AcidBase Reactions Importance of AcidBase Reactions Acidbase chemistry
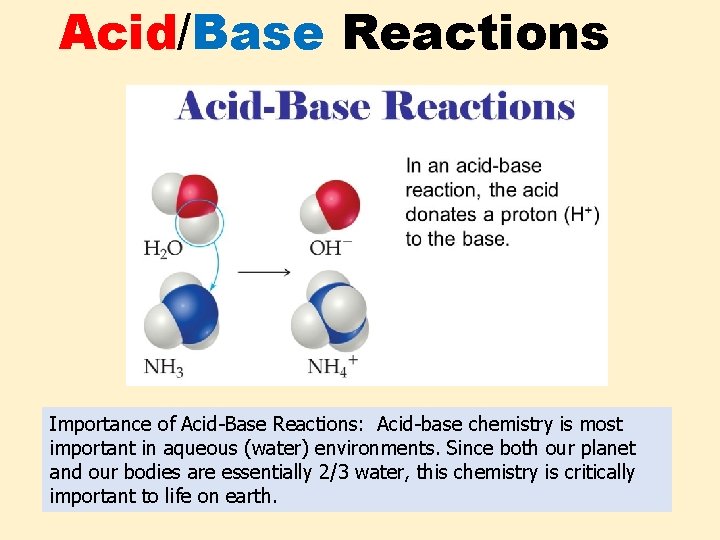
Acid/Base Reactions Importance of Acid-Base Reactions: Acid-base chemistry is most important in aqueous (water) environments. Since both our planet and our bodies are essentially 2/3 water, this chemistry is critically important to life on earth.
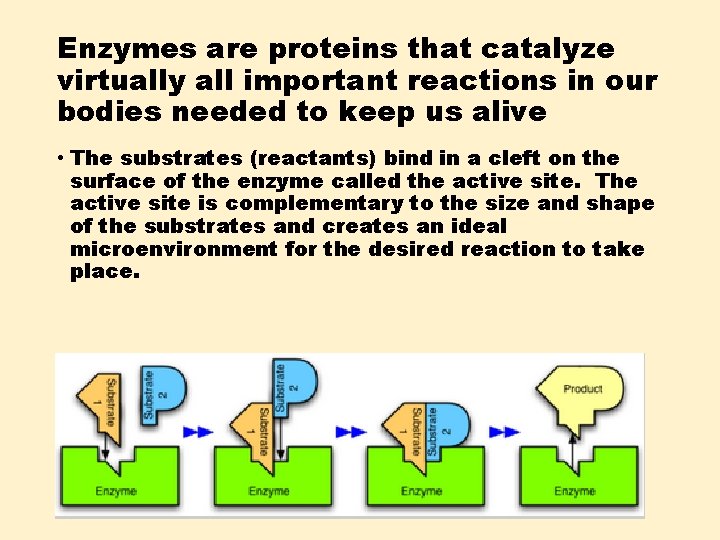
Enzymes are proteins that catalyze virtually all important reactions in our bodies needed to keep us alive • The substrates (reactants) bind in a cleft on the surface of the enzyme called the active site. The active site is complementary to the size and shape of the substrates and creates an ideal microenvironment for the desired reaction to take place.

Proteins can be unfolded by changes in the amount of acid and base present in the environment • When the shape of a protein changes, it can no longer catalyze the chemical reactions in our cells needed to keep us alive. Very acidic or basic solution
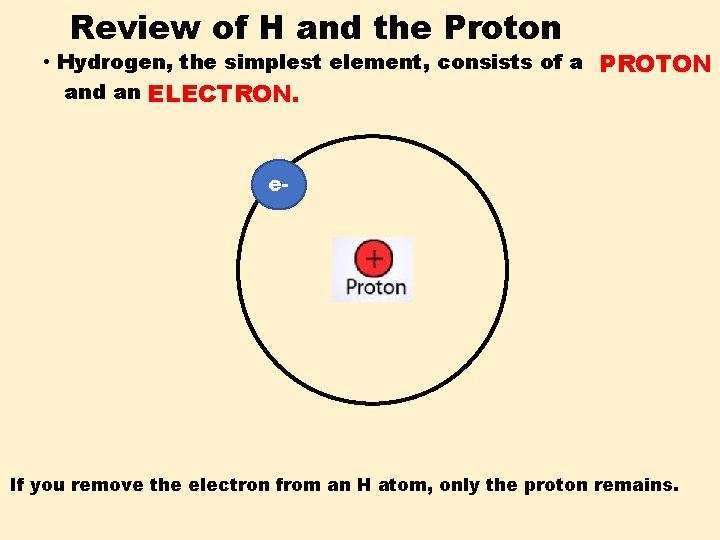
Review of H and the Proton • Hydrogen, the simplest element, consists of a PROTON and an ELECTRON. e- If you remove the electron from an H atom, only the proton remains.
