ACIDS AND BASES 1 Weak Strong Acetic Acid

ACIDS AND BASES
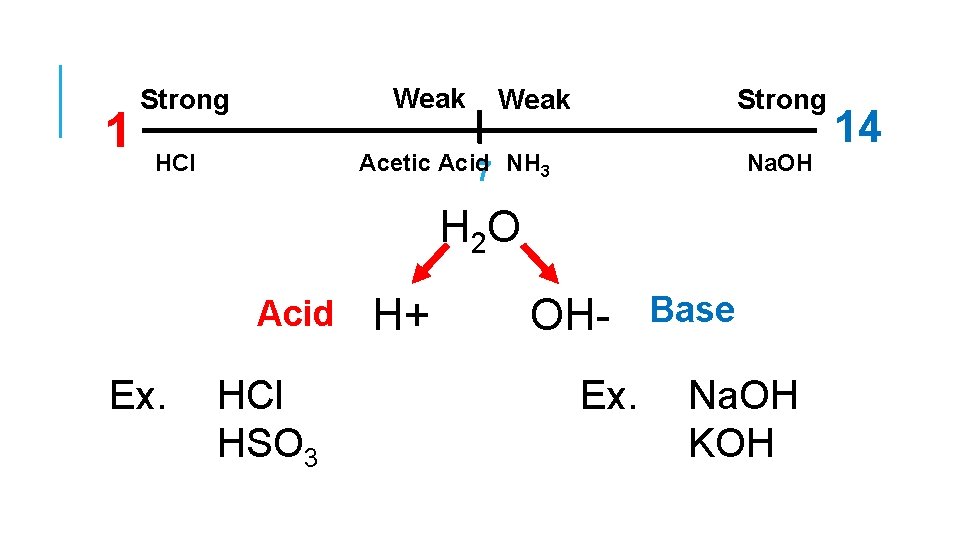
1 Weak Strong Acetic Acid 7 NH 3 HCl Na. OH H 2 O Acid Ex. HCl HSO 3 H+ OH- Base Ex. Na. OH KOH 14

ARRHENIUS ACIDS Substances that produce H+ (hydrogen ion, proton) in soln. Properties of acids are related to the H+ ions they produce Increase H+ concentration when added to H 2 O Decrease OH- concentration when added to H 2 O Decreases p. H, x<7 Turn blue litmus paper red Sour No effect on phenolphthalein, stays colorless React with certain metals to produce salt and H 2

ARRHENIUS ACIDS • Not all compounds that contain hydrogens are acids. Ex. CH 3 COOH : acetic acid, i. e. Vinegar • Only hydrogens in very polar bonds are ionizable. • Hydrogen ions are stabilized by solvation. Some definitions: Monoprotic Acids – acids that contain one ionizable hydrogen. Ex. HNO 3 Diprotic Acids – acids that contain two ionizable hydrogens. Ex. H 2 SO 4 Triprotic Acids – acids that contain three ionizable hydrogens. Ex. H 3 PO 4

ARRHENIUS BASE Substances that produce OH- (hydroxide ion) in soln. Properties of bases are related to the OH- ions they produce Increase OH- concentration when added to H 2 O Decrease H+ concentration when added to H 2 O Increases p. H, x>7 Turn red litmus paper blue Bitter and slippery Turns colorless phenolphthalein, pink. Most bases are ionic compounds.
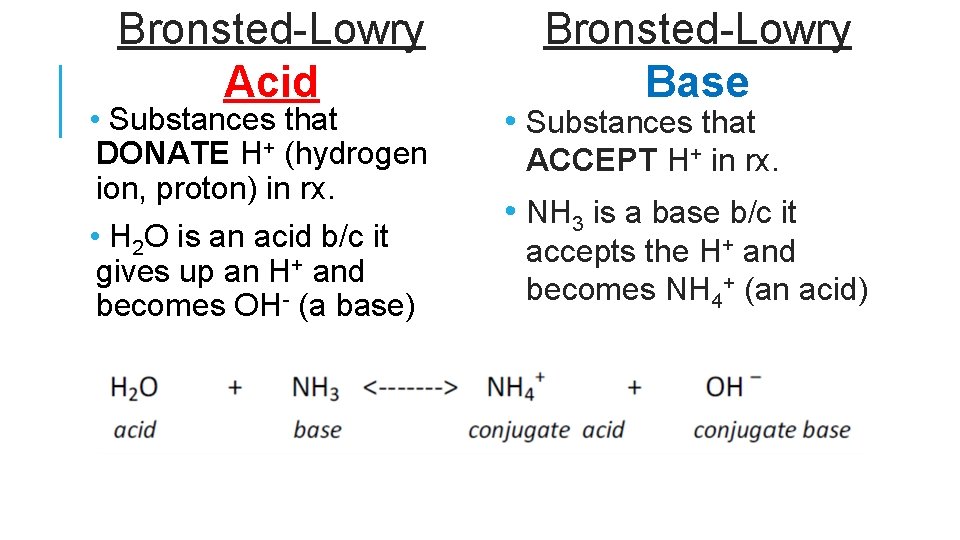
Bronsted-Lowry Acid • Substances that DONATE H+ (hydrogen ion, proton) in rx. • H 2 O is an acid b/c it gives up an H+ and becomes OH- (a base) Bronsted-Lowry Base • Substances that ACCEPT H+ in rx. • NH 3 is a base b/c it accepts the H+ and becomes NH 4+ (an acid)

