Acids and Bases Acids and Bases Acid when

Acids and Bases

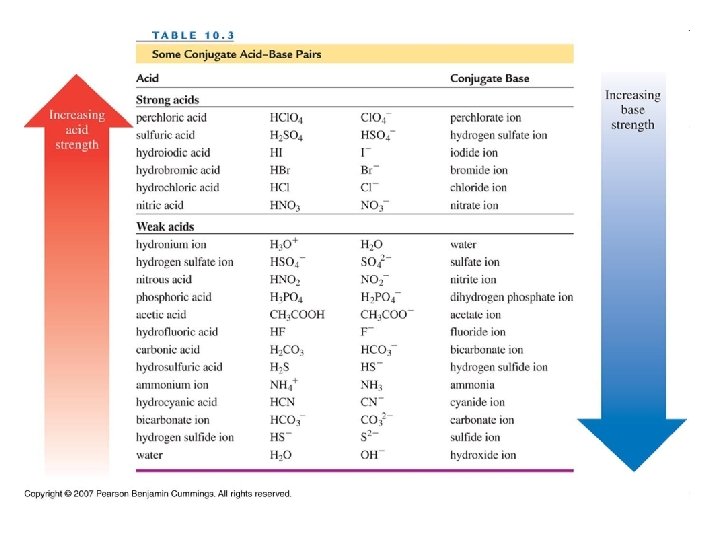
Acids and Bases • Acid: when dissolved in water, increases the concentration of H+ (arrenhius acid) • HCl H 2 O H+ + Cl • HCl + H 2 O H 3 O+ + Cl- • Strong acid: an acid that completely ionizes/dissociates in water • HNO 3 + H 2 O H 3 O+ + NO 3 • Strong electrolyte
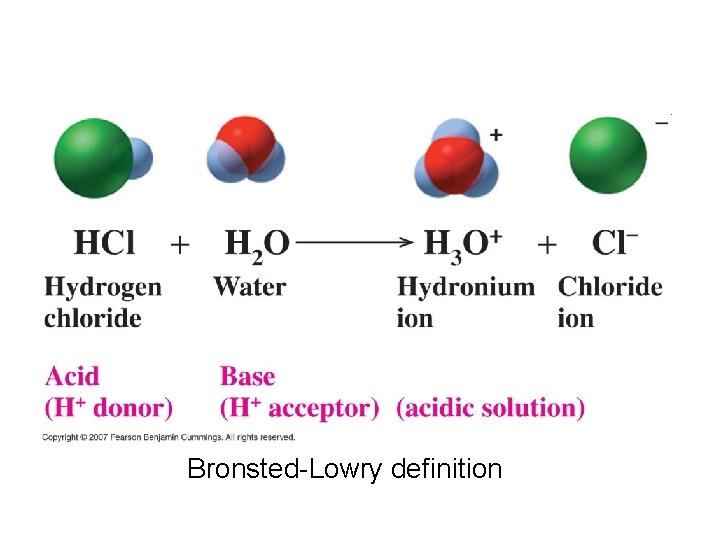
Bronsted-Lowry definition
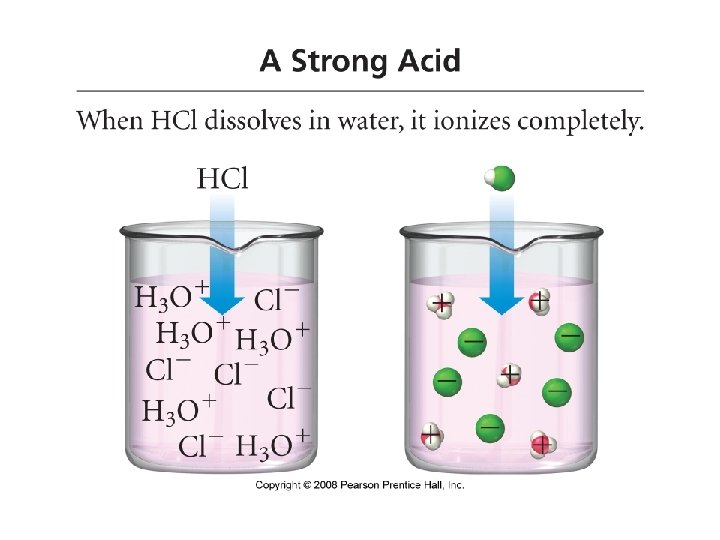