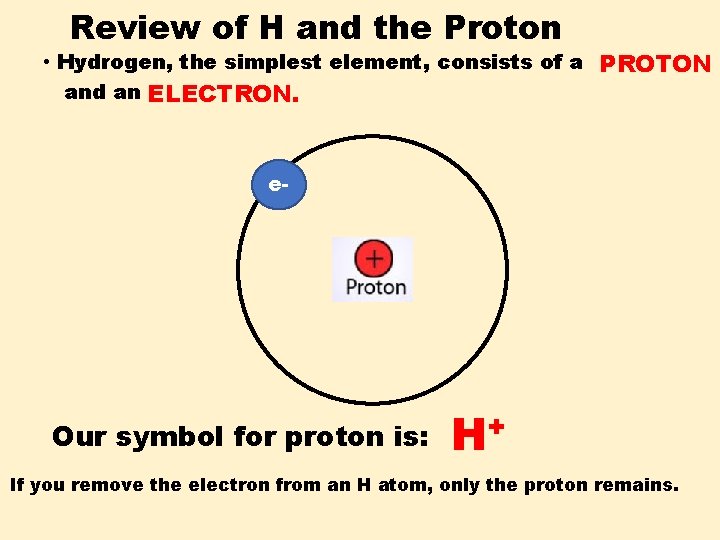
Review of H and the Proton • Hydrogen, the simplest element, consists of a PROTON and an ELECTRON. e- Our symbol for proton is: H+ If you remove the electron from an H atom, only the proton remains.
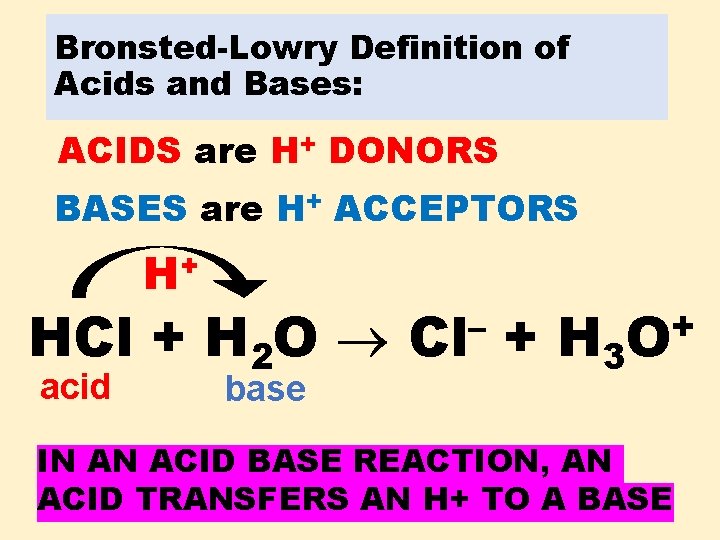
Bronsted-Lowry Definition of Acids and Bases: ACIDS are H+ DONORS BASES are H+ ACCEPTORS + H HCl + H 2 O acid base – Cl + H 3 + O IN AN ACID BASE REACTION, AN ACID TRANSFERS AN H+ TO A BASE
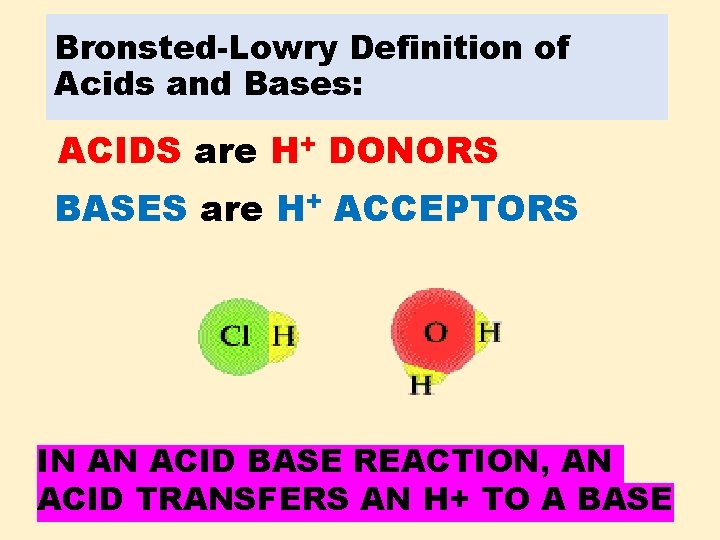