Bronsted- Lowry Theory CONJUGATE ACIDS & BASES Conjugate - coupled, connected, or related, in particular. In the case of the reverse reaction Øthe conjugate acid is the acid; H+ donor on the “product” side Øthe conjugate base is base; H+ acceptor on the “product” side. • In essence, the reversible reaction of ammonia and water has two acids and two bases.

Bronsted- Lowry Theory CONJUGATE ACIDS & BASES ØWhich substance is the B-L acid (H+ donor)? ØWhich substance is the B-L base (H+ acceptor)? ØWhat substance is the conjugate base? ØWhat substance is the conjugate acid?

Bronsted- Lowry Theory CONJUGATE ACIDS & BASES Conjugate acids are always paired with a base, and conjugate bases are always paired with an acid. • A conjugate acid-base pair consists of two ions or molecules related by the loss or gain of one hydrogen ion.

Bronsted- Lowry Theory CONJUGATE ACIDS & BASES ØWhich substance is the B-L acid (H+ donor)? ØWhich substance is the B-L base (H+ acceptor)? ØWhat substance is the conjugate base? ØWhat substance is the conjugate acid?
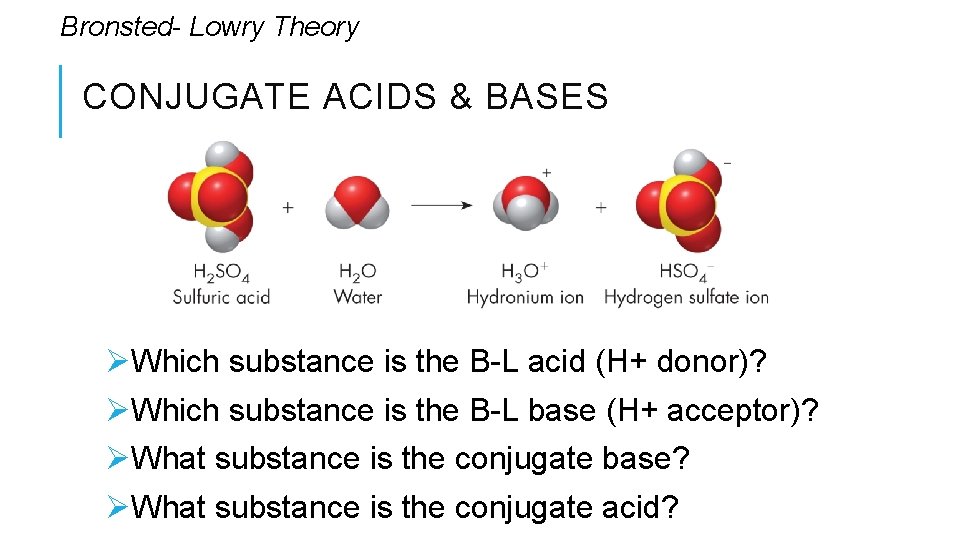
Bronsted- Lowry Theory CONJUGATE ACIDS & BASES ØWhich substance is the B-L acid (H+ donor)? ØWhich substance is the B-L base (H+ acceptor)? ØWhat substance is the conjugate base? ØWhat substance is the conjugate acid?