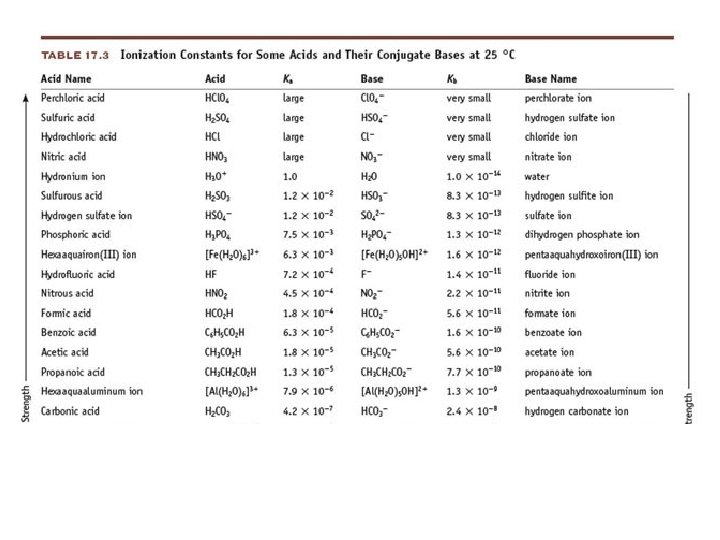
• Strong Acids: • • • Hydrochloric Acid Nitric Acid Sulfuric Acid Perchloric Acid Hydrobromic Acid Hydroiodic Acid HCl HNO 3 H 2 SO 4 HCl. O 4 HBr HI
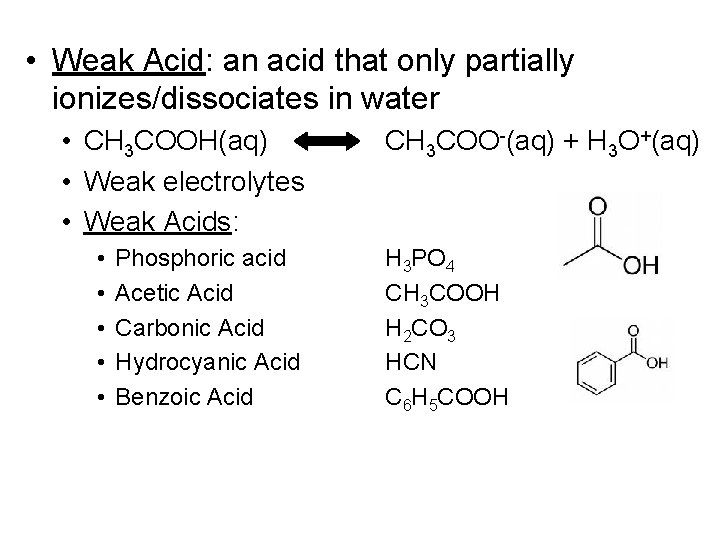
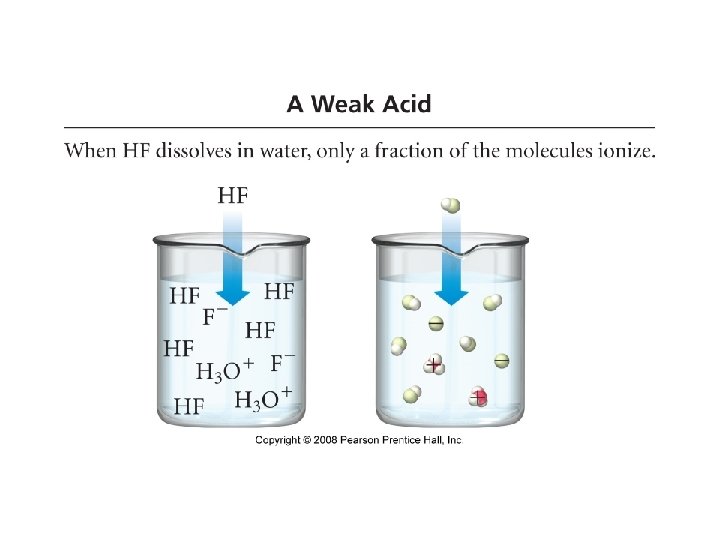
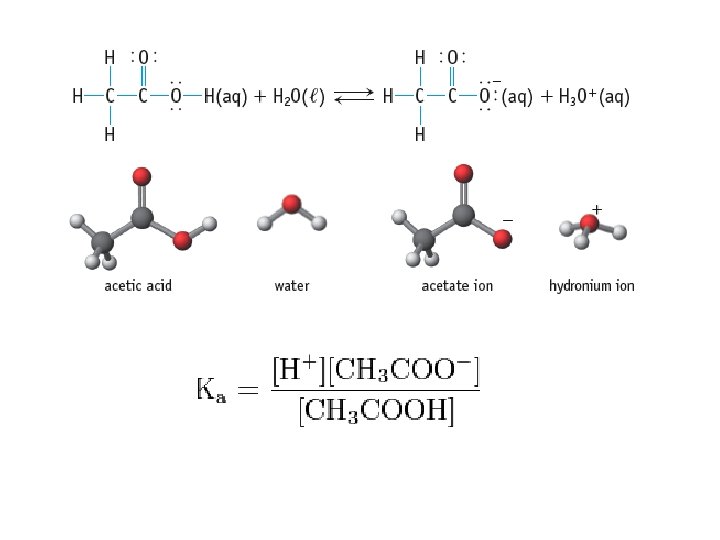
• Weak Acid: an acid that only partially ionizes/dissociates in water • CH 3 COOH(aq) • Weak electrolytes • Weak Acids: • • • Phosphoric acid Acetic Acid Carbonic Acid Hydrocyanic Acid Benzoic Acid CH 3 COO-(aq) + H 3 O+(aq) H 3 PO 4 CH 3 COOH H 2 CO 3 HCN C 6 H 5 COOH

Polyprotic Acids • Polyprotic acids: acids that can release more than one H+ • Sulfuric Acid • H 2 SO 4(aq) • HSO 4 -(aq) + H 3 O+(aq) SO 42 -(aq) + H 3 O+(aq)