Bronsted-Lowry Definition of Acids and Bases: ACIDS are H+ DONORS BASES are H+ ACCEPTORS IN AN ACID BASE REACTION, AN ACID TRANSFERS AN H+ TO A BASE

Properties of Acids and Bases ACIDS • Taste: Sour • Feel: Stings Wine, vinegar, aspirin lemons, limes BASES • Taste: Bitter • Feel: Slippery Drano, Baking Soda, Window, Cleaners
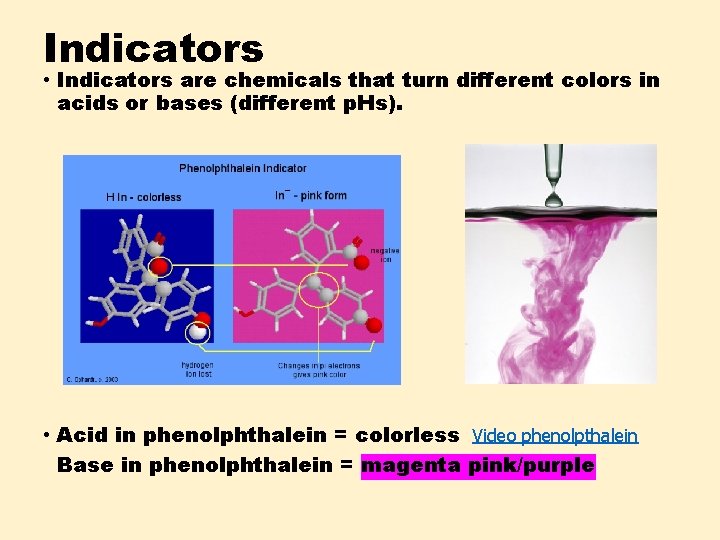
Indicators • Indicators are chemicals that turn different colors in acids or bases (different p. Hs). • Acid in phenolphthalein = colorless Video phenolpthalein Base in phenolphthalein = magenta pink/purple

