AcidBase Titration Technique used to determine the concentration

Acid/Base Titration • Technique used to determine the concentration of an acid or base by comparison with a standard. • A neutralization reaction is carried out. • Rely on color change to signal end of reaction.

Titration: Monoprotic Acid + Monohydroxy Base • HA + MOH MA + H 2 O • Use the relationship in the balanced equation to find the unknown concentration. • At the end-point, # moles HA = # moles MOH

How to calculate # of moles? • Remember Molarity! • Molarity = # moles solute Liters of solution • Rearranging: # moles solute = Molarity X Liters of solution

Acid-Base Titration • Carefully controlled neutralization reaction. • Requires a standard solution and an acid-base indicator • Standard solution has an acid or base of known concentration.

Titration • Standard solution is slowly added to unknown solution. • As the solutions mix, a neutralization reaction occurs. • Eventually, enough standard solution is added to neutralize the unknown solution. Equivalence point Simple Titration Animation

Equivalence point • Total number of moles of H+ ions donated by acid = total + number of moles of H accepted by base. • Total moles H+ = total moles OH-

Titration • End-point = point at which indicator changes color. • If the indicator is chosen correctly, the end-point is very close to the equivalence point.
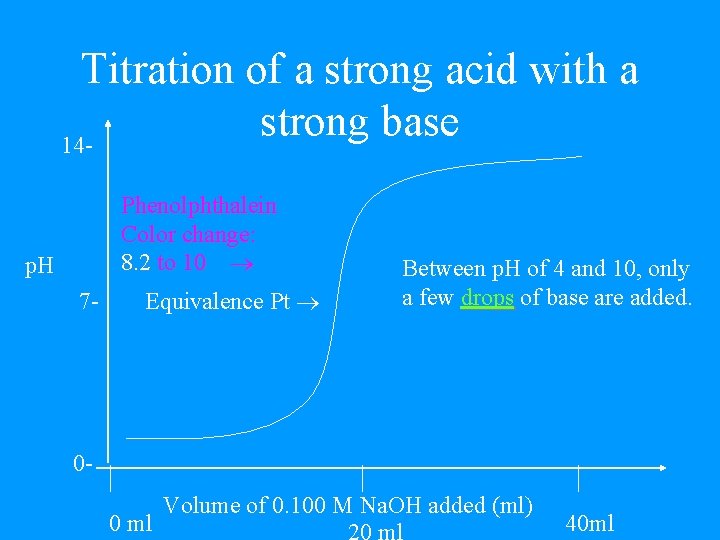
Titration of a strong acid with a strong base 14 Phenolphthalein Color change: 8. 2 to 10 p. H Equivalence Pt 7 - 0 - 0 ml Between p. H of 4 and 10, only a few drops of base are added. Volume of 0. 100 M Na. OH added (ml) 40 ml

MH+VH+ = MOH-VOH • • • MH+ = molarity of H+ MOH- = molarity of OHVH+ = volume of H+ VOH- = volume of OHIf you know 3, you can find the 4 th.

Ma. Va = Mb. Vb • True for monoprotic acids and monohydroxy bases.

Titration Problem #1 • In a titration of 40. 0 m. L of a nitric acid solution, the end point is reached when 35. 0 m. L of 0. 100 M Na. OH is added. Calculate the concentration of the nitric acid solution.

Neutralization Reaction • HNO 3 + Na. OH H 2 O + Na. NO 3 • 1 H in acid & 1 OH in base. • M a V a = M b. V b

Variables • Ma = ? • Va = 40. 0 m. L • Mb = 0. 100 M • Vb = 35. 0 m. L

Plug and Chug • X (40. 0 m. L) = (0. 100 M )(35. 0 m. L) • X =. 0875 M

Titration Problem #2 • What is the concentration of a hydrochloric acid solution if 50. 0 m. L of a 0. 250 M KOH solution are needed to neutralize 20. 0 m. L of the HCl solution of unknown concentration?

Neutralization Reaction • KOH + HCl H 2 O + KCl • 1 H in acid & 1 OH in base • M a V a = M b. V b

Variables • Ma = X • Va = 20. 0 m. L • Mb = 0. 250 M • Vb = 50. 0 m. L

Plug and Chug • X (20. 0 m. L) = (0. 250 M) (50. 0 m. L) • X = 0. 625 M

Titration Problem #3 • What is the concentration of a sulfuric acid solution if 50. m. L of a 0. 25 M Ba(OH)2 solution are needed to neutralize 20 m. L of the H 2 SO 4 solution of unknown concentration?

Neutralization Reaction • H 2 SO 4 + Ba(OH)2 2 H 2 O + Ba. SO 4 • • H 2 SO 4 is a strong diprotic acid. Ba(OH)2 is a strong dihydroxy base. # of H’s in acid = # of OH’s in base. M a V a = M b. V b

Variables • Ma = X • Va = 20. m. L • Mb = 0. 25 M • Vb = 50. m. L

Plug and Chug • X (20. ) = (0. 25 M) (50. m. L) • X = 0. 625 M = MH+ • To calculate the Molarity of the H 2 SO 4, adjust for the fact that the acid is diprotic.
![[H+] vs. [H 2 SO 4] • H 2 SO 4 + • Molarity [H+] vs. [H 2 SO 4] • H 2 SO 4 + • Molarity](http://slidetodoc.com/presentation_image_h/ef87ea68ba8f21bd315686707078b6dc/image-23.jpg)
[H+] vs. [H 2 SO 4] • H 2 SO 4 + • Molarity of H 2 SO 4 = ½ [H+] + 2 H 2 SO 4 • ½ (0. 625 M) = 0. 31 M H 2 SO 4

Other kinds of Titrations • Titrations based on redox reactions are also used. • Same ideas: rely on color change to signal completion of reaction. • Use stoichiometric equalities to determine concentration of unknown. Redox titration Animation

Salts • A salt is an ionic compound containing positive ions other than H+ and negative ions other than OH • Hydrolysis is the opposite of neutralization. – Literally means “water splitting. ” – A reaction between water and the ions of a dissolved salt. • When a salt undergoes hydrolysis, it reacts with water to form an acid and a base. Acid + Base Salt + Water

Hydrolysis • To predict which solution occurs when salts react with water, i. e. , neutral, acidic or basic: – Identify the cation and anion present in the salt – Determine if from a strong or a weak acid/base • General Rule: strong acid + strong base neutral salt strong acid + weak base acidic salt weak acid + strong base basic salt

Buffers • Buffers are solutions that resist changes in p. H when limited amounts of acid or base are added. • A buffer is a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. • The mixture of ions and molecules resist changes in p. H by reacting with any H+ or OH- added to the solution.
- Slides: 27