AcidBase Titrations Introduction 3 Overview Titrations are Important
Acid-Base Titrations Introduction 3. ) Overview Ø Ø Ø Titrations are Important tools in providing quantitative and qualitative data for a sample. To best understand titrations and the information they provide, it is necessary to understand what gives rise to the shape of a typical titration curve. To do this, acid-base equilibria are used to predict titration curve shapes. p. Ka of His in the His-Asp catalytic dyad that catalyzes the oxidation of glucose 6 -phosphate Biochemistry, Vol. 41, No. 22, 2002 6945
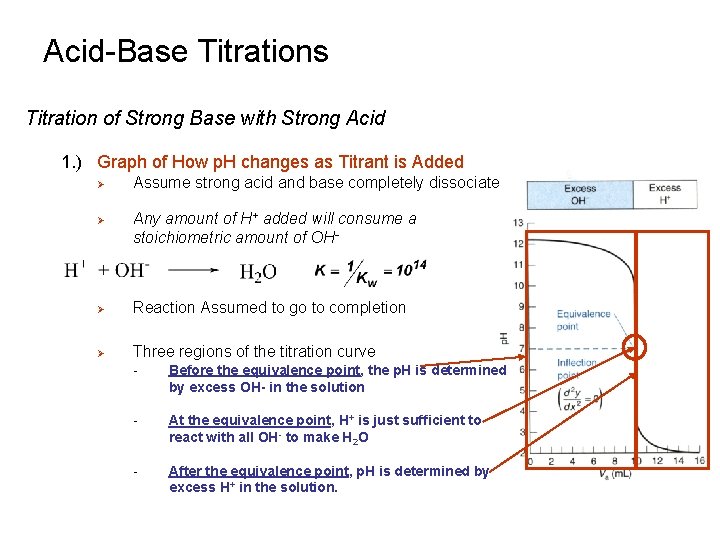
Acid-Base Titrations Titration of Strong Base with Strong Acid 1. ) Graph of How p. H changes as Titrant is Added Ø Ø Assume strong acid and base completely dissociate Any amount of H+ added will consume a stoichiometric amount of OH- Ø Reaction Assumed to go to completion Ø Three regions of the titration curve - Before the equivalence point, the p. H is determined by excess OH- in the solution - At the equivalence point, H+ is just sufficient to react with all OH- to make H 2 O - After the equivalence point, p. H is determined by excess H+ in the solution.

Acid-Base Titrations Titration of Strong Base with Strong Acid 1. ) Graph of How p. H changes as Titrant is Added Ø Ø Remember, equivalence point is the ideal goal Actually measure End Point - Ø Different Regions require different kinds of calculations - Ø Marked by a sudden physical change: color, potential Illustrated examples The “true” titration reaction is: Titrant Analyte

Acid-Base Titrations Titration of Strong Base with Strong Acid 2. ) Volume Needed to Reach the Equivalence Point Ø Titration curve for 50. 00 m. L of 0. 02000 M KOH with 0. 1000 M HBr Ø At equivalence point, amount of H+ added will equal initial amount of OH- mmol of HBr at equivalence point mmol of OHbeing titrated When 10. 00 m. L of HBr has been added, the titration is complete. Prior to this point, there is excess OH- present. After this point there is excess H+ present.

Acid-Base Titrations Titration of Strong Base with Strong Acid 3. ) Before the Equivalence Point Ø Titration curve for 50. 00 m. L of 0. 02000 M KOH with 0. 1000 M HBr - Equivalence point (Ve) when 10. 00 m. L of HBr has been added When 3. 00 m. L of HBr has been added, reaction is 3/10 complete Calculate Remaining [OH-]: Initial volume of OH- Fraction of OH Initial concentration Dilution Factor Remaining of OH- Calculate [H+] and p. H: Total volume