Properties continued • Acids always react with active metals. nitric HCl Bases generally do not react with metals. Exception: Al + Na. OH

Sulfuric Acid oxidizing sugar H 2 SO 4 with Sugar Video
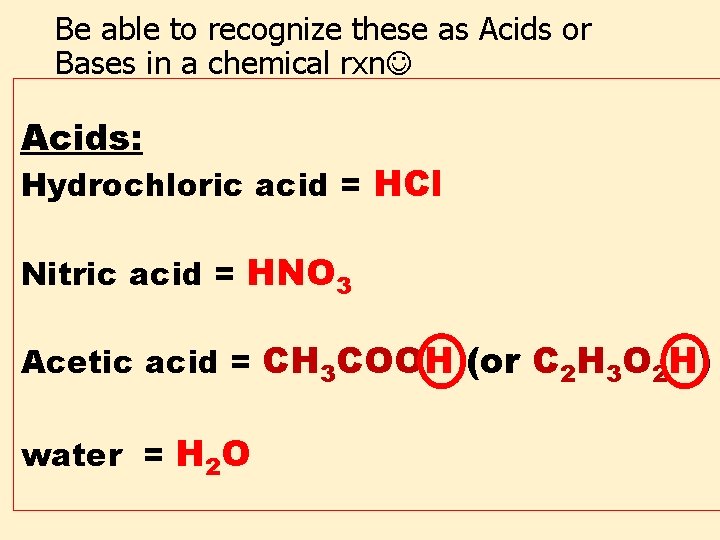
Be able to recognize these as Acids or Bases in a chemical rxn Acids: Hydrochloric acid = HCl Nitric acid = HNO 3 Acetic acid = CH 3 COOH (or C 2 H 3 O 2 H) water = H 2 O

Be able to recognize these as Acids or Bases in a chemical rxn • Bases: Ammonia (covalent) = NH 3 Na. OH Ions: Na+1 + OH-1 Sodium hydroxide (ionic) = KOH + OH-1 Potassium hydroxide (ionic) = Ions: K+1 Water = H 2 O

