Acids and Bases Arrhenius Acids and Bases Arrhenius

Acids and Bases

Arrhenius Acids and Bases • Arrhenius acid: Break apart (dissociate) to yield hydrogen ions (H+) in water (aqueous) solutions HCl → H+ + Cl • Arrhenius base: Break apart (dissociate) to yield hydroxide ions (OH-) in water (aqueous) solutions Na. OH → Na+ + OH-

Naming Acids and Bases Acids: Remember this chart? ? ? Anion Ending Acid Name -ide hydro-(stem)-ic acid -ite (stem)-ous acid -ate (stem)-ic acid Bases: cation name + hydroxide
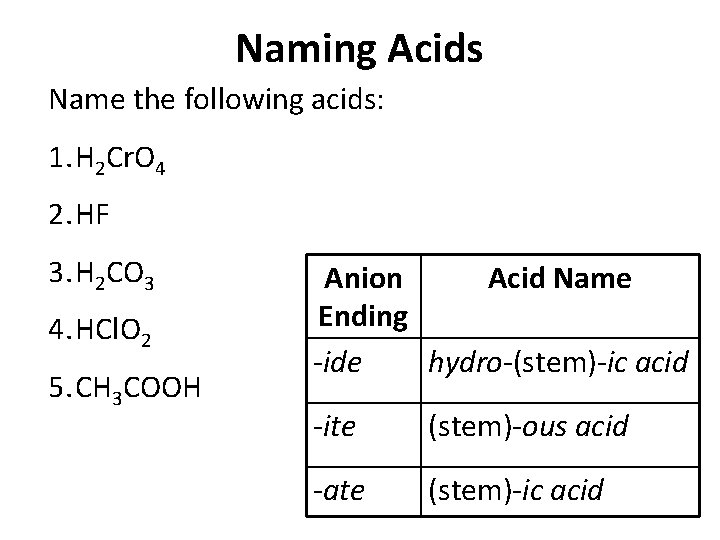
Naming Acids Name the following acids: 1. H 2 Cr. O 4 2. HF 3. H 2 CO 3 4. HCl. O 2 5. CH 3 COOH Anion Acid Name Ending -ide hydro-(stem)-ic acid -ite (stem)-ous acid -ate (stem)-ic acid

Naming Bases Name the following bases: 1. Ba(OH)2 2. KOH 3. Al(OH)3 Cation name + hydroxide

More Naming Practice Name the following acids/bases or write their formula: 1. H 3 PO 4 4. Hydrobromic acid Anion Acid Name Ending -ide hydro-(stem)-ic acid -ite (stem)-ous acid 5. Lithium hydroxide -ate 2. Nitrous acid 3. Mg(OH)2 6. H 2 S (stem)-ic acid

The p. H Scale
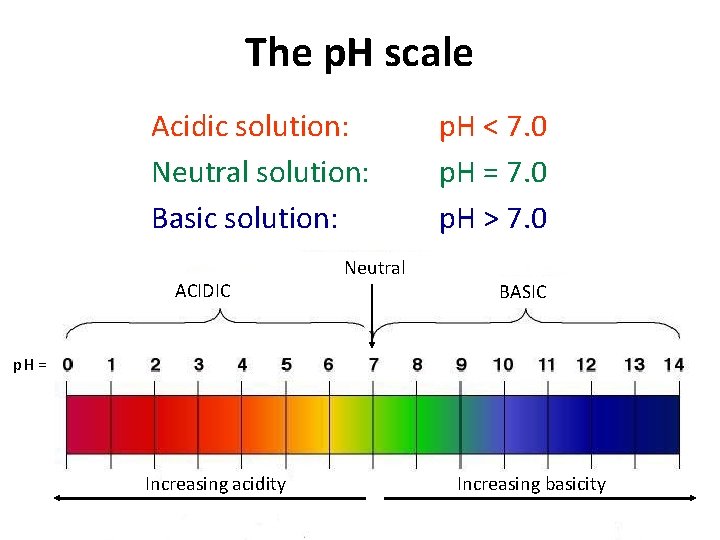
The p. H scale Acidic solution: Neutral solution: Basic solution: ACIDIC Neutral p. H < 7. 0 p. H = 7. 0 p. H > 7. 0 BASIC p. H = Increasing acidity Increasing basicity

