Acids and Bases Acidity Basicity and p H

Acids and Bases Acidity, Basicity, and p. H

Self-Ionization of Water � Water is both an acid and a base. We call this _________. � By the BrØnsted-Lowry definition, this means that water can donate a proton AND accept a proton. H 2 O ( l) + H 2 O ( l) H 3 O+(aq) + OH-(aq) process is known as the self-ionization of water. � This

Concentration of Water � The self-ionization of water makes even pure water have ions within it. � In pure water, the two ions (acidic and basic) must share the same concentration (1: 1 ratio) � Experiments 1. 00 x 10 -7 show that this concentration is M at 25°C. [H 3 O+] = [OH-] = 1. 00 x 10 -7 M
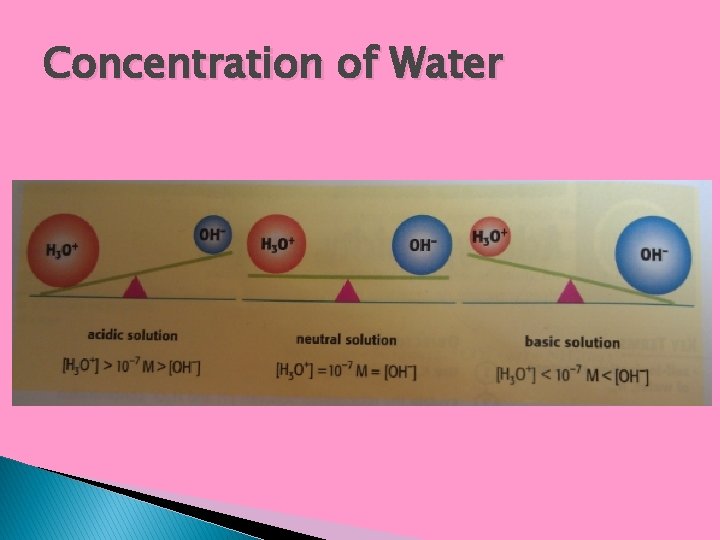
Concentration of Water

Equilibrium Constant � The equilibrium constant is an expression that relates the concentrations of species involved in an equilibrium. � To write an equilibrium constant expression, you must know the concentrations of the products and the reactants at equilibrium. Keq = [Products] / [Reactants] **Excluding solids and liquids

Equilibrium Constant � Consider the equilibrium of water: H 2 O ( l) + H 2 O ( l) 2 H 2 O(l) � This H 3 O+(aq) + OH-(aq) equation’s Equilibrium Constant is: Keq = (1. 00 x 10 -7) = 1. 00 x 10 -14 � This Kw is a special equilibrium constant, known as

Self-Ionization Constant of Water � Keq = (1. 00 x 10 -7) = 1. 00 x 10 -14 � This is the Self-Ionization Constant of Water ◦ Known as Kw � We can use this information about water and the relationship of hydronium and hydroxide to figure out various concentrations in solutions [H 3 O+][OH-] = Kw = 1. 00 x 10 -14
![Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M](http://slidetodoc.com/presentation_image/4a4ef03e875a620f727680c874b144dc/image-8.jpg)
Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M solution of 1. HCl = a strong acid or a strong base? I. So, is this a hydroxide conc or a hydronium conc? II. Since this is a strong acid it completely _______, implying that [HCl] = [H 3 O+] 2. Remember the self-ionization constant of water I. Kw = [H 3 O+][OH-] = 1. 00 x 10 -14 3. Plug in your variables and calculate what your are asked to find.
![Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M](http://slidetodoc.com/presentation_image/4a4ef03e875a620f727680c874b144dc/image-9.jpg)
Practice � What HCl? is [OH-] in a 3. 00 x 10 -5 M solution of Kw = [H 3 O+][OH-] = 1. 00 x 10 -14 = (3. 00 x 10 -5 M)[OH-] 1. 00 x 10 -14 = [OH-] 3. 00 x 10 -5 M [OH-] = 3. 33 x 10 -10 M

p. H �A solution is neutral when the concentration of hydroxide is exactly equal to the concentration of the hydronium. p. H = 7 � If the conc of the hydronium is stronger than the conc of the hydroxide than the solution with have a p. H of <7. � If the conc of the hydroxide is stronger than the conc of the hydronium than the solution with have a p. H of >7.

p. H � The p. H is a value used to express the acidity or alkalinity of a solution; it is defined as the logarithm of the reciprocal of the concentration of hydronium ions. �p = power, H = hydrogen � p. H = power of hydrogen p. H = -log[H 3 O+] = 10 -p. H

p. H Scale � p. H is a logarithmic scale, meaning an increase in a value is an increase of 10 x � i. e. A solution whose p. H is 2. 0 has a [H 3 O+] that is 10 x greater than a solution whose p. H is 3. 0 and a 100 x greater than a solution whose p. H is 4. 0

Practice #1 � What is the p. H of the following solutions: ◦ 0. 00010 M solution of HNO 3? ◦ 0. 0136 M solution of KOH?

Practice #2 � What are the concentrations of the hydronium and hydroxide ions in a sample of rain that has a p. H of 5. 05? Gather info: p. H = 5. 05 Kw = 1. 00 x 10 -14 [H 3 O+] = ? [OH-] = ? � Plan your work

Measuring p. H � Method #1 � Method #2 ◦ Uses indicators ◦ Quick and simple ◦ Does not, however, give very precise results ◦ Uses a p. H meter ◦ Very precise ◦ More complicated and expensive

Measuring p. H � Certain dyes with change different colors in solutions of different p. H � These dyes are known as indicators. ◦ A compound that can reversibly change color depending on the p. H of the solution or other chemical change. � The color after the addition of the indicator can help determine the relative p. H of the solution.

Litmus Paper � Litmus is a dye used as an indicator that is soaked into filter paper � It serves as a quick, cheap, convenient check of a solution’s p. H. � Blue Litmus Paper – Turns red under acidic conditions � Red Litmus Paper – Turns blue under basic conditions

Measuring p. H � p. H meters are a probe that is submerged into a solution. � An electrical voltage develops between two electrodes and the probe digitally reads out a p. H value. � The voltage is a measure of the conductivity of the hydronium solution.

Homework � Page 547, # 1, 2, 5, 6, 7, 8, 9, 10 � Due tomorrow!! � Vocab also due on Wednesday.
- Slides: 19