Bronsted- Lowry Theory AMPHOTERIC SUBSTANCES A substance that can act as either an acid or a base is said to be amphoteric. • Water is amphoteric. –In the reaction with hydrochloric acid, water accepts a proton and is therefore a base. –In the reaction with ammonia, water donates a proton and is therefore an acid.

LEWIS ACIDS AND BASES • A Lewis acid is a substance that can accept a pair of electrons to form a covalent bond. • A Lewis base is a substance that can donate a pair of electrons to form a covalent bond. The Lewis definitions include all the Brønsted-Lowry acids and bases.
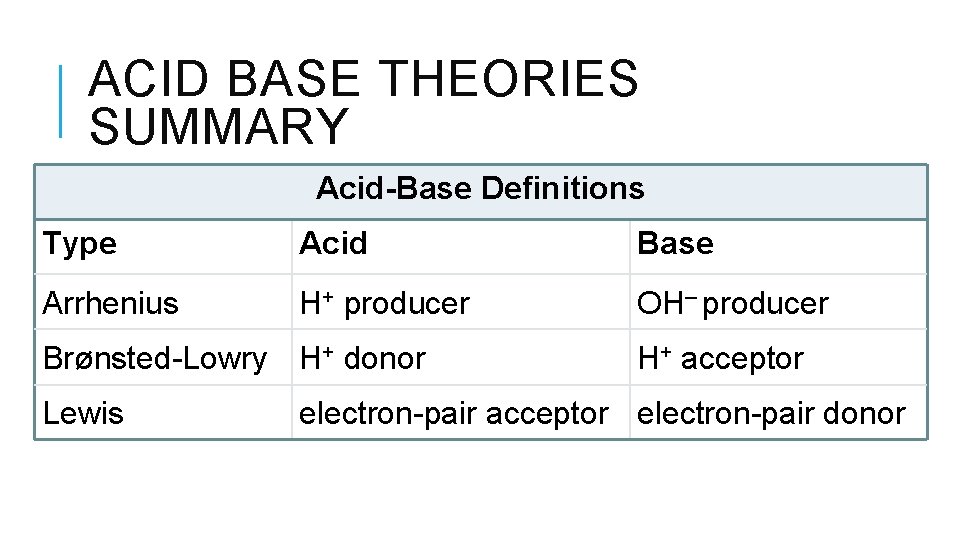
ACID BASE THEORIES SUMMARY Acid-Base Definitions Type Acid Base Arrhenius H+ producer OH– producer Brønsted-Lowry H+ donor Lewis H+ acceptor electron-pair donor
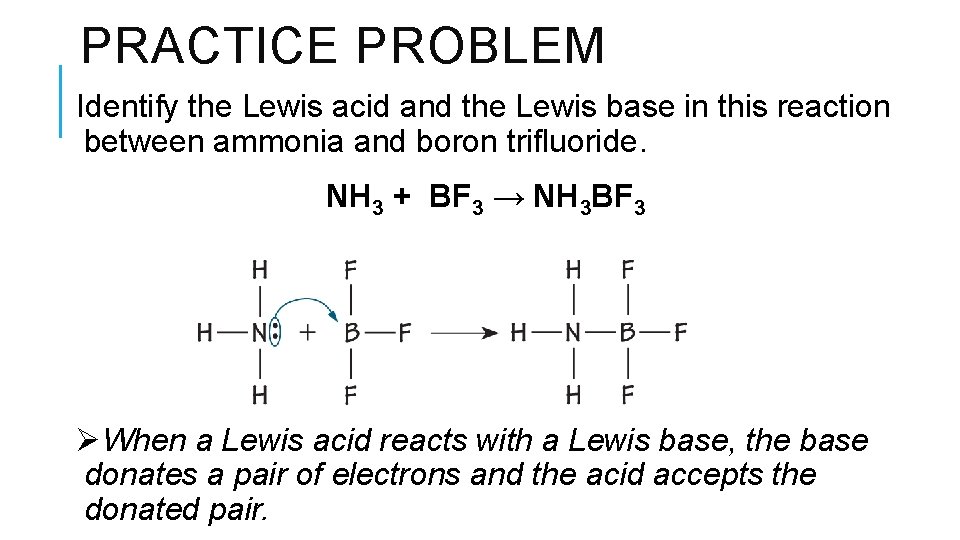
PRACTICE PROBLEM Identify the Lewis acid and the Lewis base in this reaction between ammonia and boron trifluoride. NH 3 + BF 3 → NH 3 BF 3 ØWhen a Lewis acid reacts with a Lewis base, the base donates a pair of electrons and the acid accepts the donated pair.

