Acids and Bases Arrhenius Acids and Bases Neutralization

Acids and Bases Arrhenius Acids and Bases Neutralization Reactions and Acid – Base Titrations

Arrhenius Acids and Bases • Citrus Fruits like oranges and lemons contain acetic acid. • Cleaning products as glass cleaners contain bases • Different theories have been developed to explain what is an acid and what is a base • The earliest theory has been developed by Swedish Scientist Svanté Arrhenius in 1884. • His theory was part of his doctoral thesis and was hard to be accepted by the examiners, he was barely awarded his degree • 11 years later, in 1905, he received the Nobel Prize for this theory

Arrhenius Acids and Bases • Uses the concepts of ions in solution to explain the nature of Acids and Bases. • Acids are molecular compounds and form ions when dissolved in water. HCl (g) H⁺ (aq) + Cl⁻ (aq) • By forming ions, acids undergo an ionization • Ionization : the process of forming ions

Arrhenius Acids and Bases Acids • • According to Arrhenius, an acid must contain a hydrogen atom that can become a hydrogen ion. Acids are molecular compounds and form ions when dissolved in water: HCl (g) • • Bases • • H⁺ (aq) + Cl⁻ (aq) By forming ions, acids undergo an ionization Ionization : the process of forming ions • • According to Arrhenius, a base must contain a hydroxide group -OH that can become a hydroxide ion OH⁻. Bases are ionic compounds that break apart into ions when dissolved in water: Na. OH (s) Na⁺ (aq) + OH⁻ (aq) By breaking apart, a base undergo a dissociation. Dissociation : the process in which ions break apart when dissolved in solution.

Properties of Acids and Bases Properties Acids Bases Taste sour bitter Texture No characteristic texture Slippery to the touch p. H Less than 7 Greater than 7 Indicator Colors • Litmus • Red • phenolphthalein • colorless • Blue • pink Corrosion Corrode tissue but not metals Corrode tissue and metals Reactions • With metals • React with active metals to produce H 2 (gas) • With carbonates • React with carbonates to produce CO 2 (gas) Electrical Conductivity Conduct electricity • No reaction Conduct electricity Common examples Citrus fruits, vinegar, carbonated drinks, vitamin C Soap, baking soda, oven cleaner, household ammonia
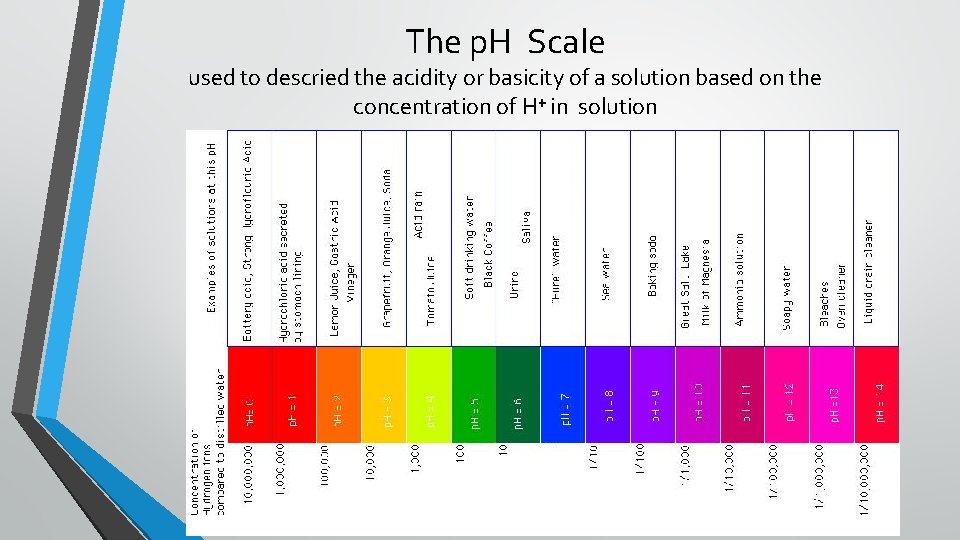
The p. H Scale used to descried the acidity or basicity of a solution based on the concentration of H⁺ in solution

