ACIDBASE REACTIONS Monday 42114 ACIDBASE REACTIONS When an

ACID-BASE REACTIONS Monday, 4/21/14

ACID-BASE REACTIONS When an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt and water. The H+ cation of the acid combines with the OHanion of the base to form water. The compound formed by the remaining ions is called a salt. Acid + Base Water + Salt Ex: HCl + Na. OH H 2 O + Na. Cl
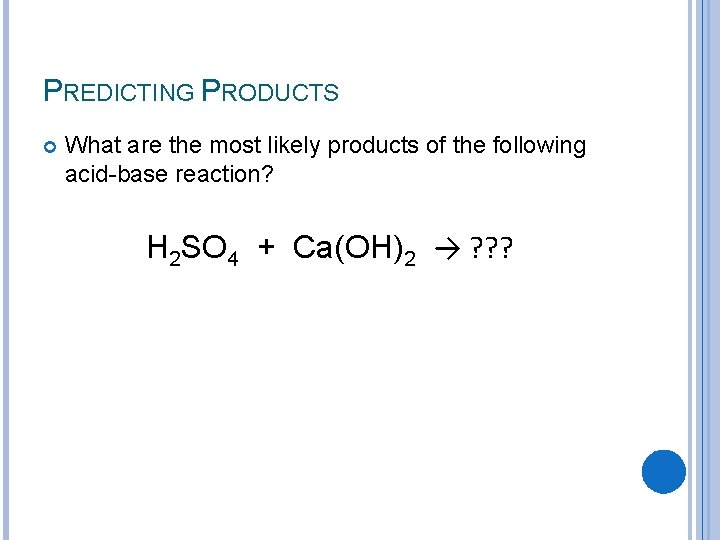
PREDICTING PRODUCTS What are the most likely products of the following acid-base reaction? H 2 SO 4 + Ca(OH)2 → ? ? ?

PREDICTING PRODUCTS What are the most likely products of the following acid-base reaction? H 2 SO 4 + Ca(OH)2 → H 2 O + Ca. SO 4 Check: Do the charges on the salt =0 (remember criss-cross)? H 2 SO 4 + Ca(OH)2 → H 2 O + Ca. SO 4

NEUTRALIZATION VS. BUFFERS

TYPES OF ACIDS AND BASES Strong Acids: completely dissociate (break apart) in solution to form H+ ions. Strong Bases: completely dissociate in solution to form OH- ions. Weak Acids: Only partially dissociate; some H+ ions stay bonded to the rest of the acid molecule. Weak Bases: Only partially dissociate; some OHions stay bonded to the rest of the base molecule.

STRONG ACIDS AND STRONG BASES Both fully ionize (break apart) The reaction leads to a p. H=7 solution, with only the water and salt remaining.

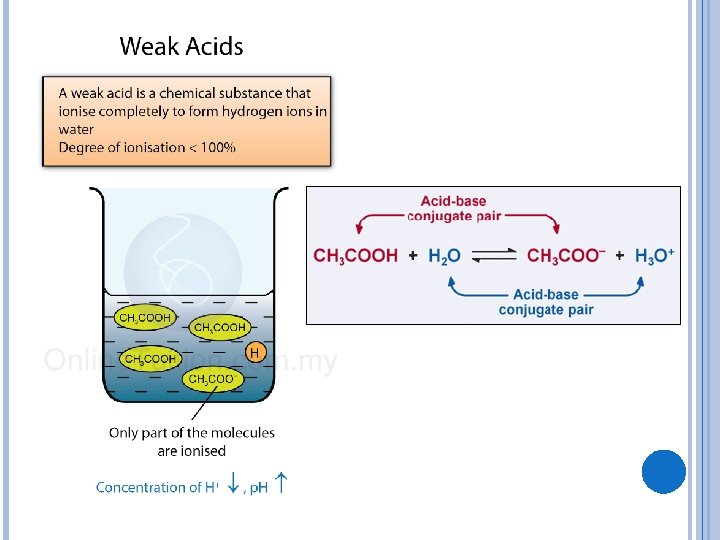
WEAK ACIDS AND WEAK BASES These do not fully break apart There is more than just water and salt remaining! � Weak Acid: HA, H+, A- Example: HCN leads to HCN, H+, and CN� Weak Base: BOH, OH-, B+ Example: NH 4 OH leads to NH 4 OH, NH 4+, and OH-

BUFFER REACTIONS Having the extra weak acid and weak base present means that the solution can still neutralize additional acids or bases. This allows the solution to RESIST a change in p. H. Left over weak acid and base neutralizes whatever else is added in! This is called a “buffer. ”
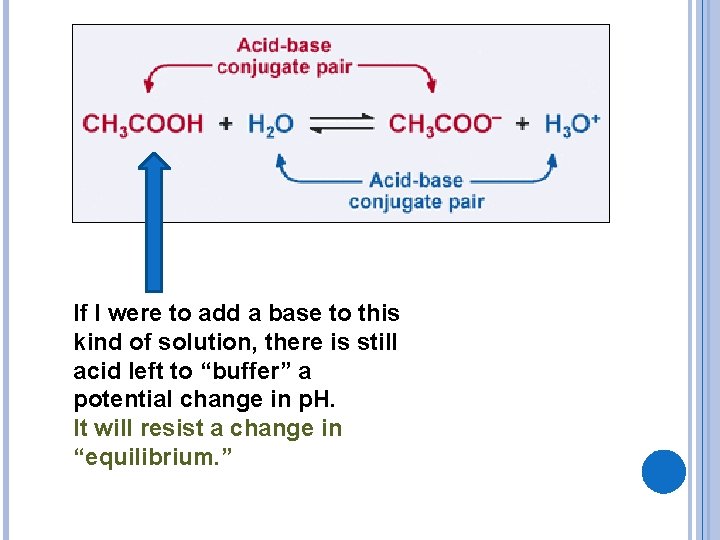
If I were to add a base to this kind of solution, there is still acid left to “buffer” a potential change in p. H. It will resist a change in “equilibrium. ”

YOUR JOB Complete the Neutralization and Buffer Practice for tomorrow! We will begin our unit project on Wednesday. (It will be partner/small group)
- Slides: 12