ADDITIONAL PRACTICE PROBLEMS Identify the Lewis acid and Lewis base in each of the reactions. 1. H+ + H 2 O H 3 O+ 2. Al. Cl 3 + Cl- Al. Cl 43. Would you predict PCl 3 to be a Lewis acid or a Lewis base in a typical reaction? Explain your prediction. 4. KOH + HBr KBr + H 2 O 5. HCl + H 2 O H 3 O+ + Cl-
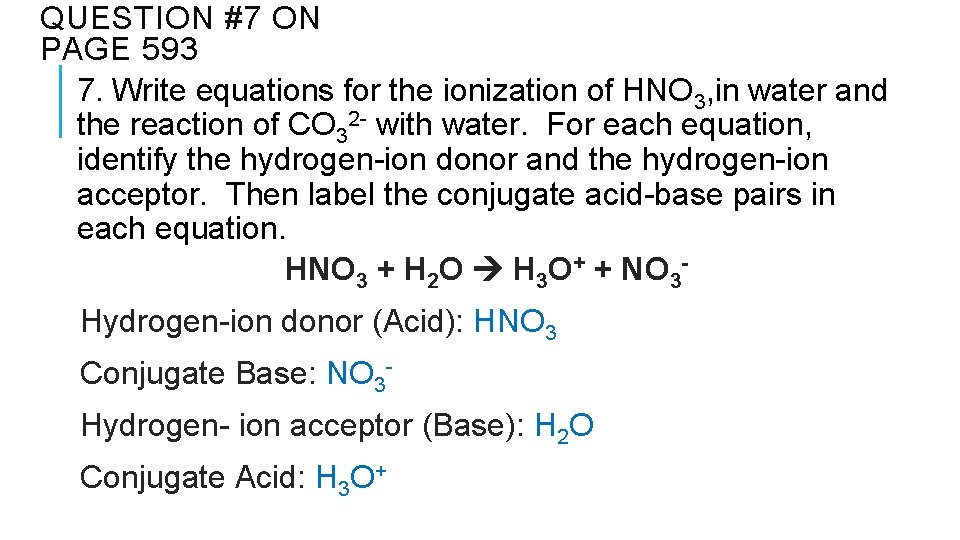
QUESTION #7 ON PAGE 593 7. Write equations for the ionization of HNO 3, in water and the reaction of CO 32 - with water. For each equation, identify the hydrogen-ion donor and the hydrogen-ion acceptor. Then label the conjugate acid-base pairs in each equation. HNO 3 + H 2 O H 3 O+ + NO 3 Hydrogen-ion donor (Acid): HNO 3 Conjugate Base: NO 3 Hydrogen- ion acceptor (Base): H 2 O Conjugate Acid: H 3 O+

ACID BASE INDICATORS Substances that change colors in the presence of an acid or base.

SECTION 2 Hydrogen Ions and Acidity

ION PRODUCT CONSTANT FOR WATER The ionization of water is a reversible reaction, so Le Châtelier’s principle applies. ØAdding either hydrogen ions or hydroxide ions to an aqueous solution is a stress on the system. ØIn response, the equilibrium will shift toward the formation of water. • The concentration of the other ion will decrease.

ION PRODUCT CONSTANT FOR WATER The ionization of water is a reversible reaction, so Le Châtelier’s principle applies. In any aqueous solution, when [H+] increases, [OH−] decreases. Likewise, when [H+] decreases, [OH−] increases.