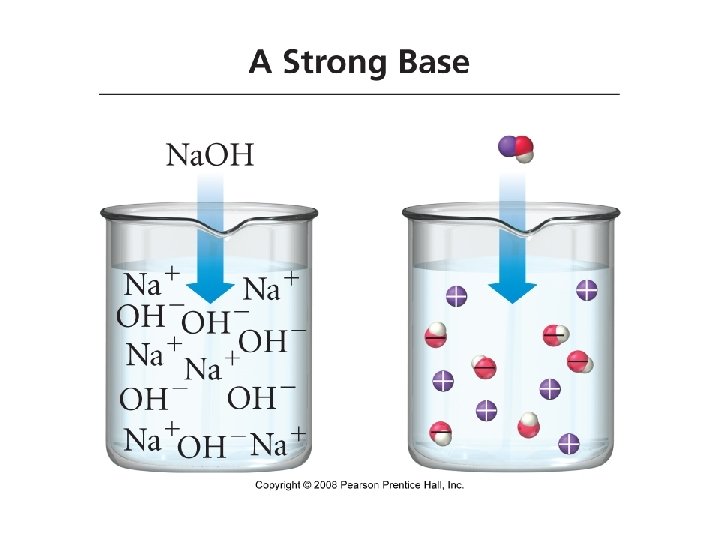
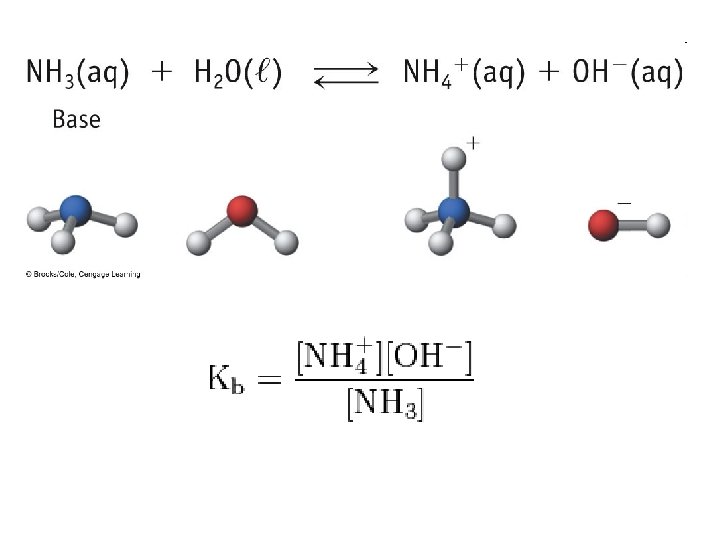
Bases • Base: a substance that, when put in water, increases the concentration of OH- ions or a substance that accepts H+ ions • Na. OH(aq) Na+(aq) + OH-(aq) • Strong Bases: bases that completely ionize in water

