AcidBase Titrations Titrations n Titration Curve always calculate

Acid/Base Titrations

Titrations n Titration Curve – always calculate equivalent point first n Strong Acid/Strong Base n Regions that require “different” calculations n n B/F any base is added Half-way point region At the equivalence point After the equivalence point

Strong Acid/Strong Base n 50 m. L of 0. 02000 M KOH Titrated with 0. 1000 M HBr n First -find Volume at equivalence n n M 1 V 1 = M 2 V 2 (0. 050 L)(0. 02000 M) = 0. 1000 V V = 10. 0 m. L

Strong Acid/Strong Base n n 50. 00 m. L of 0. 02000 M KOH Titrated with 0. 1000 M HBr Second – find initial p. H = - log. AH ~ -log [H+] p. OH = -log. AOH ~ -log [OH-] p. H = 12. 30 n

Strong Acid/Strong Base n n n Before After 50 m. L of 0. 02000 M KOH Titrated with 0. 1000 M HBr (~6 ml) Third– find p. H at mid-way volume n KOH (aq) + HBr (aq) -> H 2 O (l) + KBr(aq) 0. 001000 mol 0. 000400 mol 0. 0006000 mol Limiting Reactant p. H = 11. 8

Strong Acid/Strong Base n 50 m. L of 0. 02000 M KOH Titrated with 0. 1000 M HBr n Fourth – find p. H at equivalence point n n Before After KOH (aq) + HBr (aq) -> H 2 O (l) + KBr(aq) 0. 001000 mol p. H = 7. 0 0. 0010000 mol

Strong Acid/Strong Base n 50 m. L of 0. 02000 M KOH Titrated with 0. 1000 M HBr n Finally – find p. H after equivalence point n n Before After 12 ml KOH (aq) + HBr (aq) -> H 2 O (l) + KBr(aq) 0. 001000 mol p. H = 2. 5 0. 001200 mol 0. 0002000 mol Limiting Reactant 0. 0010000 mol
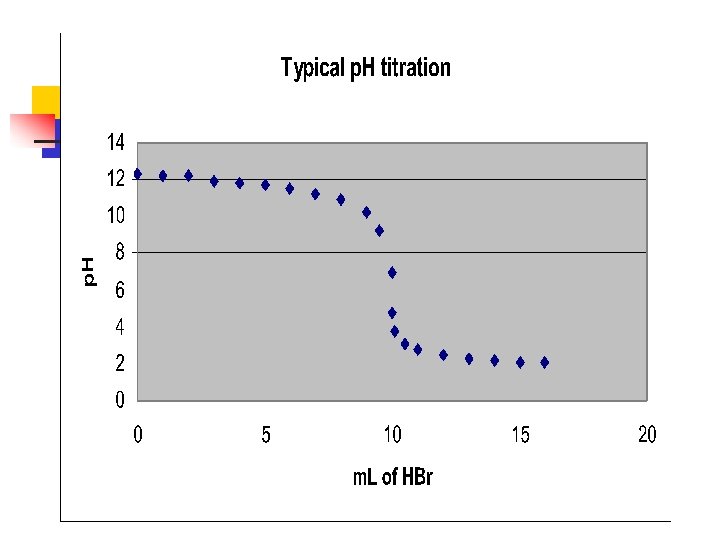


Titration of WEAK acid with a strong base
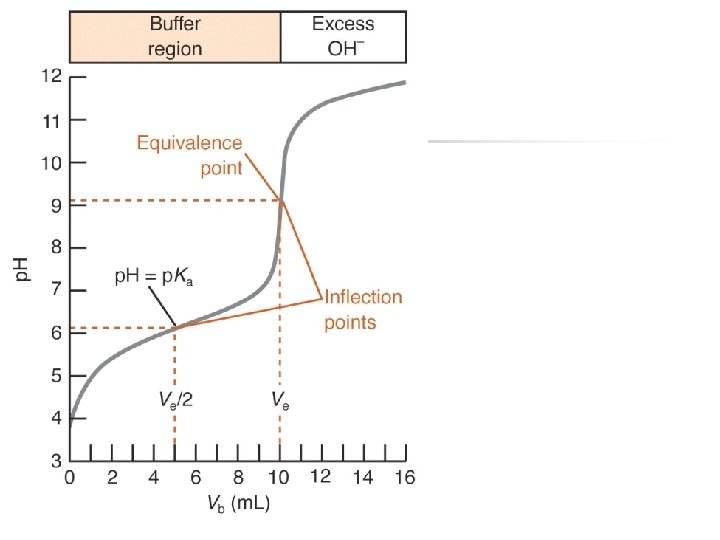

Titration of a weak acid solution with a strong base. n 25. 0 m. L of 0. 1000 M acetic acid Ka = 1. 8 x 10 -5 Titrant = 0. 100 M Na. OH n First, calculate the volume at the equivalence-point n n n M 1 V 1 = M 2 V 2 (0. 0250 L) 0. 1000 M = 0. 1000 M (V 2) V 2 = 0. 0250 L or 25. 0 m. L

