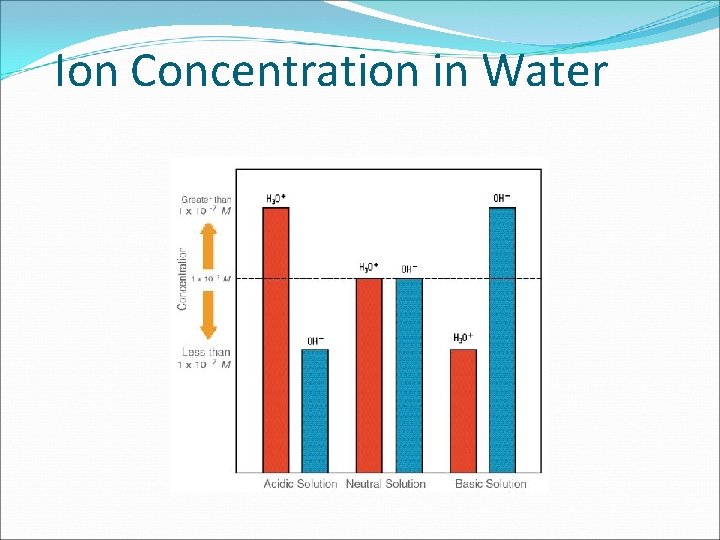
Ion Concentration in Water

Kw – Ionization Constant for Water In pure water at 25 C: [H 3 O+] = 1 x 10 -7 mol/L (M) [OH-] = 1 x 10 -7 mol/L (M) Kw is a constant at 25 C: Kw = [H 3 O+][OH-] Kw = (1 x 10 -7) = 1 x 10 -14 M 2
![Concentrations of H 3 O+ and OHSince [H 3 O+] = [OH-] in pure Concentrations of H 3 O+ and OHSince [H 3 O+] = [OH-] in pure](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-34.jpg)
Concentrations of H 3 O+ and OHSince [H 3 O+] = [OH-] in pure water, it is neutral. When [H 3 O+] [OH-] , the solution is acidic. When [H 3 O+] [OH-], the solution is basic.
![Concentrations of H 3 O+ and OHKw = [H 3 O+] [OH- ] = Concentrations of H 3 O+ and OHKw = [H 3 O+] [OH- ] =](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-35.jpg)
Concentrations of H 3 O+ and OHKw = [H 3 O+] [OH- ] = 1. 0 x 10 -14 M 2 [H 3 O+] = Kw = 1. 0 X 10 -14 M 2 [OH-] [OH -] = Kw. = [H 3 O+] 1. 0 X 10 -14 M 2 [H 3 O+ ] Assume that strong acids and bases are completely ionized in solution
![Examples of Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10 Examples of Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-36.jpg)
Examples of Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10 -14 M 2 = [H 3 O+] [OH-] EX. If you have 2. 0 x 10 -4 M H 3 O+, then Kw = 1. 0 x 10 -14 M 2 = (2. 0 x 10 -4 ) x [OH-] = Kw = 1. 0 x 10 -14 M 2 = 0. 5 x 10 -10 M OH[H 3 O+] 2. 0 x 10 -4 M
![Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10 -14 M Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10 -14 M](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-37.jpg)
Calculating [H 3 O+] and [OH-] Kw = 1. 0 x 10 -14 M 2 = [H 3 O+] [OH-] From the previous slide: [OH-] = 0. 5 x 10 -10 M [H 3 O+] = 2. 0 x 10 -4 M [H 3 O+] [OH-] , the solution is acidic.

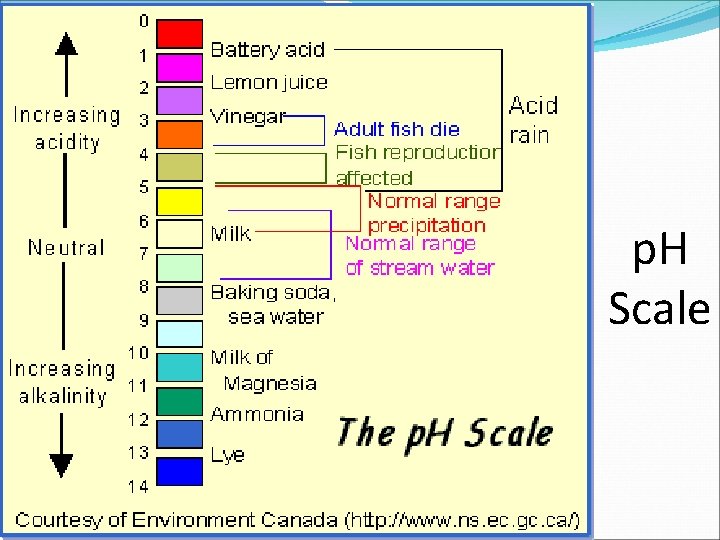
p. H Scale �Expressing the acidity or alkalinity in terms of concentration (how much— concentrated or dilute) of ions can be cumbersome since the values tend to be small. � A more convenient way to express concentration is to use p. H: p. H “hydrogen power” (French “pouvoir hydrogene”)