Patterns: Acid-Base Conjugate Pairs HA → H+ + AAcid conjugate base of HA HA and A- are conjugate pairs because they differ by an H+ B- + H + Base → HB conjugate acid of B- B- and HB are conjugate pairs because they differ by an H+
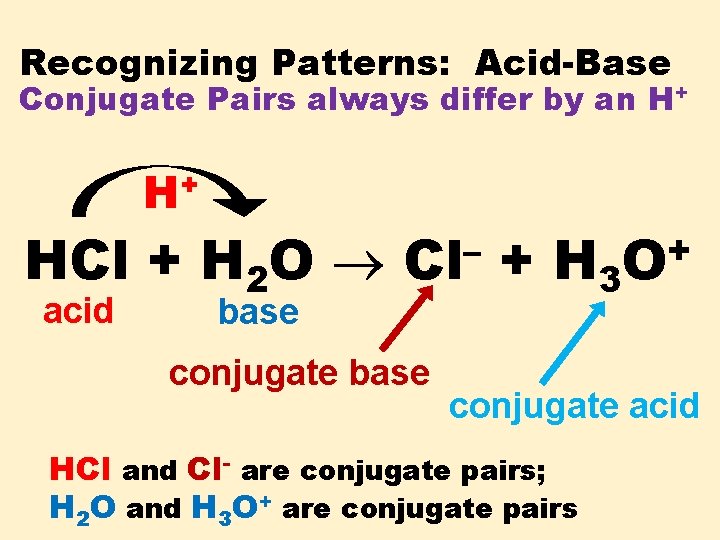
Recognizing Patterns: Acid-Base Conjugate Pairs always differ by an H+ + H HCl + H 2 O acid base – Cl conjugate base + H 3 + O conjugate acid HCl and Cl- are conjugate pairs; H 2 O and H 3 O+ are conjugate pairs
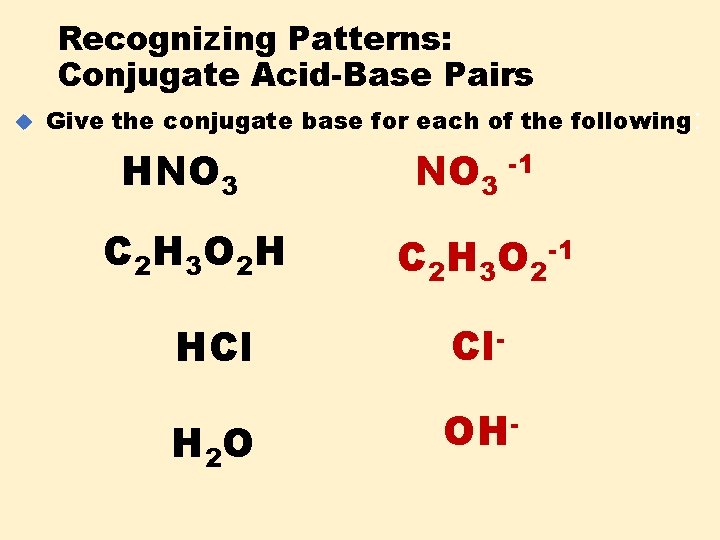