ION PRODUCT CONSTANT FOR WATER Acidic Basic HCl(aq) → H+(aq) + Cl−(aq) Na. OH(aq) → Na+(aq) + OH−(aq) • A solution in which [H+] is greater than [OH−] is an acidic solution. • The [H+] is greater than 1 × 10− 7 M. • A basic solution is one in which [H+] is less than [OH−]. • The [H+] is less than 1 × 10− 7 M. AKA alkaline solutions

ION PRODUCT CONSTANT FOR WATER For aqueous solutions, the product of the hydrogen-ion concentration and the hydroxide-ion concentration equals 1. 0 × 10− 14. The product of the concentrations of the hydrogen ions and the hydroxide ions in water is called the ionproduct constant for water (Kw). Kw = [H+] × [OH−] = 1. 0 × 10− 14
![Sample Problem 19. 2 USING THE ION PRODUCT CONSTANT FOR WATER If the [H+] Sample Problem 19. 2 USING THE ION PRODUCT CONSTANT FOR WATER If the [H+]](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-24.jpg)
Sample Problem 19. 2 USING THE ION PRODUCT CONSTANT FOR WATER If the [H+] in a solution is 1. 0 × 10− 5 M, is the solution acidic, basic, or neutral? What is the [OH−] of this solution? [H+] is 1. 0 × 10− 5 M, which is greater than 1. 0 × 10− 7 M. Rearrange Kw = [H+] × [OH−] = Kw [H+] [OH −] = 1. 0 × 10− 9 M

RELATING PH TO H + ION CONCENTRATION Mathematically, p. H is defined as the –log of H+ ion concentration of a soln. p. H = -log[H+] or [H+] = 10 -p. H Øp. H is a measure of how much H+ are in a solution. If H+ concen. Is not given as 1 x 10 -x : Ex. [H+] = 5. 4 x 10 -8 M p. H = -log(5. 4 x 10 -8) = 7. 3

CALCULATING PH To calculate the p. H of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The p. H is then calculated using the expression: p. H = - log [H 3 O+] Example: Find the p. H of a 0. 0025 M HCl solution. The HCl is a strong acid and is 100% ionized in water. The hydronium ion concentration is 0. 0025 M. Thus: p. H = - log (0. 0025) = - ( - 2. 60) = 2. 60

CALCULATING PH MORE PROBLEMS Example: What is the p. H of a solution with a hydrogen-ion concentration of 4. 2 × 10− 10 M? p. H = - log [H 3 O+] p. H = - log [4. 2 x 10 -10 M] p. H = - (-9. 37675) p. H = 9. 38 (Base)

BELL RINGER ACTIVITY 1. 2.
![p. H = - log [H 3 O+] CALCULATING THE HYDRONIUM ION CONCENTRATION FROM p. H = - log [H 3 O+] CALCULATING THE HYDRONIUM ION CONCENTRATION FROM](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-29.jpg)
p. H = - log [H 3 O+] CALCULATING THE HYDRONIUM ION CONCENTRATION FROM PH The hydronium ion concentration can be found from the p. H by the reverse of the mathematical operation employed to find the p. H. [H 3 O+] = 10 -p. H Example: What is the hydronium ion concentration in a solution that has a p. H of 8. 34? 8. 34 = - log [H 3 O+] - 8. 34 = log [H 3 O+]= 10 -8. 34 = 4. 57 x 10 -9 M

CALCULATING THE HYDRONIUM ION CONCENTRATION FROM PH The p. H of an unknown solution is 6. 35. What is the hydrogen-ion concentration? 6. 35 = - log [H 3 O+] - 6. 35 = log [H 3 O+]= 10 -6. 35 = 4. 5 x 10 -7 M
![Practice Problems Calculate the [H+] for each solution. A. p. H = 5. 00 Practice Problems Calculate the [H+] for each solution. A. p. H = 5. 00](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-31.jpg)
Practice Problems Calculate the [H+] for each solution. A. p. H = 5. 00 B. p. H = 12. 38 • When the p. H is a whole number, you can assume the coefficient to be 1 • When the p. H is a decimal, use the antilog. What are the hydrogen ion concentrations for solutions with the following p. H values? A. p. H = 4. 00 B. p. H = 11. 55

Relationship between p. H and p. OH. • p. H scale ranges from 014. • For aqueous solutions, the product of the hydrogen-ion concentration and the hydroxide-ion concentration equals 1. 0 +] + [OH−] = 1. 0 − 14 [H × 10− 14.