p. H Scale �p. H � a measure of the concentration of H 3 O+ ions in solution � measured with a p. H paper, p. H meter or an indicator with a wide color range � Ranges from 0 -14 � Each number is 10 x more concentrated than the next. (2 has 10 x more H 3 O+ ions than 3) INCREASING ACIDITY NEUTRAL INCREASING BASICITY

p. H Scale � The p. H of a solution is defined as the negative of the common logarithm of the hydronium ion concentration. [H 3 O+] p. H is expressed by the following: p. H = - log [H 3 O+] The common logarithm of a number is the power to which 10 must be raised to equal the number.
![p. H Scale � When the concentration of [H 3 O+] in solution is p. H Scale � When the concentration of [H 3 O+] in solution is](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-41.jpg)
p. H Scale � When the concentration of [H 3 O+] in solution is an integral power of 10, such as 1 M or 0. 01 M = 1. 0 x 10 -2, the p. H of the solution is the exponent of the hydronium ion’s concentration with the sign changed. EX. [H 3 O+] = 1. 0 x 10 -5 M p. H = 5
![p. H Scale �Example: A neutral solution has a [H 3 O+] of 1. p. H Scale �Example: A neutral solution has a [H 3 O+] of 1.](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-42.jpg)
p. H Scale �Example: A neutral solution has a [H 3 O+] of 1. 0 x 10 -7 The p. H is determined as follows: p. H = - log [H 3 O+] = -log (1. 0 x 10 -7) = -(-7) = 7

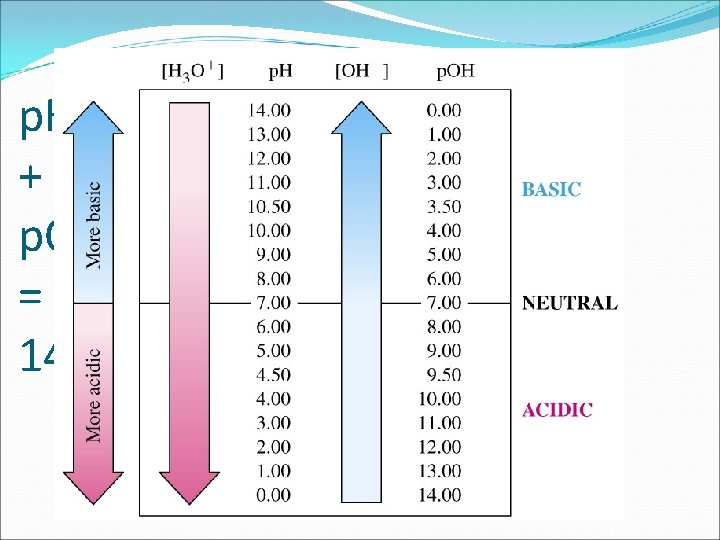
p. H Scale � p. OH can also be determined, however it can only be determined from [OH-] concentration p. OH = - log [OH-] A neutral solution would have [OH-] = 1. 0 x 10 -7 M. Therefore, the p. OH = 7 p. H + p. OH = 14. 0

p. H and p. OH Calculations

p. H SCALE OF COMMON HOUSEHOLD PRODUCTS

Sample Problem: �What is the p. H of a 1. 0 x 10 -3 M Na. OH solution? � Since [Na. OH] is 1 x 10 -3 M, and it’s a 1: 1 ratio of Na: OH, then [OH-]=1. 0 x 10 -3 �Using this we can solve for p. OH �Once we have p. OH, then p. H=14 -p. OH