Titration of a weak acid solution with a strong base. n n 25. 0 m. L of 0. 1000 M acetic acid Ka = 1. 8 x 10 -5 Titrant = 0. 100 M Na. OH Second, Calculate the initial p. H of the acetic acid solution

Titration of a weak acid solution with a strong base. n 25. 0 m. L of 0. 1000 M acetic acid Ka = 1. 8 x 10 -5 Titrant = 0. 100 M Na. OH n Third, Calculate the p. H at some intermediate volume n n

Titration of a weak acid solution with a strong base. n 25. 0 m. L of 0. 1000 M acetic acid Ka = 1. 8 x 10 -5 Titrant = 0. 100 M Na. OH n Fourth, Calculate the p. H at equivalence n n

Titration of a weak acid solution with a strong base. n 25. 0 m. L of 0. 1000 M acetic acid Ka = 1. 8 x 10 -5 Titrant = 0. 100 M Na. OH n Finally calculate the p. H after the addition 26. 0 m. L of Na. OH n n
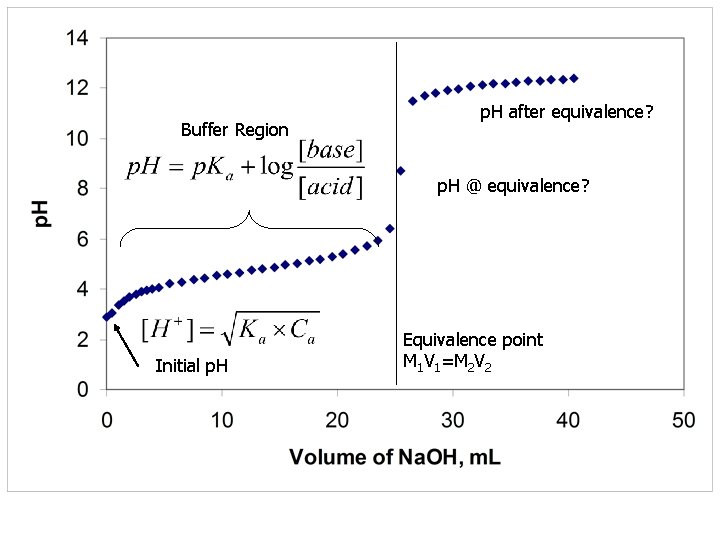
Buffer Region p. H after equivalence? p. H @ equivalence? Initial p. H Equivalence point M 1 V 1=M 2 V 2
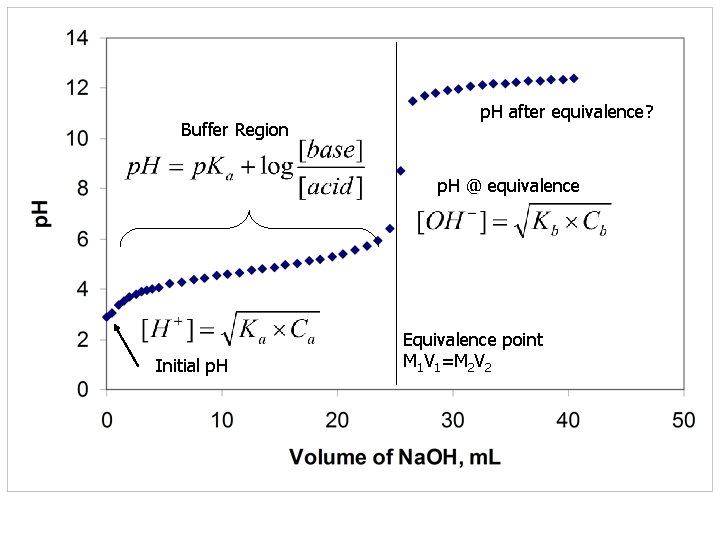
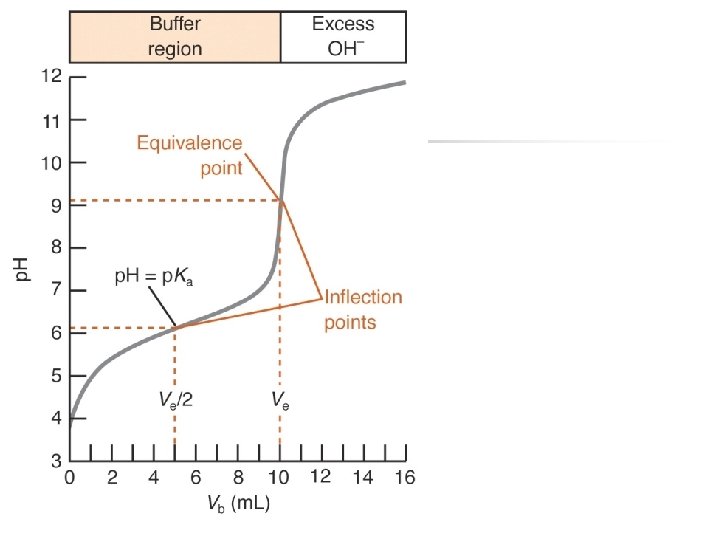
Buffer Region p. H after equivalence? p. H @ equivalence Initial p. H Equivalence point M 1 V 1=M 2 V 2
![Buffer Region p. H after equivalence Dominated by remaining [OH-] p. H @ equivalence Buffer Region p. H after equivalence Dominated by remaining [OH-] p. H @ equivalence](http://slidetodoc.com/presentation_image_h/eaa7bdba56887df0e9ef6c0868ad52f0/image-20.jpg)
Buffer Region p. H after equivalence Dominated by remaining [OH-] p. H @ equivalence Initial p. H Equivalence point M 1 V 1=M 2 V 2
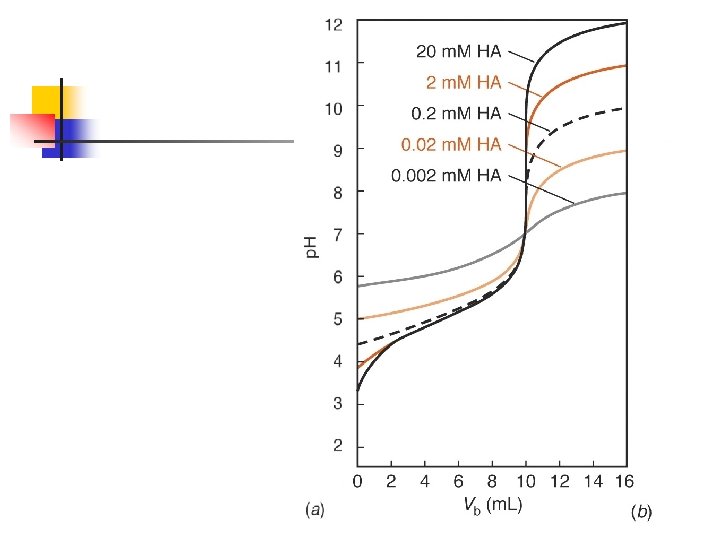