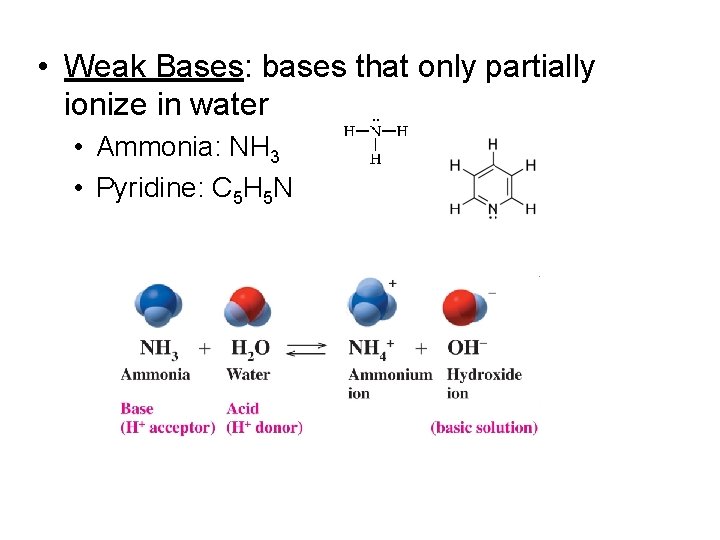
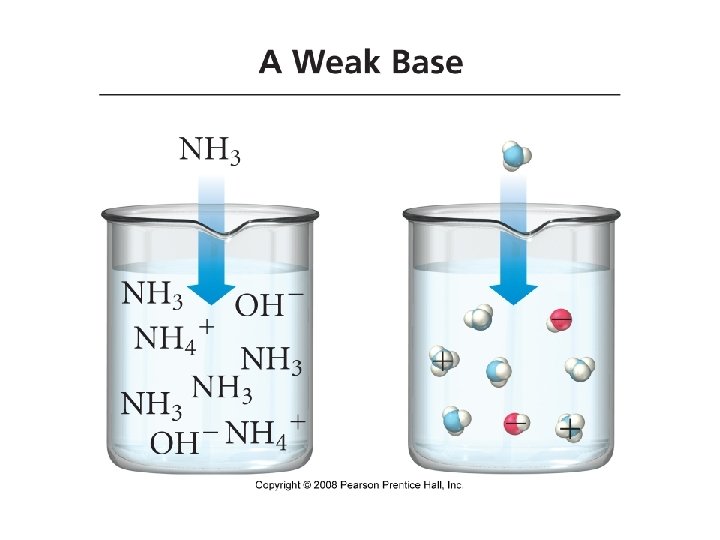
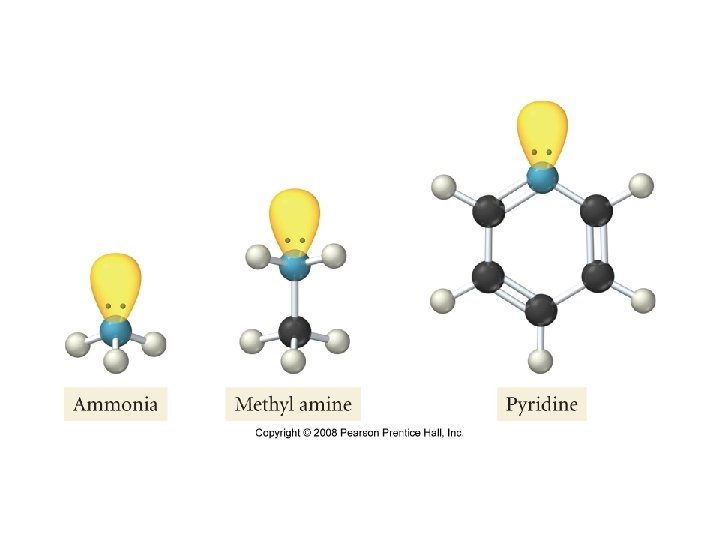
• Weak Bases: bases that only partially ionize in water • Ammonia: NH 3 • Pyridine: C 5 H 5 N
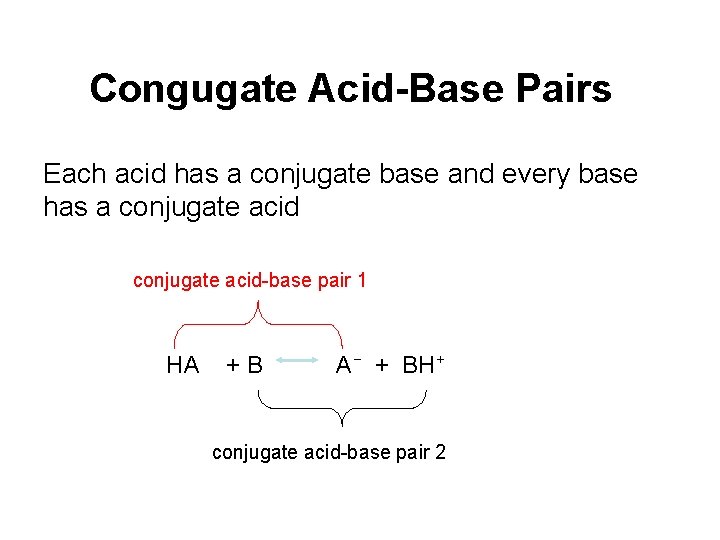
Congugate Acid-Base Pairs Each acid has a conjugate base and every base has a conjugate acid-base pair 1 HA +B A− + BH+ conjugate acid-base pair 2
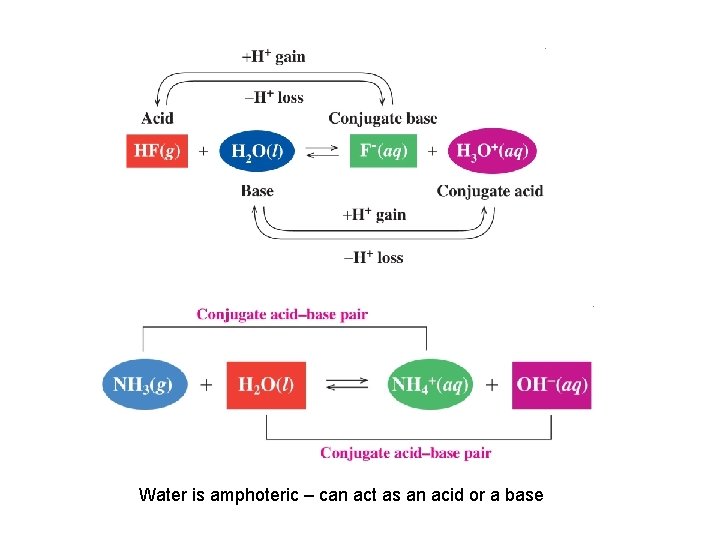
Water is amphoteric – can act as an acid or a base

Neutralization Reactions • When strong acids and bases in aqueous solution react with each other, they form water and a salt. • HX(aq) + MOH(aq) HOH(l) + MX(aq) Water Salt • HCl(aq) + Na. OH(aq) H 2 O(l) + Na. Cl(aq)

Acid Ionization Constant • Acid Ionization Constant (Ka): the equilibrium constant for the ionization reaction of an acid with water • HA + H 2 O A - + H 3 O + • Large Ka = Strong acid • Small Ka = Weak acid

Base Ionization Constant • Base Ionization Constant (Kb): the equilibrium constant for the ionization reaction of a base with water B + H 2 O OH- + BH+ • Large Kb = Strong base • Small Kb = Weak base