Sample Problem What is the p. H of a solution if the [H 3 O+] is 3. 4 x 10 -5 M? Use the calculator to find: p. H = -log [H 3 O+] = -log (3. 4 x 10 -5 )
![+ O] Finding [H 3 p. H and p. OH �[H 3 O+] = + O] Finding [H 3 p. H and p. OH �[H 3 O+] =](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-48.jpg)
+ O] Finding [H 3 p. H and p. OH �[H 3 O+] = 10 (-p. H) �[OH-] = 10 (-p. OH) and [OH ] from
![Sample Problem What is the [OH-] of a solution whose p. H = 3. Sample Problem What is the [OH-] of a solution whose p. H = 3.](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-49.jpg)
Sample Problem What is the [OH-] of a solution whose p. H = 3. 00?
![Formulas! Finding [H 3 O+] and [OH-] Finding p. H and p. OH p. Formulas! Finding [H 3 O+] and [OH-] Finding p. H and p. OH p.](http://slidetodoc.com/presentation_image_h2/4e166d6686b84a33db3eece4f4a1933a/image-50.jpg)
Formulas! Finding [H 3 O+] and [OH-] Finding p. H and p. OH p. H = -log (H 3 O+) p. OH = -log (OH-) p. H + p. OH = 14 [H 3 O+] = Kw [OH-] [OH -] = Kw [H 3 O+] Finding [H 3 O+] and [OH-] from p. H, p. OH [H 3 O+] = 10 (-p. H) [OH-] = 10 (-p. OH)

H+, OH-, and p. H
p. H Scale
p. H + p. OH = 14

Measuring p. H with widerange paper

Narrow-Range p. H Paper
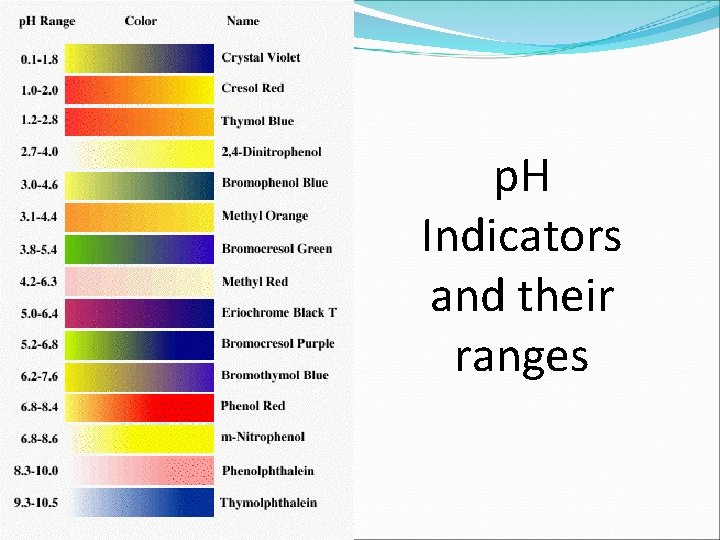
p. H Indicators and their ranges

Neutralization & Titrations ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic • Salts can be neutral, acidic, or basic. • Neutralization does not mean p. H = 7.

If you have equal amounts… �Strong Acid + Strong Base = Neutral (p. H=7) �Strong Acid + Weak Base = Acidic (p. H < 7) �Weak Acid + Strong Base = Basic (p. H>7)