Recognizing Patterns: Conjugate Acid-Base Pairs u Give the conjugate base for each of the following : HNO 3 C 2 H 3 O 2 H NO 3 -1 C 2 H 3 O 2 -1 HCl Cl- H 2 O OH-
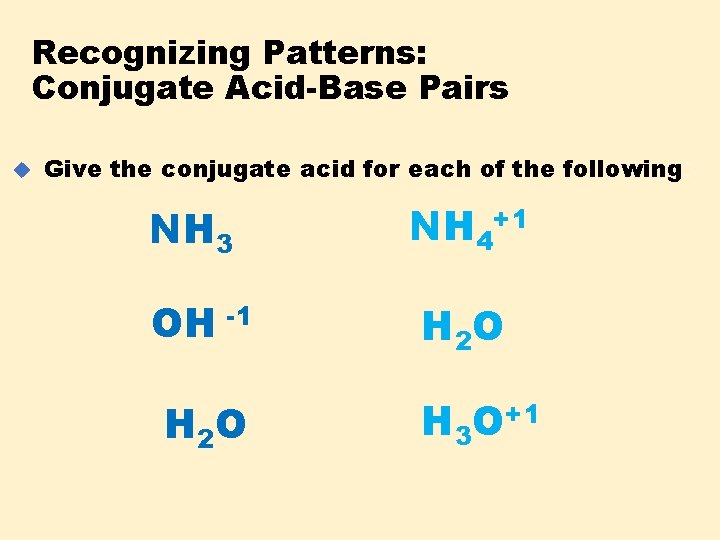
Recognizing Patterns: Conjugate Acid-Base Pairs u Give the conjugate acid for each of the following : NH 3 NH 4+1 OH -1 H 2 O H 3 O+1
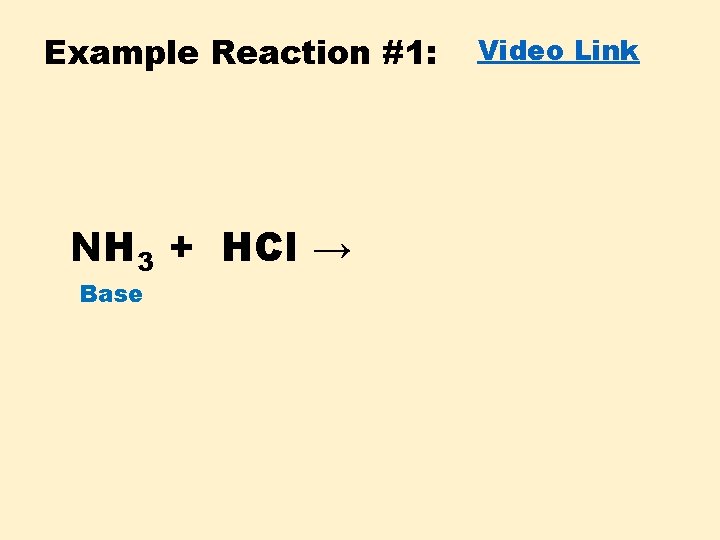
Example Reaction #1: NH 3 + HCl → Base Video Link
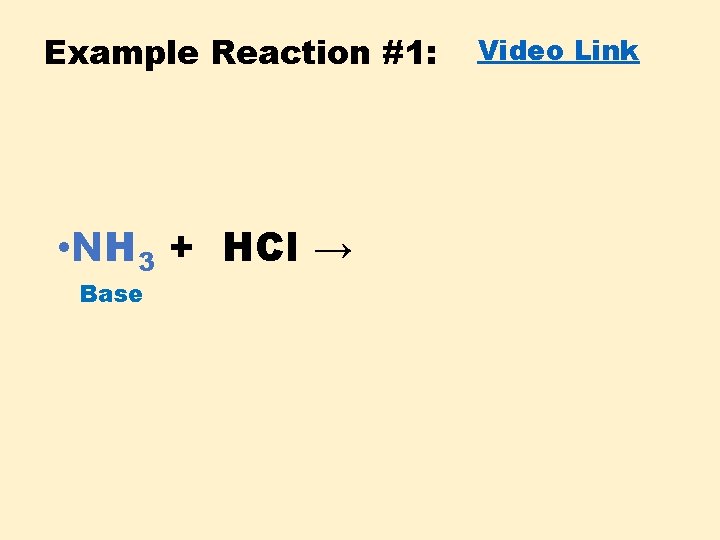
Example Reaction #1: • NH 3 + HCl → Base Video Link
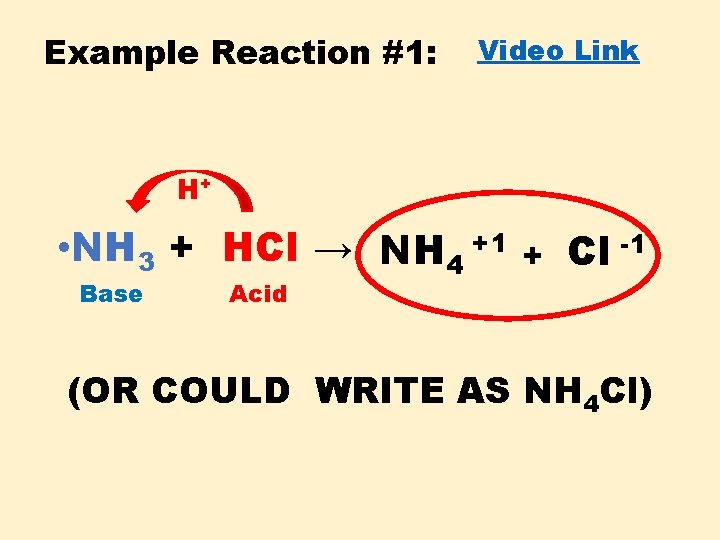
Example Reaction #1: Video Link H+ • NH 3 + HCl → NH 4 +1 Base Acid + Cl -1 (OR COULD WRITE AS NH 4 Cl)