Acid-Base Titrations Titration of Strong Base with Strong Acid 4. ) At the Equivalence Point Ø Titration curve for 50. 00 m. L of 0. 02000 M KOH with 0. 1000 M HBr - Just enough H+ has been added to consume OHp. H determined by dissociation of water Kw x - x Kw= 1 x 10 -14 p. H at the equivalence point for any strong acid with strong base is 7. 00 Not true for weak acid-base titration

Acid-Base Titrations Titration of Strong Base with Strong Acid 5. ) After the Equivalence Point Ø Titration curve for 50. 00 m. L of 0. 02000 M KOH with 0. 1000 M HBr - Adding excess HBr solution When 10. 50 m. L of HBr is added Calculate volume of excess H+: Calculate excess [H+]: Calculate p. H: Volume of excess H+ Initial concentration Dilution factor of H+ Total volume
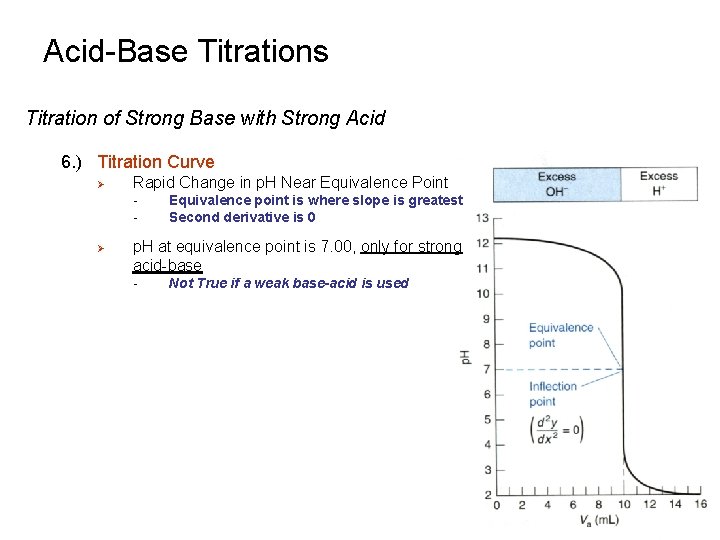
Acid-Base Titrations Titration of Strong Base with Strong Acid 6. ) Titration Curve Ø Rapid Change in p. H Near Equivalence Point - Ø Equivalence point is where slope is greatest Second derivative is 0 p. H at equivalence point is 7. 00, only for strong acid-base - Not True if a weak base-acid is used

Acid-Base Titrations Titration of Weak Acid with Strong Base 1. ) Four Regions to Titration Curve Ø Before any added base, just weak acid (HA) in water - p. H determined by Ka Ka Ø With addition of strong base buffer - Ø p. H determined by Henderson Hasselbach equation At equivalence point, all HA is converted into A- Weak base with p. H determined by Kb Kb

Acid-Base Titrations Titration of Weak Acid with Strong Base 1. ) Four Regions to Titration Curve Ø Beyond equivalence point, excess strong base is added to A- solution - p. H is determined by strong base Similar to titration of strong acid with strong base 2. ) Illustrated Example: Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH - MES is a weak acid with p. Ka = 6. 27 K - Reaction goes to completion with addition of strong base

Acid-Base Titrations Titration of Weak Acid with Strong Base 3. ) Volume Needed to Reach the Equivalence Point Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH - Reaction goes to completion with addition of strong base Strong plus weak react completely mmol of base mmol of HA

Acid-Base Titrations Titration of Weak Acid with Strong Base 4. ) Region 1: Before Base is Added Ø Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Simply a weak-acid problem Ka Calculate [H+]: F-x Calculate p. H: Ka= 10 -6. 27 x x

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 2: Before the Equivalence Point Ø Ø Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Adding OH- creates a mixture of HA and A- Buffer Calculate p. H from [A-]/[HA] using Henderson-Hasselbach equation Simply the difference Simply of volumes of initialratio quantities Calculate [A-]/HA]: Amount of added Na. OH is 3 m. L with equivalence point is 10 m. L Relative Initial quantities (HA≡ 1) Relative Final quantities Calculate p. H: 1 - -