Titration standard solution �Titration �Process in which a standard solution is used to determine the concentration of an unknown solution
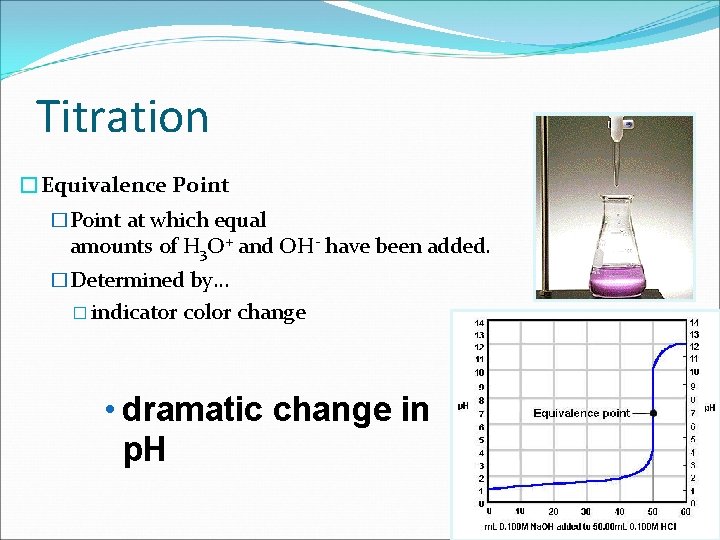
Titration �Equivalence Point �Point at which equal amounts of H 3 O+ and OH- have been added. �Determined by… � indicator color change • dramatic change in p. H
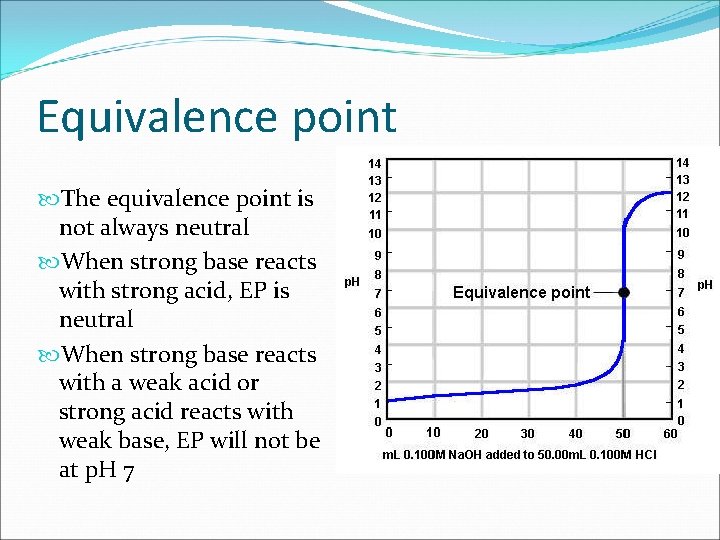
Equivalence point The equivalence point is not always neutral When strong base reacts with strong acid, EP is neutral When strong base reacts with a weak acid or strong acid reacts with weak base, EP will not be at p. H 7
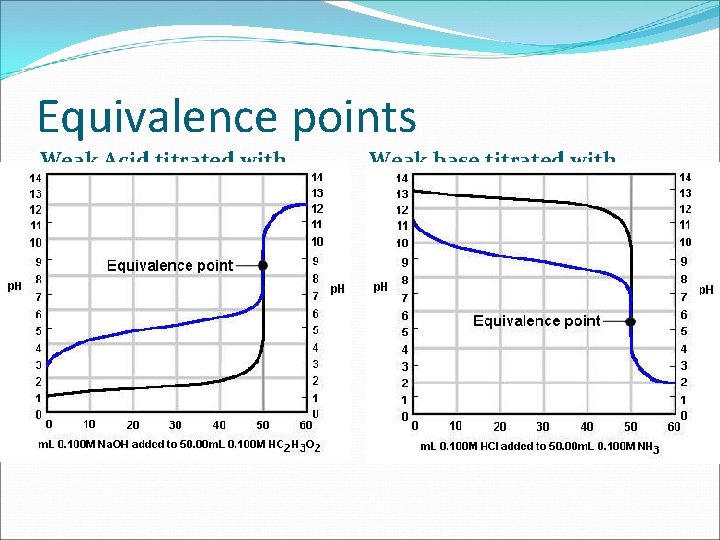
Equivalence points Weak Acid titrated with strong base Weak base titrated with strong acid


Titration + O moles H 3 = moles M V n = M V n OH M: Molarity V: volume n: # of H+ ions in the acid or OH- ions in the base

Titration � 42. 5 m. L of 1. 3 M KOH are required to neutralize 50. 0 m. L of H 2 SO 4. Find the molarity of H 2 SO 4. H 3 O + M=? V = 50. 0 m. L n=2 OHM = 1. 3 M V = 42. 5 m. L n=1 MV# = MV# M(50. 0 m. L)(2) =(1. 3 M)(42. 5 m. L)(1) M = 0. 55 M H 2 SO 4

Or you can solve by setting up factor label �m. L KOH moles KOH (using molarity 1. 3 moles / 1 liter) moles of H 2 SO 4 (from rxn stoich ratios) �Then divide moles H 2 SO 4 by volume H 2 SO 4 in L (change from m. L)
- Slides: 66