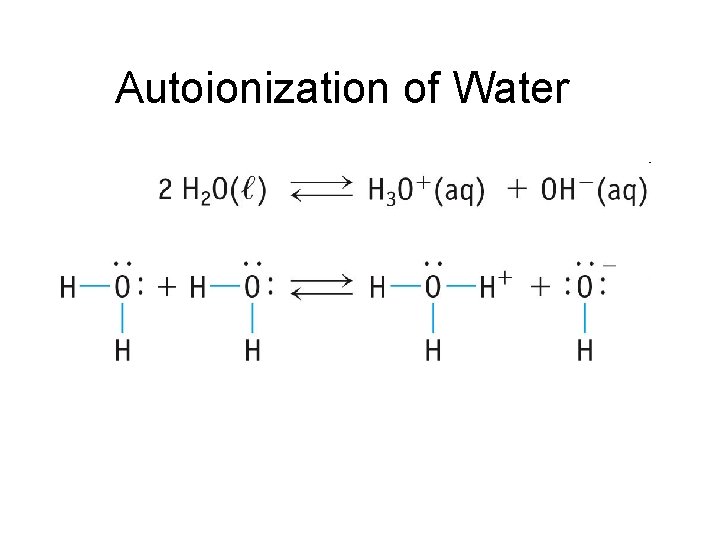
Autoionization of Water
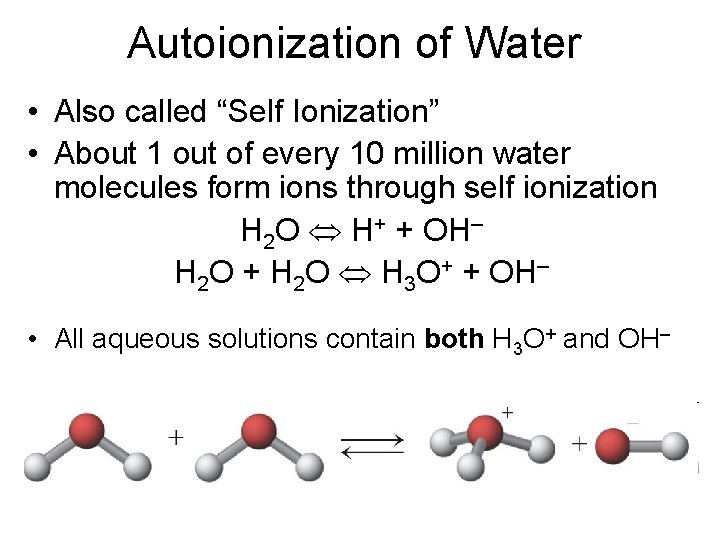
Autoionization of Water • Also called “Self Ionization” • About 1 out of every 10 million water molecules form ions through self ionization H 2 O H+ + OH– H 2 O + H 2 O H 3 O+ + OH– • All aqueous solutions contain both H 3 O+ and OH–

Ion Product Constant for Water • Ion Product Constant for Water (Kw): the numerical value obtained by multiplying the molar concentrations for hydronium and hydroxide ions present in pure water at 25°C • Kw = [H 3 O+][OH-] = 1. 00 x 10 -14 at 25 o. C • the concentration of H 3 O+ and OH– are equal in pure water • [H 3 O+] = [OH–] = 10 -7 M @ 25°C

Ion Product of Water • the product of the H 3 O+ and OH– concentrations is always the same number • Kw =[H 3 O+][OH–] = 1. 00 x 10 -14 @ 25°C – if you measure one of the concentrations, you can calculate the other • as [H 3 O+] increases the [OH–] must decrease so the product stays constant – inversely proportional
![[H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10 [H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10](http://slidetodoc.com/presentation_image_h2/d57fb41ab6c7087f4701ff3e994e6d1c/image-30.jpg)
[H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10 -5 + H OH- [OH-]10 -14 10 -13 10 -11 10 -9 10 -7 10 -9 Base 10 -11 H+ + H OH OH 10 -7 10 -5 10 -3 10 -14 H+ OH 10 -1 100 Even though it may look like it, neither H+ nor OH- will ever be 0
![Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–] Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–]](http://slidetodoc.com/presentation_image_h2/d57fb41ab6c7087f4701ff3e994e6d1c/image-31.jpg)
Acidic and Basic Solutions • Neutral solutions have equal [H 3 O+] and [OH–] • [H 3 O+] = [OH–] = 1 x 10 -7 • acidic solutions have a larger [H 3 O+] than [OH–] • [H 3 O+] > 1 x 10 -7; [OH–] < 1 x 10 -7 • basic solutions have a larger [OH–] than [H 3 O+] • [H 3 O+] < [OH–] • [H 3 O+] < 1 x 10 -7; [OH–] > 1 x 10 -7
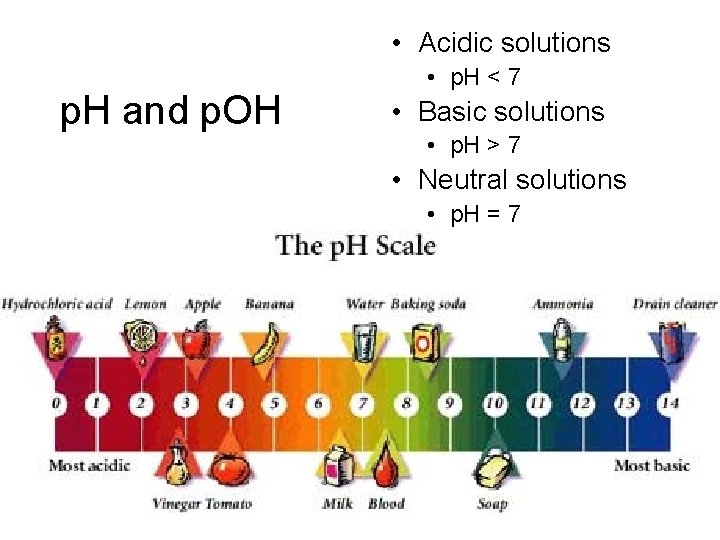
• Acidic solutions p. H and p. OH • p. H < 7 • Basic solutions • p. H > 7 • Neutral solutions • p. H = 7
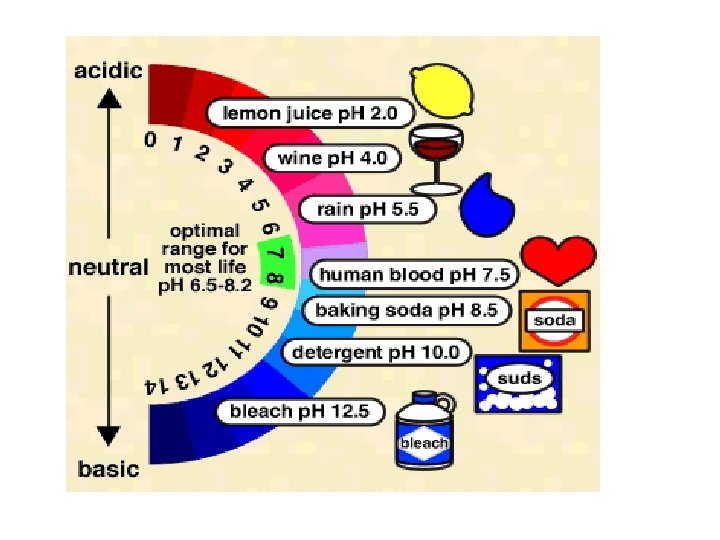