CALCULATING POH To calculate the p. OH of a solution you need to know the concentration of the hydroxide ion in moles per liter (molarity). The p. OH is then calculated using the expression: p. OH = - log [OH-] Example: What is the p. OH of a solution that has a hydroxide ion concentration of 4. 82 x 10 -5 M? p. OH = - log [4. 82 x 10 -5] = - ( - 4. 32) = 4. 32
![CALCULATING PH FROM [OH−] If you know the [OH−] of a solution, you can CALCULATING PH FROM [OH−] If you know the [OH−] of a solution, you can](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-34.jpg)
CALCULATING PH FROM [OH−] If you know the [OH−] of a solution, you can find its p. H. 1. You can use the ion-product constant to determine [H+] for a known [OH−]. 2. Then you use [H+] to calculate the p. H.
![PRACTICE PROBLEM What is the p. H of a solution if the [OH-] = PRACTICE PROBLEM What is the p. H of a solution if the [OH-] =](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-35.jpg)
PRACTICE PROBLEM What is the p. H of a solution if the [OH-] = 4. 0 x 10 -11 M? 1. Start with the ion-product constant to find [H+]. Kw + + − + 2. Rearrange the equation solve Kw = [OH ]to × [H ] for [H ] = [OH−] 3. [H+] = 1. 0 × 10− 14 4. 0 × 10− 11 = 0. 25 × 10− 3 M [H+] = 2. 5 × 10− 4 M 4. p. H = −log[H+] = −log(2. 5 × 10− 4) = −(− 3. 60205) p. H = 3. 60
![PRACTICE PROBLEMS 1. Calculate the p. H of each solution A. [OH-] = 4. PRACTICE PROBLEMS 1. Calculate the p. H of each solution A. [OH-] = 4.](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-36.jpg)
PRACTICE PROBLEMS 1. Calculate the p. H of each solution A. [OH-] = 4. 3 x 10 -5 M B. [OH-] = 4. 3 x 10 -11 M 2. Calculate the p. H of each solution A. [H+] = 5. 0 x 10 -5 M B. [H+] = 8. 3 x 10 -10 M


MORE PRACTICE PROBLEMS Find the p. H and the p. OH of each solution. A. [H+] = 1 x 10 -4 M B. [H+] = 0. 0015 M What are the p. H and p. OH values of the following solutions, based on their hydrogen-ion concentrations? A. [H+] = 1 x 10 -12 M B. [H+] = 0. 045 M Use either p. H Calculations worksheet #1 or #2 here

Complete Topic 8 Acids, Bases, and Salts note packet page 146, practice problems # 26 -29.

+ H RELATIVE CONCENTRATIONS OF SOLUTIONS The lower the p. H, the more H+ ions are in a soln. As [H+] in a soln. increases, p. H of the soln. decreases. Ex. A soln. with a p. H of 3 has more H+ ions then a soln. of p. H 4 Difference in H+ of two soln. = 10 (difference in p. H) That Means:


RELATIVE H+ CONCENTRATIONS OF SOLN. • Complete Topic 8 Acids, Bases, and Salts note packet page 146, practice problems # 30 -32.

STRONG VS WEAK ACIDS § Determined by degree in which the acid dissociates in water. ØStrong Acid – completely ionized in aqueous soln. + +(aq) + Cl–(aq) • High concentration of H O HCl(g) + H 2 O(l) → H O 3 3 Ex. ØWeak Acid – partially ionized in aqueous soln. • Low concentration of H 3 O+ Ex. CH 3 COOH(aq) + H 2 O(l) H 3 O+(aq) + CH 3 COO– (aq)


