Acids and Bases an Introduction Acids Sour Taste

Acids and Bases: an Introduction

Acids Sour Taste Electrolytes Identified by the presence of H+ ions (Arrhenius). Proton (H+) donor (Bronsted-Lowry). Common Acids: vinegar, citric acid, sulfuric acid, hydrochloric acid.
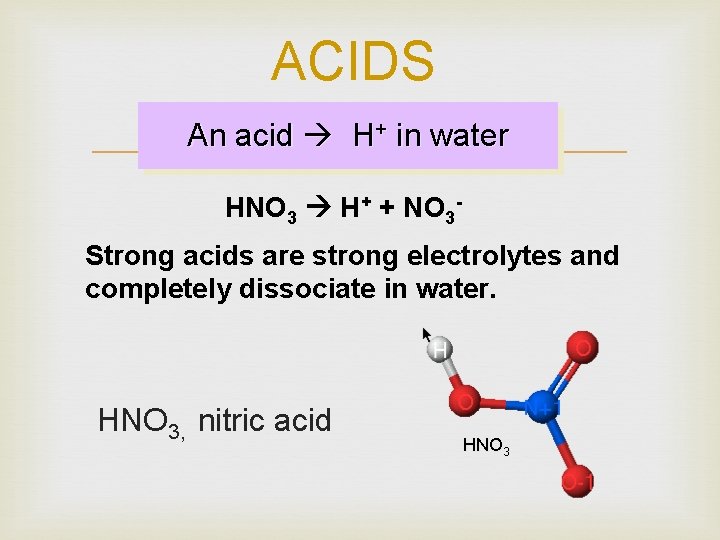
ACIDS An acid H+ in water HNO 3 H+ + NO 3 Strong acids are strong electrolytes and completely dissociate in water. HNO 3, nitric acid HNO 3
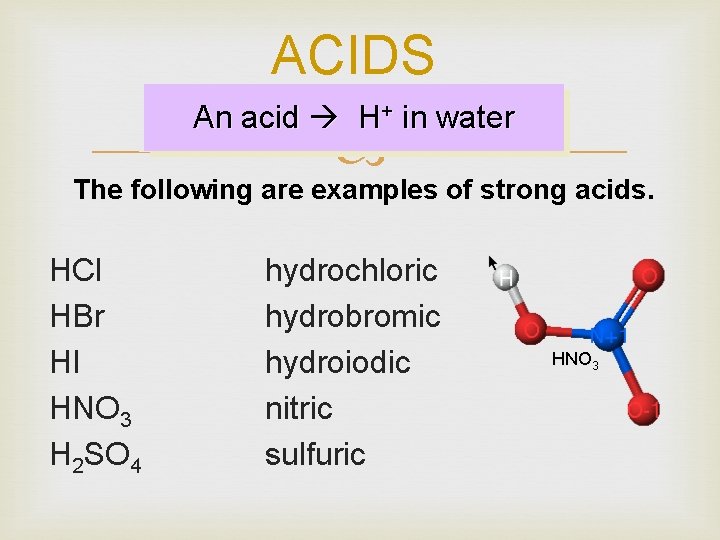
ACIDS An acid H+ in water The following are examples of strong acids. HCl HBr HI HNO 3 H 2 SO 4 hydrochloric hydrobromic hydroiodic nitric sulfuric HNO 3
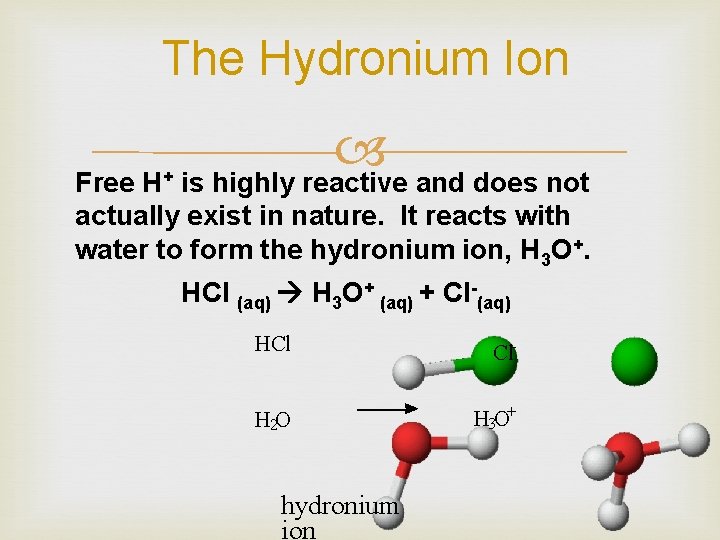
The Hydronium Ion is highly reactive and does not Free H+ actually exist in nature. It reacts with water to form the hydronium ion, H 3 O+. HCl (aq) H 3 O+ (aq) + Cl-(aq) HCl Cl- H 2 O H 3 O+ hydronium ion
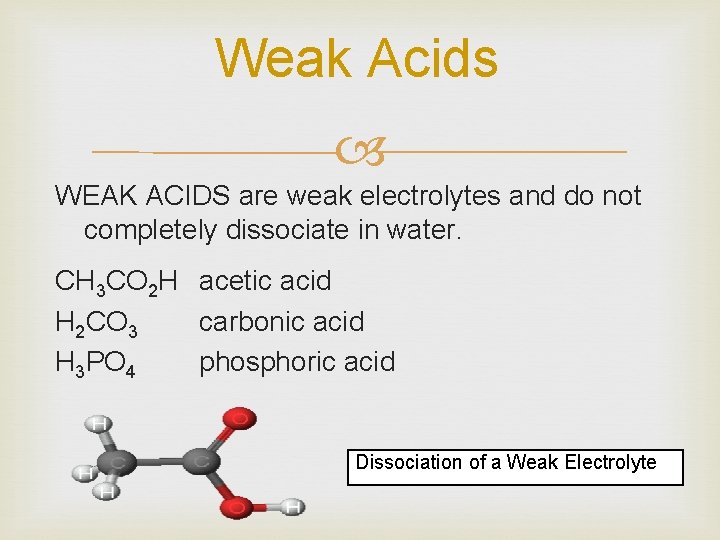
Weak Acids WEAK ACIDS are weak electrolytes and do not completely dissociate in water. CH 3 CO 2 H acetic acid H 2 CO 3 carbonic acid H 3 PO 4 phosphoric acid Dissociation of a Weak Electrolyte