Water is amphoteric meaning it can act as ACID or a BASE In a problem with a different chemical, identify the other chemical besides water as the acid or the base.
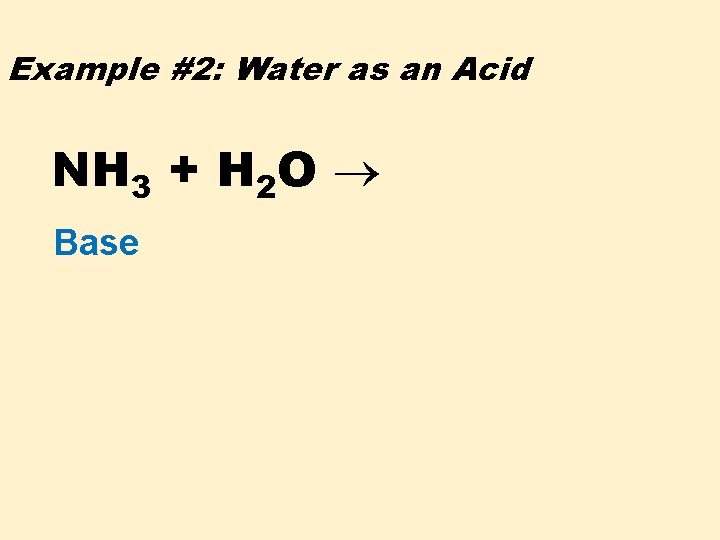
Example #2: Water as an Acid NH 3 + H 2 O Base
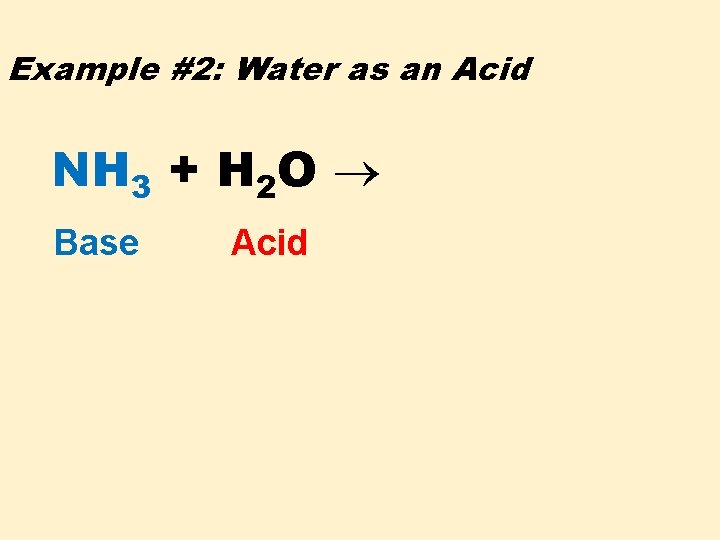
Example #2: Water as an Acid NH 3 + H 2 O Base Acid
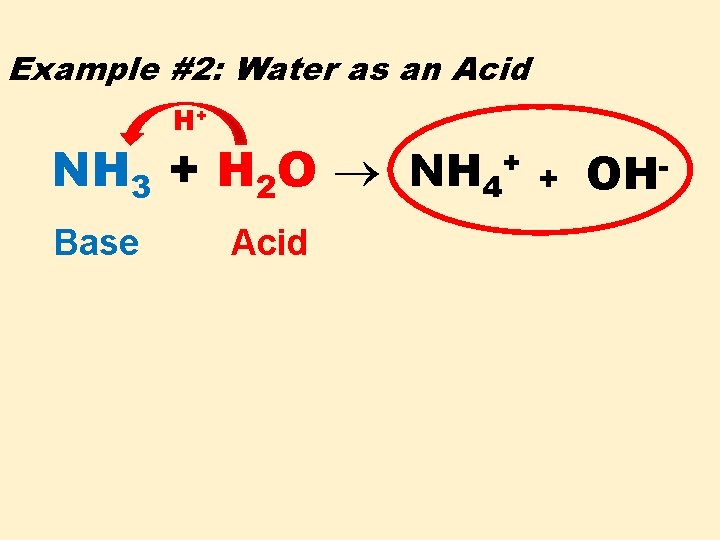
Example #2: Water as an Acid H+ NH 3 + H 2 O NH 4+ Base Acid + OH
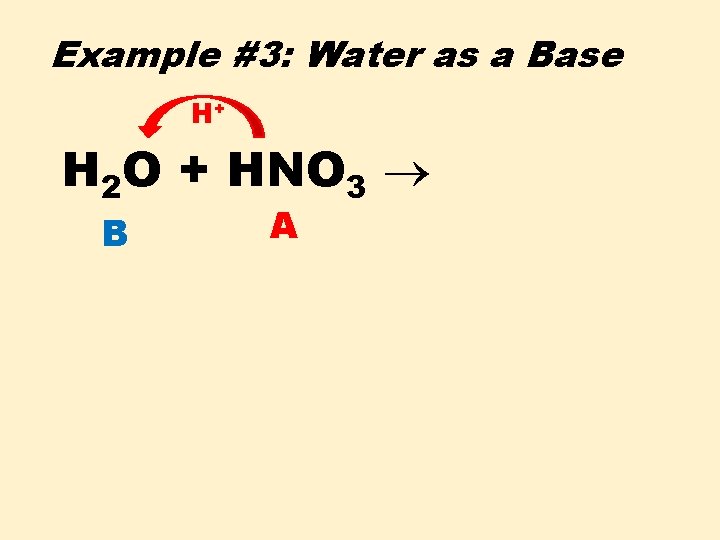
Example #3: Water as a Base H+ H 2 O + HNO 3 B A
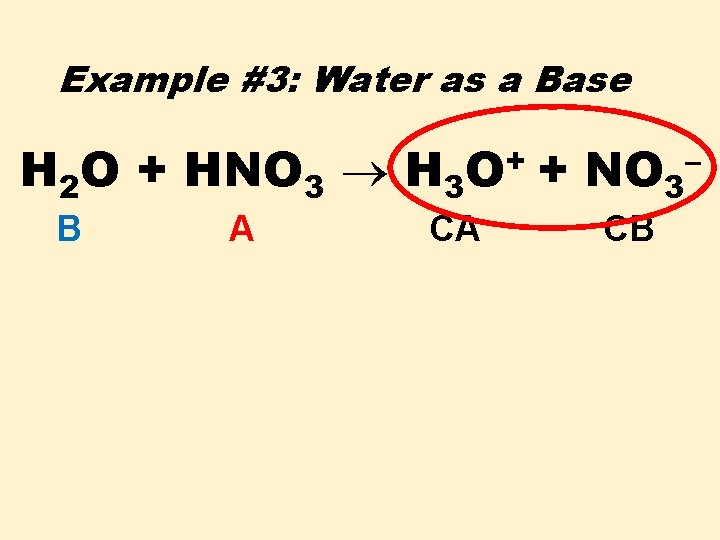