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 2: Before the Equivalence Point Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Ø p. H = p. Ka when the volume of titrant equals ½Ve Relative Initial quantities (HA≡ 1) Relative Final quantities 1 - -

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 3: At the Equivalence Point Ø Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Exactly enough Na. OH to consume HA Relative Initial quantities (HA≡ 1) 1 1 - - Relative Final quantities - - 1 1 Ø The solution only contains A- weak base Kb F-x x x

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 3: At the Equivalence Point Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Calculate Formal concentration of [A-]: A- is no longer 0. 02000 M, diluted by the addition of Na. OH Initial volume of HA Initial concentration Dilution factor of HA Total volume

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 3: At the Equivalence Point Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Calculate [OH-]: Calculate p. H: p. H at equivalence point is not 7. 00 p. H will always be above 7. 00 for titration of a weak acid because acid is converted into conjugate base at the equivalence point

Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Region 4: After the Equivalence Point Ø Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Adding Na. OH to a solution of A- Na. OH is a much stronger base than Ap. H determined by excess of OH- Calculate volume of excess OH-: Amount of added Na. OH is 10. 10 m. L with equivalence point is 10 m. L Calculate excess Calculate p. H: [OH-]: Volume of excess OH- Initial concentration Dilution factor of OH- Total volume
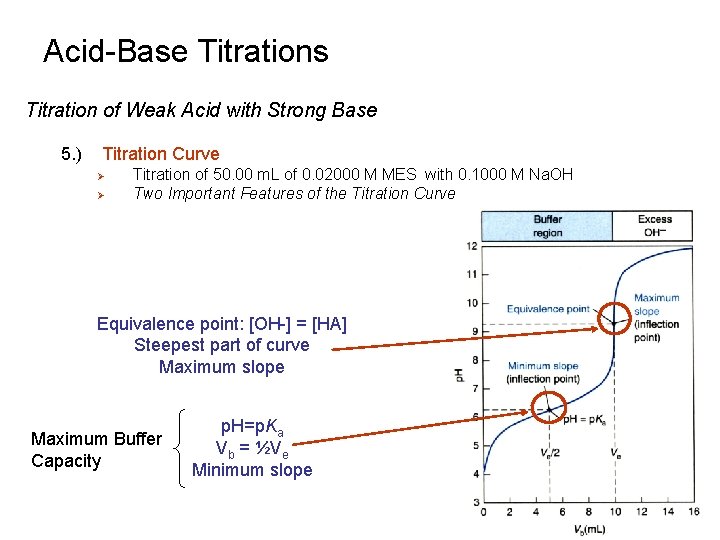
Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Titration Curve Ø Ø Titration of 50. 00 m. L of 0. 02000 M MES with 0. 1000 M Na. OH Two Important Features of the Titration Curve Equivalence point: [OH-] = [HA] Steepest part of curve Maximum slope Maximum Buffer Capacity p. H=p. Ka Vb = ½Ve Minimum slope
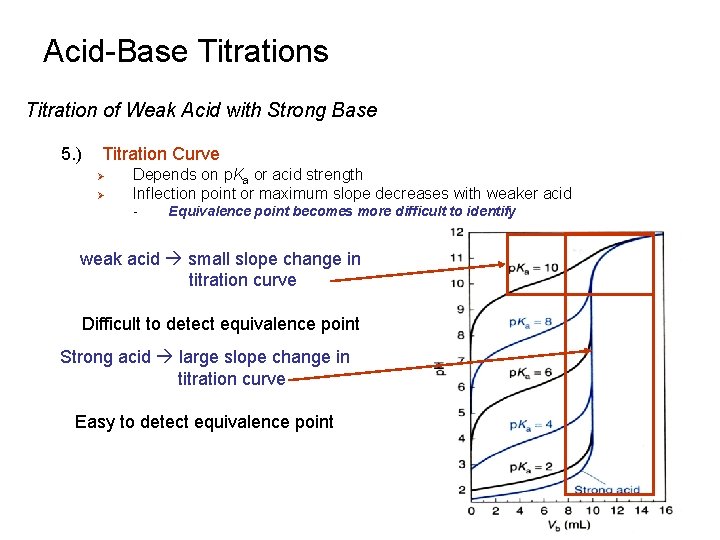
Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Titration Curve Ø Ø Depends on p. Ka or acid strength Inflection point or maximum slope decreases with weaker acid - Equivalence point becomes more difficult to identify weak acid small slope change in titration curve Difficult to detect equivalence point Strong acid large slope change in titration curve Easy to detect equivalence point
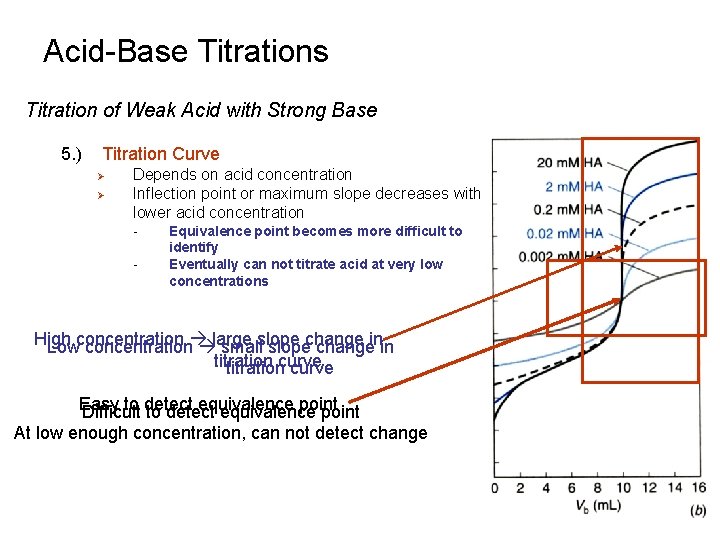
Acid-Base Titrations Titration of Weak Acid with Strong Base 5. ) Titration Curve Ø Ø Depends on acid concentration Inflection point or maximum slope decreases with lower acid concentration - Equivalence point becomes more difficult to identify Eventually can not titrate acid at very low concentrations High Lowconcentration large smallslopechangeinin titrationcurve Easy to detect equivalence point Difficult to detect equivalence point At low enough concentration, can not detect change
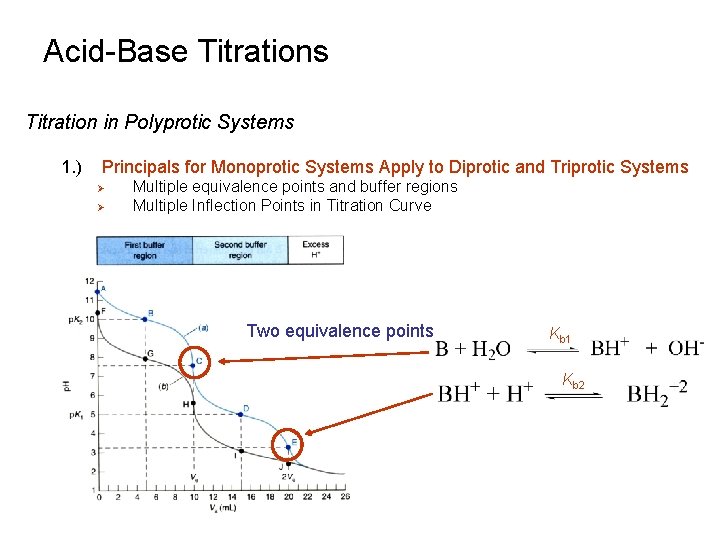
