AcidBase Properties of Salts AcidBase Properties of Salts
Acid-Base Properties of Salts

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is from a strong base, anion from a strong acid KCl, KNO 3 Na. Cl Na. NO 3 Both ions are neutral p. H of solution Neutral These salts simply dissociate in water: KCl(s) K+(aq) + Cl-(aq)

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is from a strong base, anion from a weak acid Na. C 2 H 3 O 2 KCN, Na. F Cation is neutral, Anion is basic p. H of solution Basic The basic anion can accept a proton from water: C 2 H 3 O 2 - + H 2 O HC 2 H 3 O 2 + OHbase acid base
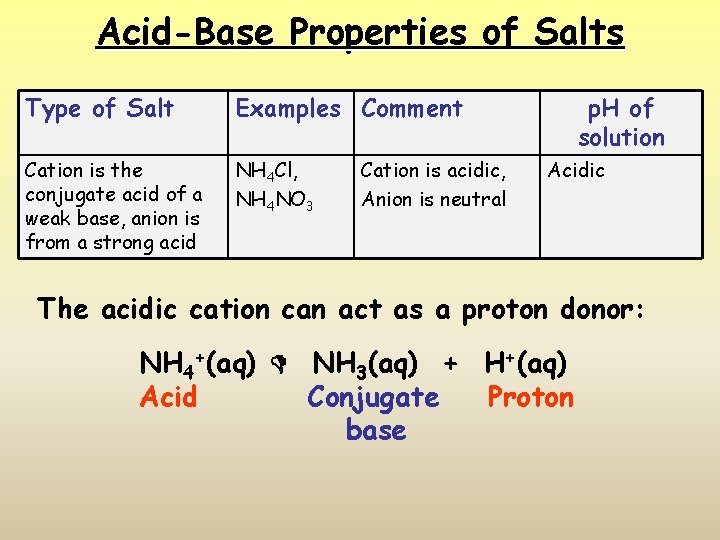
Acid-Base Properties of Salts Type of Salt Examples Comment Cation is the conjugate acid of a weak base, anion is from a strong acid NH 4 Cl, NH 4 NO 3 Cation is acidic, Anion is neutral p. H of solution Acidic The acidic cation can act as a proton donor: NH 4+(aq) NH 3(aq) + H+(aq) Acid Conjugate Proton base

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is the conjugate acid of a weak base, anion is conjugate base of a weak acid NH 4 C 2 H 3 O 2 Cation is acidic, NH 4 CN Anion is basic w w w p. H of solution See below IF Ka for the acidic ion is greater than Kb for the basic ion, the solution is acidic IF Kb for the basic ion is greater than Ka for the acidic ion, the solution is basic IF Kb for the basic ion is equal to Ka for the acidic ion, the solution is neutral

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is a highly Al(NO 3)2 charged metal ion; Fe. Cl 3 Anion is from strong acid Hydrated cation acts as an acid; Anion is neutral p. H of solution Acidic Step #1: Al. Cl 3(s) + 6 H 2 O Al(H 2 O)63+(aq) + Cl-(aq) Salt water Complex ion anion Step #2: Al(H 2 O)63+(aq) Al(OH)(H 2 O)52+(aq) + H+(aq) Acid Conjugate base Proton
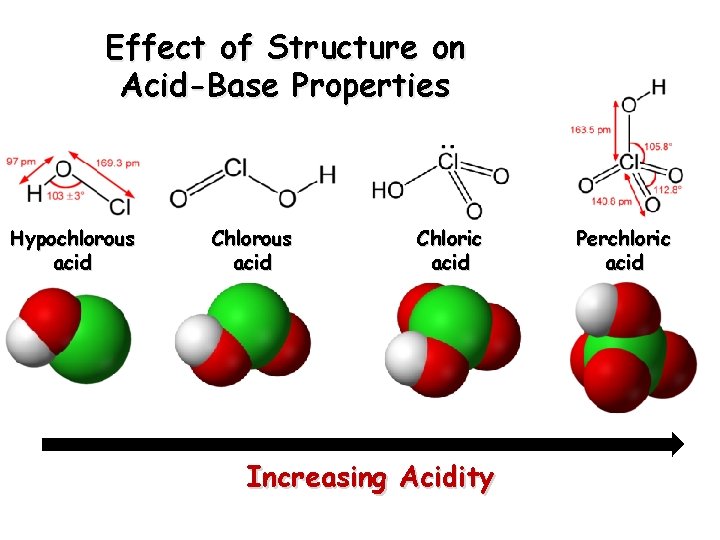
Effect of Structure on Acid-Base Properties Hypochlorous acid Chloric acid Increasing Acidity Perchloric acid
- Slides: 7