AcidBase Reactions Adding a base to an acid

Acid-Base Reactions • Adding a base to an acid neutralizes the acid’s acidic properties. This reactions is called a neutralization reaction. • The products of a neutralization reaction are water and salt. • Acid + base water + salt • A salt is an ionic compound that is composed of the anion from the acid and the cation from the base.
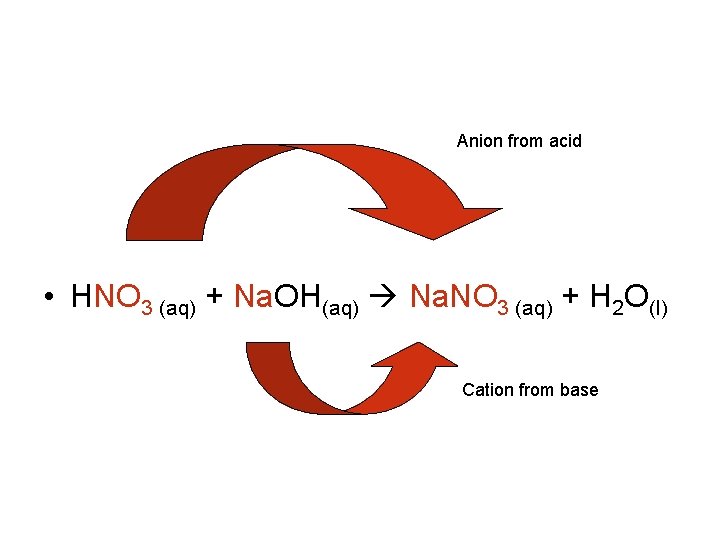
Anion from acid • HNO 3 (aq) + Na. OH(aq) Na. NO 3 (aq) + H 2 O(l) Cation from base

• The balanced chemical equation for this reaction shows that 1 mol of nitric acid reacts with 1 mol of sodium hydroxide. • If equal molar quantities of reactants are used, then the result is a neutral (p. H 7) aqueous solution. • For most neutralization reactions, there are no visible signs that a reaction is occurring. One way is to use an acid-base indicator, which is a substance that changes color in acidic or basic solutions.

• H+(indicator) H+ + indicator one color another color

Finding Concentration Example • 13. 84 m. L of hydrochloric acid just neutralizes 25. 00 m. L of a 0. 1000 mol/L solution of sodium hydroxide. What is the concentration of the hydrochloric acid? • Write the balanced equation • Calculate the amount of sodium hydroxide added, based on volume and concentration of solution • Determine the amount of hydrochloric acid needed to neutralize the sodium hydroxide • Find the concentration based on the amount and volume of hydrochloric acid solution needed.

• Balanced equation • HCl(aq) + Na. OH(aq) Na. Cl(aq) + HOH(l) • Calculate Na. OH • Amount Na. OH added (in mol) = Concentration x volume = 0. 1000 mol/L x 0. 025 00 L = 2. 500 x 10 -3 mol

• Determine HCl • HCl reacts with Na. OH in a 1: 1 ratio, so there must be 2. 500 x 10 -3 mol HCl • Concentration (mol/L) HCl = Amount (mol) volume (L) = 2. 500 x 10 -3 mol 0. 01384 L = 0. 1806 mol/L

Finding Volume Example • What volume of 0. 250 mol/L sulfuric acid is needed to react completely with 37. 2 m. L of 0. 650 mol/L potassium hydroxide? • Write a balanced equation • Calculate the amount (in mol) of potassium hydroxide • Determine the amount of sulfuric acid needed • Find the volume of sulfuric acid
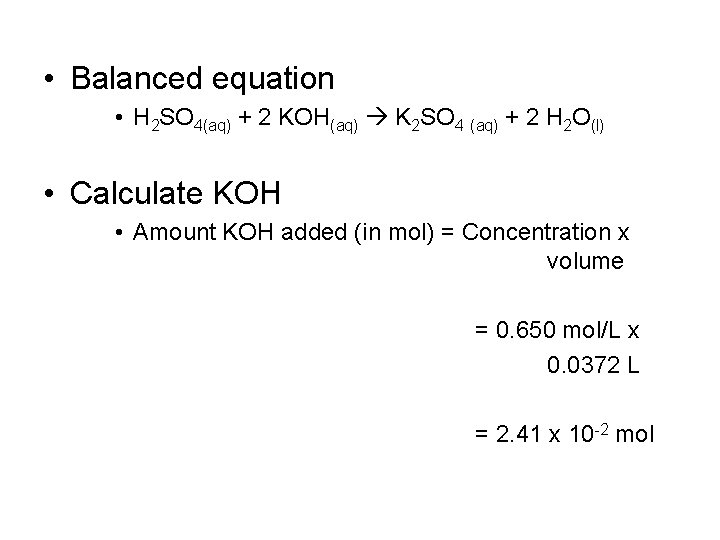
• Balanced equation • H 2 SO 4(aq) + 2 KOH(aq) K 2 SO 4 (aq) + 2 H 2 O(l) • Calculate KOH • Amount KOH added (in mol) = Concentration x volume = 0. 650 mol/L x 0. 0372 L = 2. 41 x 10 -2 mol

• Determine H 2 SO 4 • 1 H 2 SO 4 = x H 2 SO 4 2 KOH 2. 41 X 10 -2 • 1. 21 x 10 -2 mol H 2 SO 4 • Concentration (mol/L) HCl = Amount (mol) volume (L) Volume = Amount / Concentration = 1. 21 x 10 -2 mol / 0. 250 mol/L = 4. 84 x 10 -2 L
- Slides: 10