p. H (“potential hydrogen”): a way of expressing the hydrogen ion concentration p. H = - log [H+] = the hydrogen ion concentration IN MOLARITY [H+] = 10 -p. H
![Calculating p. H Example p. H = - log [H+] What is the p. Calculating p. H Example p. H = - log [H+] What is the p.](http://slidetodoc.com/presentation_image_h2/3cf8f1f06834d07e77fcf0825b7088a5/image-10.jpg)
Calculating p. H Example p. H = - log [H+] What is the p. H of a HCl solution in which [H+] = 3. 9 x 10 -4 M?
![Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a](http://slidetodoc.com/presentation_image_h2/3cf8f1f06834d07e77fcf0825b7088a5/image-11.jpg)
Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a solution if the p. H = 10. 8?
![The p. H Scale - As p. H increases, [H+] decreases - As p. The p. H Scale - As p. H increases, [H+] decreases - As p.](http://slidetodoc.com/presentation_image_h2/3cf8f1f06834d07e77fcf0825b7088a5/image-12.jpg)
The p. H Scale - As p. H increases, [H+] decreases - As p. H decreases, [H+] increases
![Calculating p. H [H+] = 10 -p. H Acidic Solution p. H < 7. Calculating p. H [H+] = 10 -p. H Acidic Solution p. H < 7.](http://slidetodoc.com/presentation_image_h2/3cf8f1f06834d07e77fcf0825b7088a5/image-13.jpg)
Calculating p. H [H+] = 10 -p. H Acidic Solution p. H < 7. 0 [H+] > 1 x 10 -7 Neutral Solution p. H = 7. 0 [H+] = 1 x 10 -7 Basic Solution p. H > 7. 0 [H+] < 1 x 10 -7
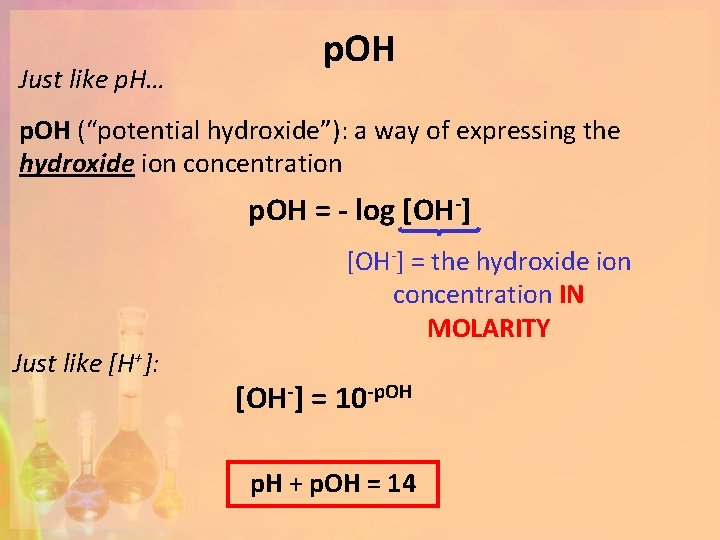
Just like p. H… p. OH (“potential hydroxide”): a way of expressing the hydroxide ion concentration p. OH = - log [OH-] = the hydroxide ion concentration IN MOLARITY Just like [H+]: [OH-] = 10 -p. OH p. H + p. OH = 14
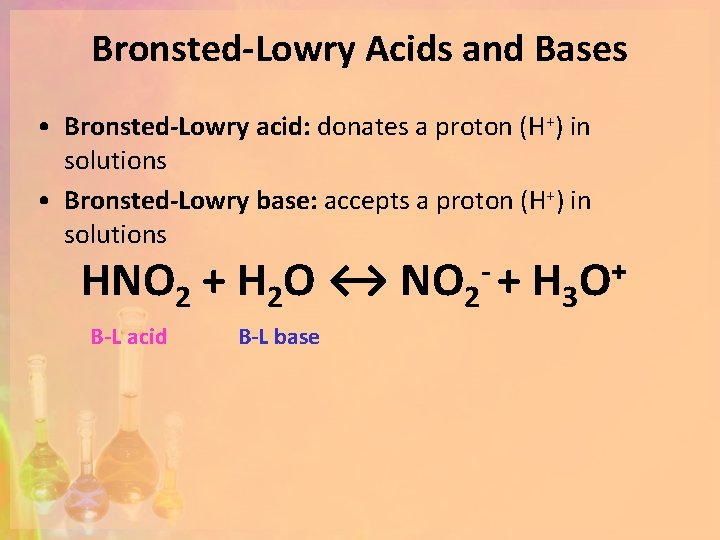
Bronsted-Lowry Acids and Bases • Bronsted-Lowry acid: donates a proton (H+) in solutions • Bronsted-Lowry base: accepts a proton (H+) in solutions HNO 2 + H 2 O ↔ NO 2 - + H 3 O+ B-L acid B-L base
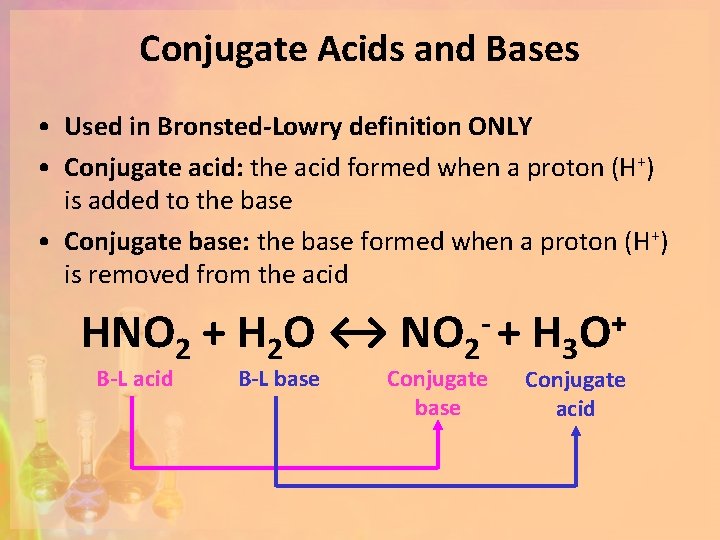
Conjugate Acids and Bases • Used in Bronsted-Lowry definition ONLY • Conjugate acid: the acid formed when a proton (H+) is added to the base • Conjugate base: the base formed when a proton (H+) is removed from the acid HNO 2 + H 2 O ↔ NO 2 - + H 3 O+ B-L acid B-L base Conjugate acid
- Slides: 16