Weak Base titrated with strong acid n n Consider a 100 ml of a 0. 0100 M base with 0. 0500 M HCl Kb = 1 x 10 -5
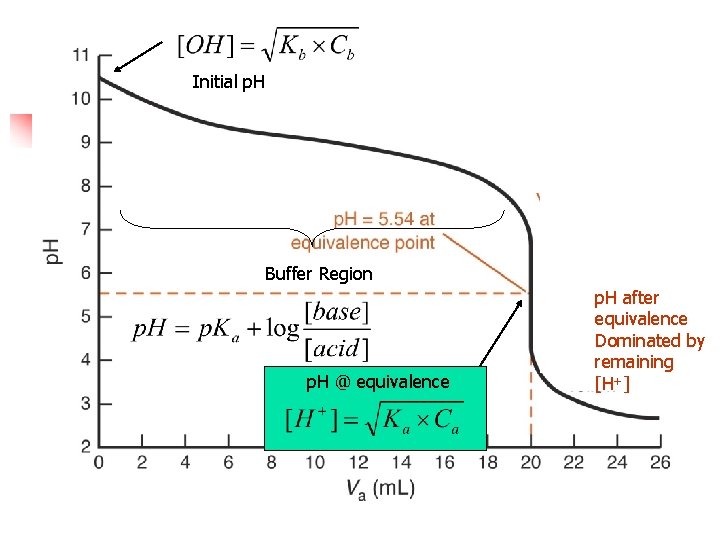
Initial p. H Buffer Region p. H @ equivalence p. H after equivalence Dominated by remaining [H+]
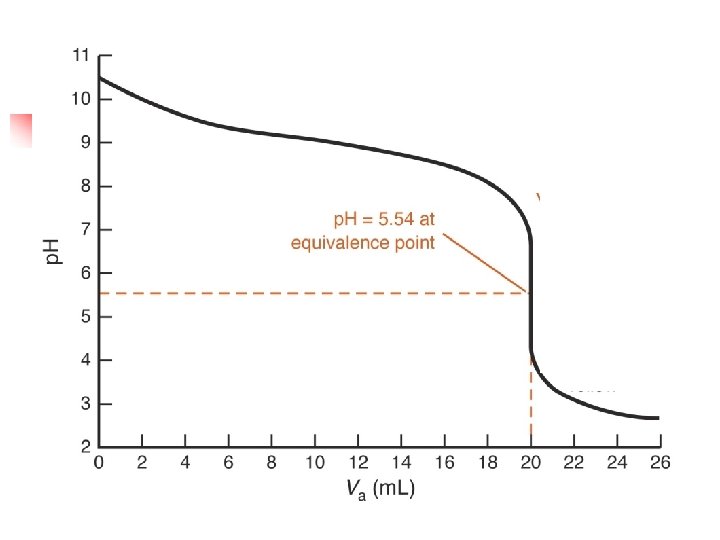
- Slides: 24