• p. H is a measure of the concentration of H 3 O+ in solution • p. H = -log [H+] OR p. H = -log [H 3 O+] • [H 3 O+] = 10 -p. H • p. OH is a measure of the concentration of OH- in solution • p. OH = -log [OH-] • [OH-] = 10 -p. OH • p. H + p. OH = 14

Classification of Water Soluble Substances • Electrolytes: solutes that separate into ions when dissolved in water (they’re soluble) – Have the ability to conduct electricity – 2 types • Strong electrolytes • Weak electrolytes
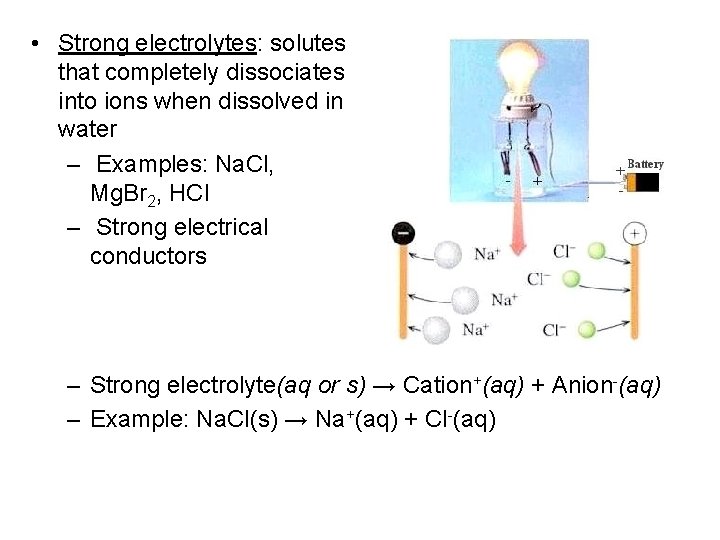
• Strong electrolytes: solutes that completely dissociates into ions when dissolved in water – Examples: Na. Cl, Mg. Br 2, HCl – Strong electrical conductors – Strong electrolyte(aq or s) → Cation+(aq) + Anion-(aq) – Example: Na. Cl(s) → Na+(aq) + Cl-(aq)
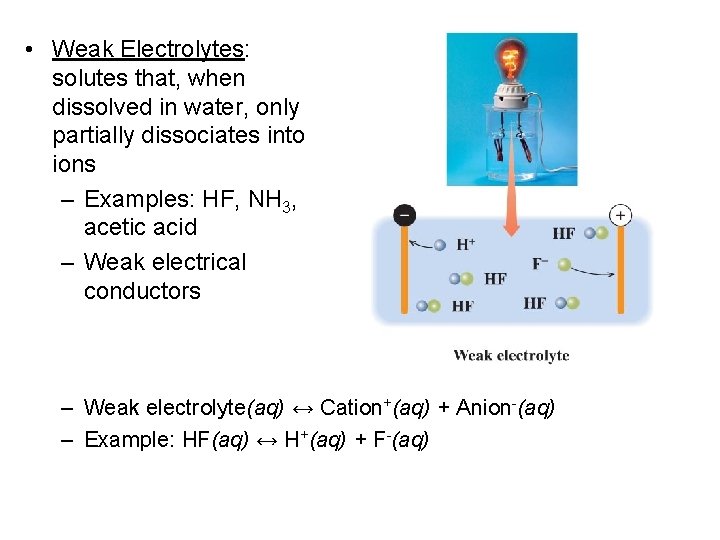
• Weak Electrolytes: solutes that, when dissolved in water, only partially dissociates into ions – Examples: HF, NH 3, acetic acid – Weak electrical conductors – Weak electrolyte(aq) ↔ Cation+(aq) + Anion-(aq) – Example: HF(aq) ↔ H+(aq) + F-(aq)