Example #3: Water as a Base H 2 O + HNO 3 H 3 B A + O CA + NO 3 CB –
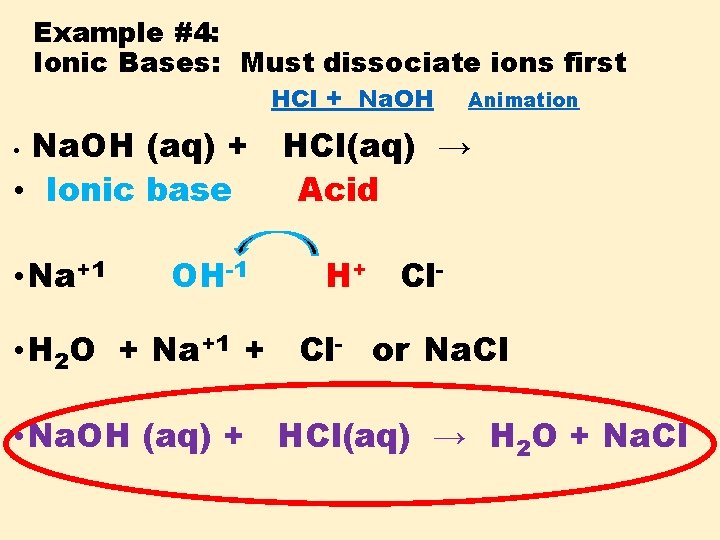
Example #4: Ionic Bases: Must dissociate ions first HCl + Na. OH Animation Na. OH (aq) + HCl(aq) → • Ionic base Acid • • Na+1 OH-1 H+ Cl- • H 2 O + Na+1 + Cl- or Na. Cl • Na. OH (aq) + HCl(aq) → H 2 O + Na. Cl
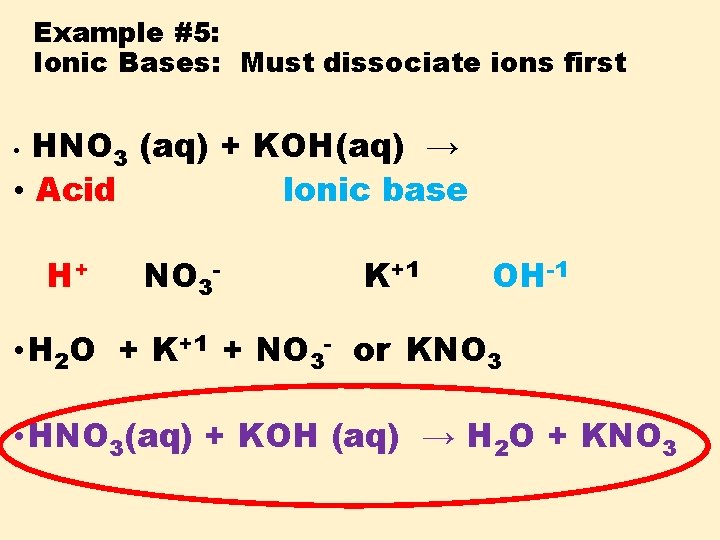
Example #5: Ionic Bases: Must dissociate ions first HNO 3 (aq) + KOH(aq) → • Acid Ionic base • H+ NO 3 - K+1 OH-1 • H 2 O + K+1 + NO 3 - or KNO 3 • HNO 3(aq) + KOH (aq) → H 2 O + KNO 3






- Slides: 35