Acid-Base Titrations Titration in Polyprotic Systems 1. ) Principals for Monoprotic Systems Apply to Diprotic and Triprotic Systems Ø Ø Multiple equivalence points and buffer regions Multiple Inflection Points in Titration Curve Two equivalence points Kb 1 Kb 2
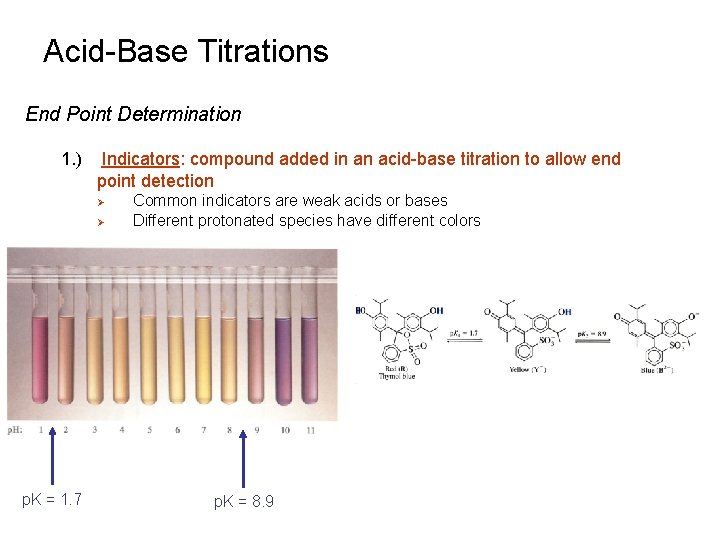
Acid-Base Titrations End Point Determination 1. ) Indicators: compound added in an acid-base titration to allow end point detection Ø Ø p. K = 1. 7 Common indicators are weak acids or bases Different protonated species have different colors p. K = 8. 9
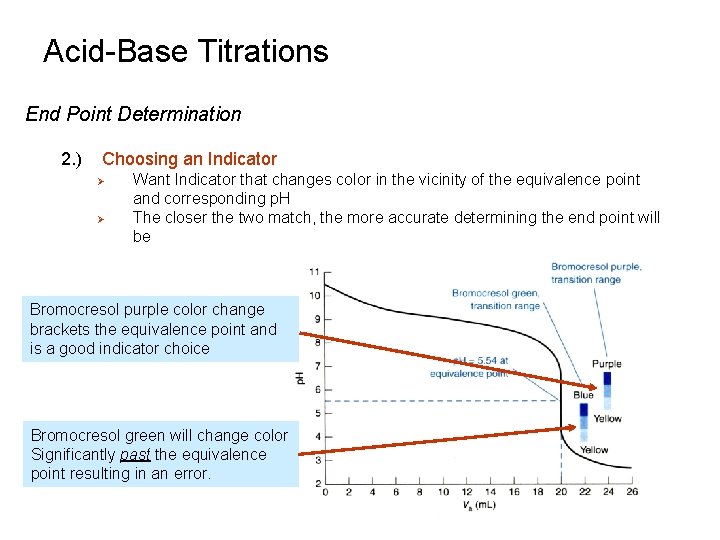
Acid-Base Titrations End Point Determination 2. ) Choosing an Indicator Ø Ø Want Indicator that changes color in the vicinity of the equivalence point and corresponding p. H The closer the two match, the more accurate determining the end point will be Bromocresol purple color change brackets the equivalence point and is a good indicator choice Bromocresol green will change color Significantly past the equivalence point resulting in an error.

Acid-Base Titrations End Point Determination 2. ) Choosing an Indicator The difference between the end point (point of detected color change) and the true equivalence point is the indicator error Amount of indicator added should be negligible Indicators cover a range of p. Hs

Acid-Base Titrations End Point Determination 3. ) Example: a) What is the p. H at the equivalence point when 0. 100 M hydroxyacetic acid is titrated with 0. 0500 M KOH? b) What indicator would be a good choice to monitor the endpoint?
- Slides: 26