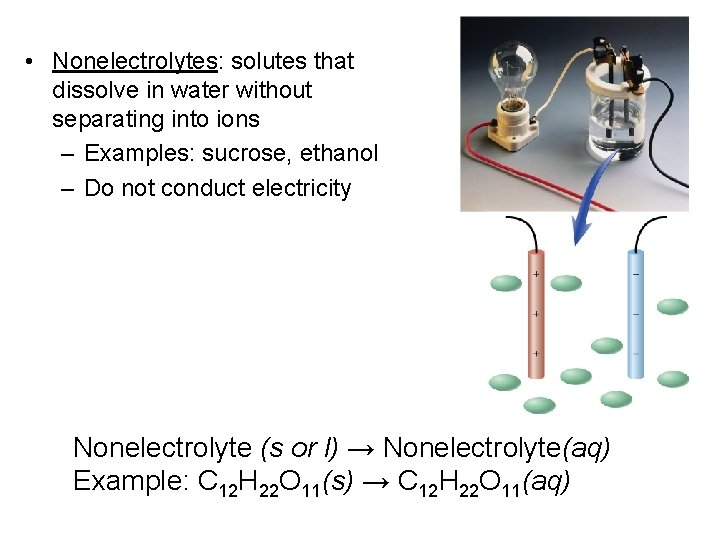
• Nonelectrolytes: solutes that dissolve in water without separating into ions – Examples: sucrose, ethanol – Do not conduct electricity Nonelectrolyte (s or l) → Nonelectrolyte(aq) Example: C 12 H 22 O 11(s) → C 12 H 22 O 11(aq)
Titration • Titration: a procedure for the quantitative analysis of a substance of unknown concentration whereby a measured quantity of another substance, of know concentration, is completely reacted with the original substance. – Often used to determine the concentration of acids and bases
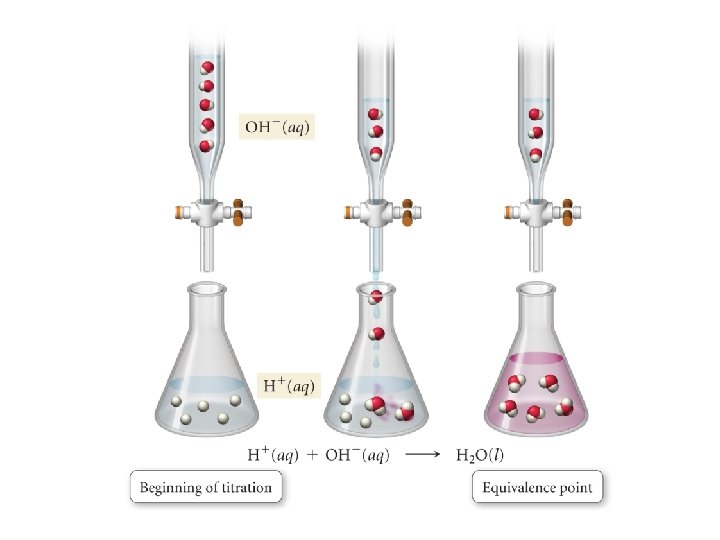
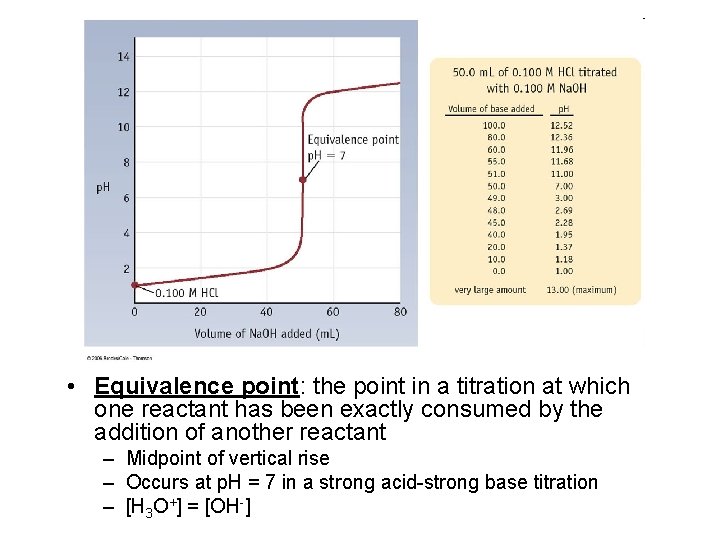
• Equivalence point: the point in a titration at which one reactant has been exactly consumed by the addition of another reactant – Midpoint of vertical rise – Occurs at p. H = 7 in a strong acid-strong base titration – [H 3 O+] = [OH-]