ACID DISSOCIATIONK a CONSTANT;
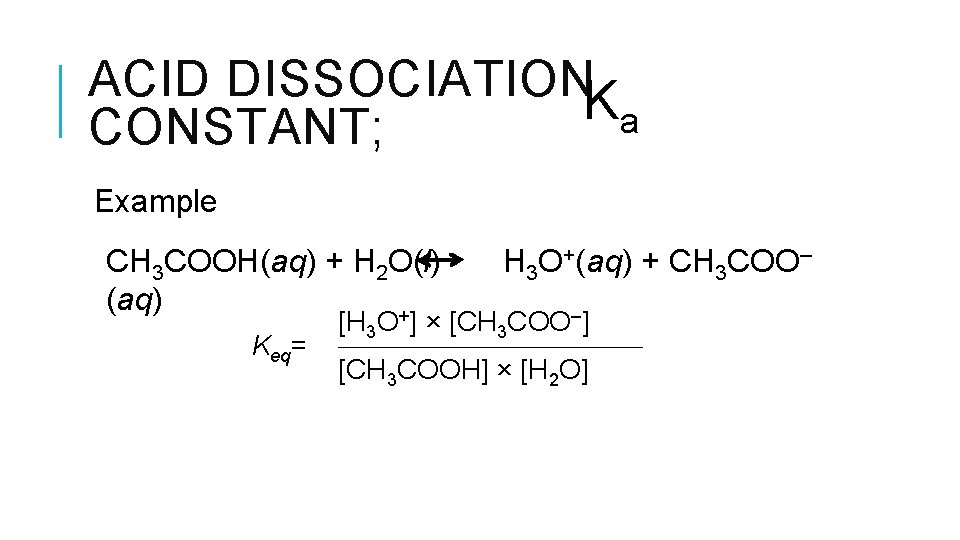
ACID DISSOCIATIONK a CONSTANT; Example CH 3 COOH(aq) + H 2 O(l) (aq) + Keq= H 3 O+(aq) + CH 3 COO– [H 3 O ] × [CH 3 COO–] [CH 3 COOH] × [H 2 O]

CALCULATING DISSOCIATION CONSTANTS You can find the Ka of an acid in water by substituting the equilibrium concentrations of the acid, [HA], the anion from the dissociation of the acid, [A−], and the hydrogen ion, [H+], into the equation below. [H+][A−] Ka = [HA]
![CALCULATING DISSOCIATION CONSTANTS In a 0. 1000 M solution of ethanoic acid, [H+] = CALCULATING DISSOCIATION CONSTANTS In a 0. 1000 M solution of ethanoic acid, [H+] =](http://slidetodoc.com/presentation_image_h2/9bb08934872582752a30a939323a6223/image-48.jpg)
CALCULATING DISSOCIATION CONSTANTS In a 0. 1000 M solution of ethanoic acid, [H+] = 1. 34 × 10− 3 M. Calculate the Ka of this acid. Refer to the table for the ionization equation for ethanoic acid. 1. Start by determining the equilibrium concentration of the ions. + − − 3 [H ] = [CH 3 COO ] = 1. 34 × 10 M (0. 1000 – 0. 00134)M = 0. 0987 M Concentration [CH 3 COOH] [H+] [CH 3 COO−] Initial 0. 1000 0 0 Change − 1. 34 × 10− 3 Equilibrium 0. 0987 1. 34 × 10− 3

STRONG VS WEAK BASES § Determined by degree in which the metal ions and hydroxide ions dissociate in water. ØStrong Base – completely ionized in aqueous soln. Na. OH(aq) + H 2 O(l) → OH-(aq) + Ex. Na+(aq) ØWeak Acid – partially ionized in aqueous soln. – + NH 3(aq) + H 2 O(l) NH 4 (aq) + OH (aq) Ex. Base Conjugate Aci d Acid Base

NEUTRALIZATION A rx between an acid and a base to produce water and a salt. Generally a double replacement reaction

TITRATION A lab process used for determining the concen. Of an unknown soln. by reacting it with a soln. of known concen. §Involves an acid and a base §When moles of H+ and OH- are equal, neutralization has occurred, and the endpoint of the titrations is reached. §Indicators are used to identify the endpoint. Change in indicator color = endpoint reached.

SALTS Ionic compounds composed of a positive ion (other than H+) and a negative ion (other than OH-) Salt is one of the products of an acid-base neutralization rx Salts are electrolytes (conduct electricity when dissolved in H 2 O) Soluble salts are better electrolytes than insoluble salts Table F can be used to determine soluble and insoluble salts

ELECTROLYTES Substances that can conduct electricity when dissolved in H 2 O Dissolve in H 2 O to produce a soln. with “+” and “–” ions. Conduct electricity b/c of mobile ions in the soln. Acids (Table K), bases (Table L), and salts are electrolytes
- Slides: 53