Bases Bitter taste Slippery Like acids, they are electrolytes Identified by the presence of OHions (Arrhenius). Proton (H+) acceptor (Bronsted. Lowry).
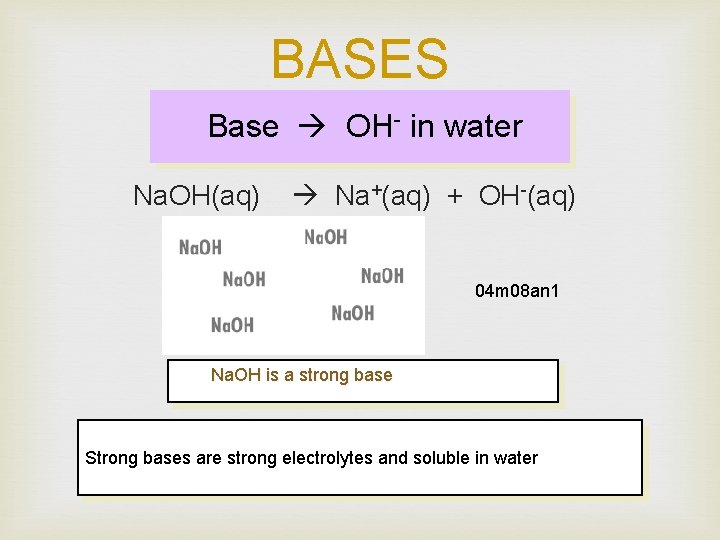
BASES Base OH- in water Na. OH(aq) Na+(aq) + OH-(aq) 04 m 08 an 1 Na. OH is a strong base Strong bases are strong electrolytes and soluble in water

Strength of Acids and Bases Based on the concentration of H+ or OH - ions in a solution. Strong Acids/Bases: completely dissociate into ions in a solution. Weak Acids/Bases: do NOT completely dissociate into ions in a solution.

Conjugate Acids/Bases Acids and bases are related to each other through the addition/loss of hydrogen ions Conjugate acid-base pairs Acids produce conjugate bases Bases produce conjugate acids

Conjugate Examples HA + H 2 O H 3 O+ + A- HNO 3 + NH 3 NH 4+ + NO 3 -

Conjugate Acid/Base Strength Stronger the acid, the weaker the conjugate base Stronger the base, the weaker the conjugate acid Weak acids/bases have strong conjugate bases/acids

Strong Acid Example: HCl

Weak Acid Example: CH 3 CO 2 H

“BIG 6”---Strong Acids (Know them!!) HCl. O 4 HI HCl HNO 3 HBr H 2 SO 4

Strong Bases (Know them!!) Group I metal hydroxides (Na. OH, KOH, etc. ) Soluble/Slightly soluble Group II metal hydroxides ( Ca(OH)2, Sr(OH)2, Ba(OH)2 )

Acid Nomenclature Binary acids– containing only 2 elements (one is hydrogen) 1) Prefix “hydro—” with binary acid 2) root name for second element 3) End the name with “IC acid” Oxyacids– acids containing hydrogen, oxygen, and a nonmetal 1) use the given polyatomic ion name from anion 2) add “IC acid”

Example 1: HBr Write name

Now you try, HCl Write name.

Example 2: Hydrofluoric acid Write chemical formula

Now you try, hydriodic acid.

Example 3: H 2 SO 4 Write the name.

Example 4: HCl. O 2 Write the name.
Try: HCl. O 3 HCl. O 4

Base Nomenclature Name of cation, name of anion/hydroxide Ex. Na. OH

Brainstorm… Briefly brainstorm how we measure the amount of hydrogen ions in a solution? How can we quickly measure the acidity or basicity of a solution?
- Slides: 26