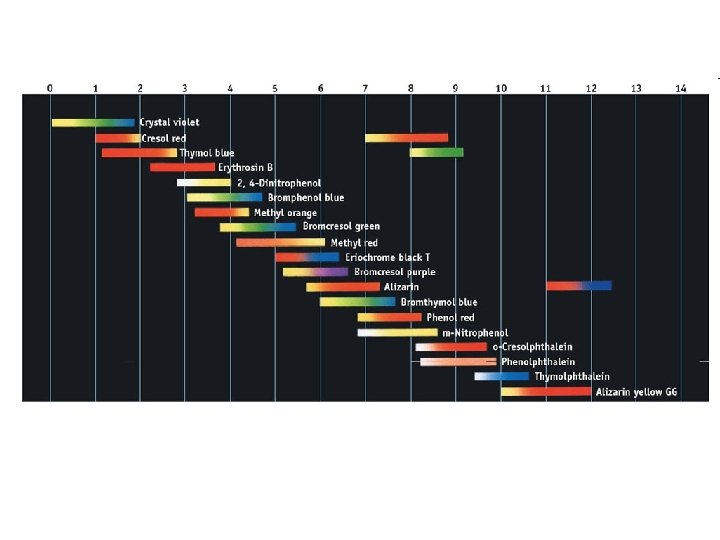
Indicators • Acid-Base Indicator: a chemical that changes color with a change in p. H – Added to solutions in small amounts in order to determine to solution’s p. H visually – Usually organic compounds – Weak acid or base – establishes an equilibrium with the H 2 O and H 3 O+ in the solution
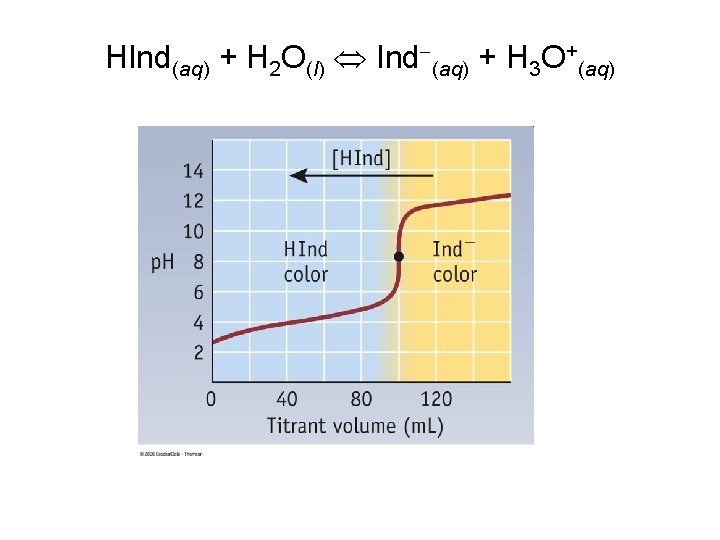
HInd(aq) + H 2 O(l) Ind (aq) + H 3 O+(aq)
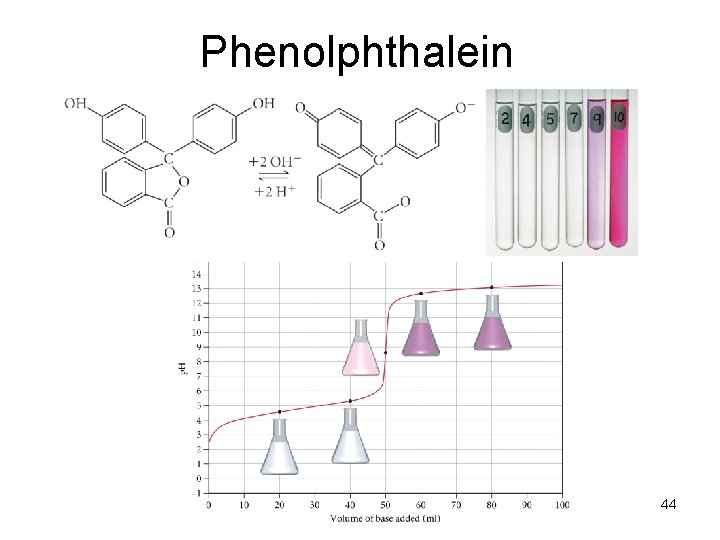
Phenolphthalein 44
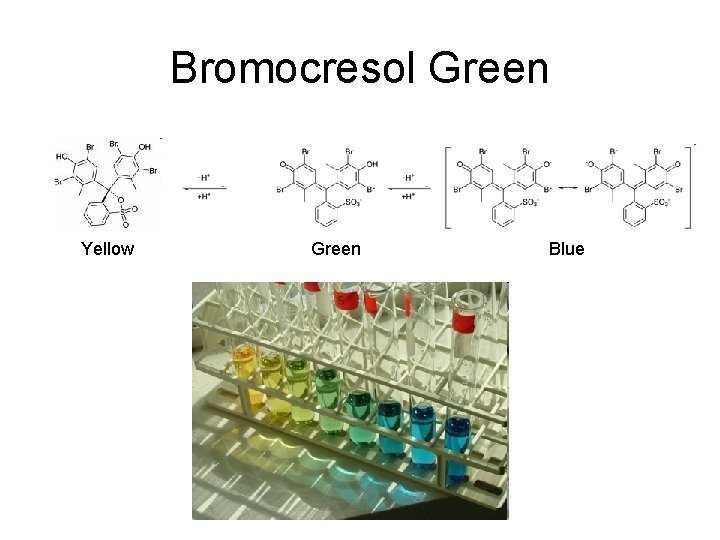
Bromocresol Green Yellow Green Blue
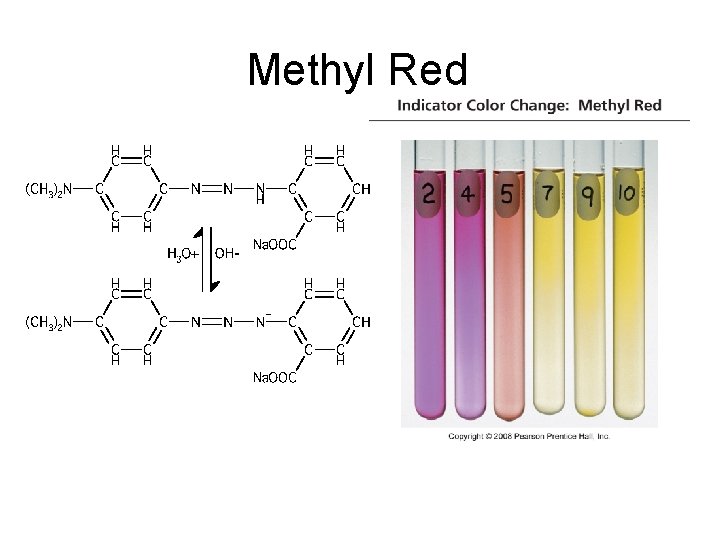
Methyl Red
- Slides: 47