Question 1: Identify each substance as an acid or a base a. HBr (aq) b. HCl. O 4 (aq) c. Sr(OH)2 (aq) d. KOH (aq) e. Ca(OH)2 (aq) f. Cs(OH) (aq) g. H 3 PO 4 (aq) h. HNO 3 (aq)
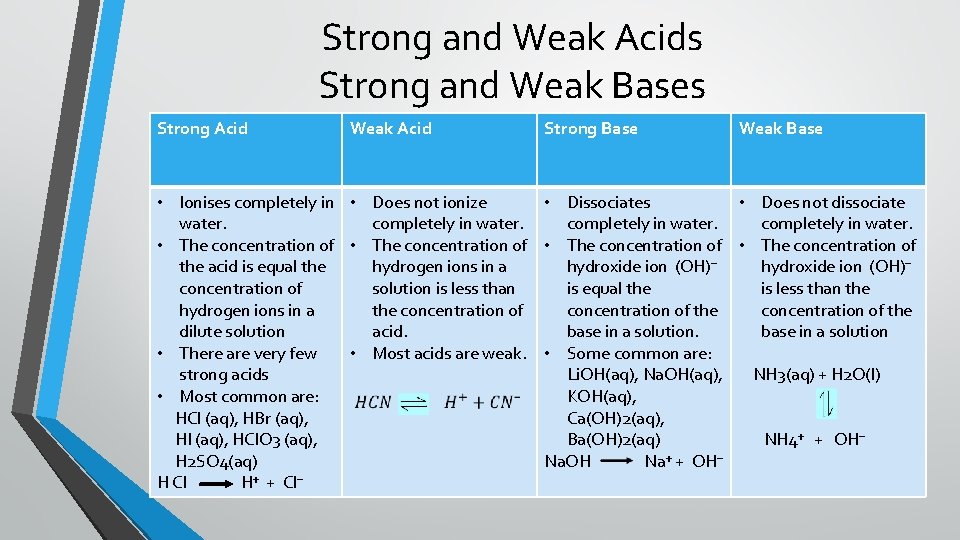
Strong and Weak Acids Strong and Weak Bases Strong Acid Weak Acid • Ionises completely in • Does not ionize water. completely in water. • The concentration of the acid is equal the hydrogen ions in a concentration of solution is less than hydrogen ions in a the concentration of dilute solution acid. • There are very few • Most acids are weak. strong acids • Most common are: HCl (aq), HBr (aq), HI (aq), HCl. O 3 (aq), H 2 SO 4(aq) H Cl H⁺ + Cl⁻ Strong Base Weak Base • Dissociates • Does not dissociate completely in water. • The concentration of hydroxide ion (OH)⁻ is equal the is less than the concentration of the base in a solution • Some common are: Li. OH(aq), Na. OH(aq), NH 3(aq) + H 2 O(l) KOH(aq), Ca(OH)2(aq), Ba(OH)2(aq) NH 4⁺ + OH⁻ Na. OH Na⁺ + OH⁻

Question 2: Consider 0. 1 mol/L solutions of HCl(aq), CH 3 COOH (aq) and NH 3(aq). List these solutions in order of increasing hydrogen ions concentration. Explain your reasoning.
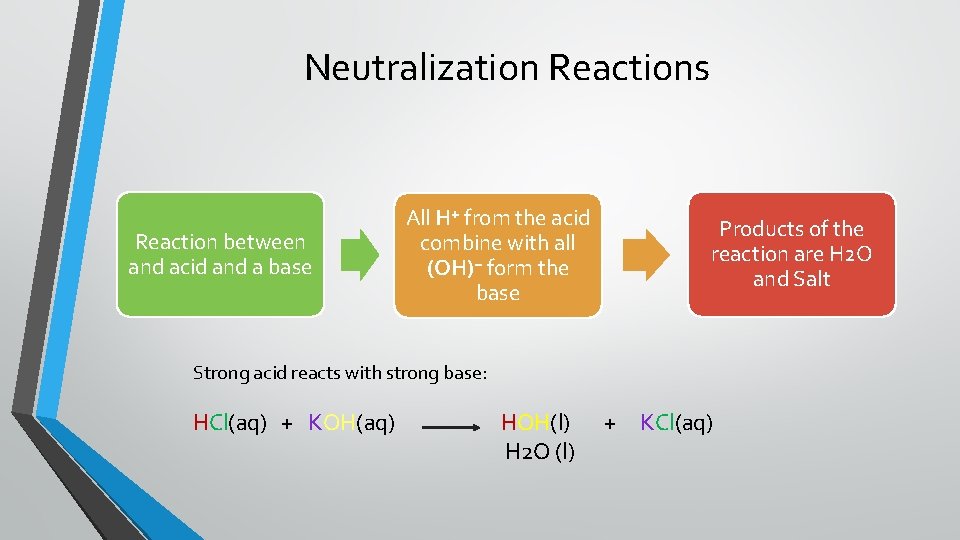
Neutralization Reactions Reaction between and acid and a base All H⁺ from the acid combine with all (OH)⁻ form the base Products of the reaction are H 2 O and Salt Strong acid reacts with strong base: HCl(aq) + KOH(aq) HOH(l) H 2 O (l) + KCl(aq)

Acid-Base Titration Performing a Neutralization Reaction Procedure called Titration Used to determine the concentration of a solution https: //www. youtube. com/watch? v=s. Fp. FCPTDv 2 w

Question 3: • A 25 m. L of nitric acid HNO 3(aq) is neutralized by 18. 55 m. L of a 0. 1750 mol/L sodium hydroxide, Na. OH(aq). What is the concentration of HNO 3(aq) solution?

Question 5: • What volume of 0. 996 mol/L barium hydroxide Ba(OH)2(aq), is needed to neutralize 25. 0 mol/L of 1. 70 mo/L nitric acid, HNO 3(aq)?
- Slides: 13