AcidBase Properties of Salts AcidBase Properties of Salt
Acid-Base Properties of Salts

Acid-Base Properties of Salt Solutions Because ions can exhibit acid or base properties (i. e. , donate or accept protons), salt solutions can be acidic or basic. -- hydrolysis: occurs when ions “react” w/H 2 O; the H 2 O is split into H+ and OH– Hydrolysis occurs when the acetate ion in sodium acetate reacts with water to produce acetic acid and hydroxide ion. OAc– + H 2 O HOAc + OH–

Anions of weak acids react with water to form OH–, and are thus basic. Example: Consider the weak acid HX(aq) H+(aq) + X–(aq) By the very definition of a weak acid, X– does NOT like to walk alone down the Boulevard of Broken Dreams. Now, say we have X–(aq) in solution from a salt. We’ll get. . . X – + H 2 O HX + OH– i. e. , X– accepts protons, acting like a base
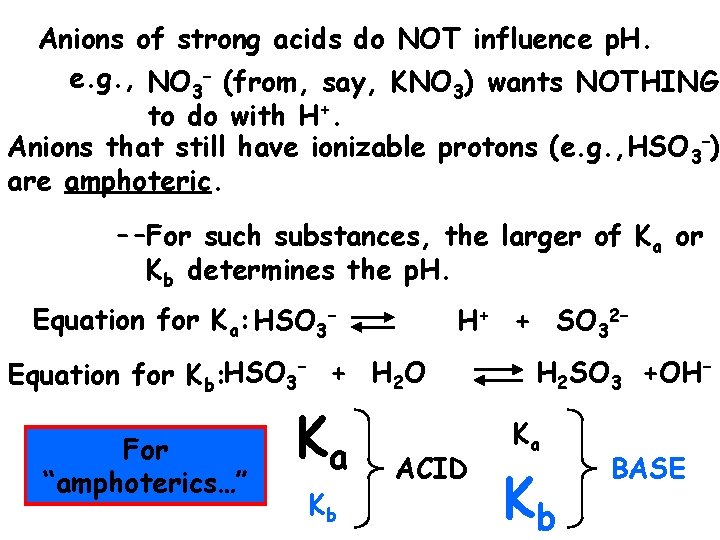
Anions of strong acids do NOT influence p. H. e. g. , NO 3– (from, say, KNO 3) wants NOTHING to do with H+. Anions that still have ionizable protons (e. g. , HSO 3–) are amphoteric. --For such substances, the larger of Ka or Kb determines the p. H. Equation for Ka: HSO 3– H+ + SO 32– Equation for Kb: HSO 3– + H 2 O For “amphoterics…” Ka Kb ACID H 2 SO 3 +OH– Ka Kb BASE
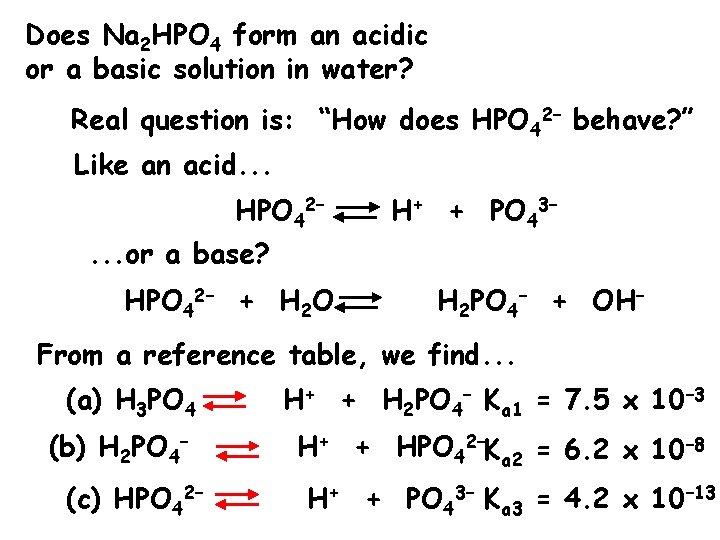
Does Na 2 HPO 4 form an acidic or a basic solution in water? Real question is: “How does HPO 42– behave? ” Like an acid. . . HPO 42– H+ + PO 43– . . . or a base? HPO 42– + H 2 O H 2 PO 4– + OH– From a reference table, we find. . . (a) H 3 PO 4 H+ + H 2 PO 4– Ka 1 = 7. 5 x 10– 3 (b) H 2 PO 4– H+ + HPO 42–Ka 2 = 6. 2 x 10– 8 (c) HPO 42– H+ + PO 43– Ka 3 = 4. 2 x 10– 13
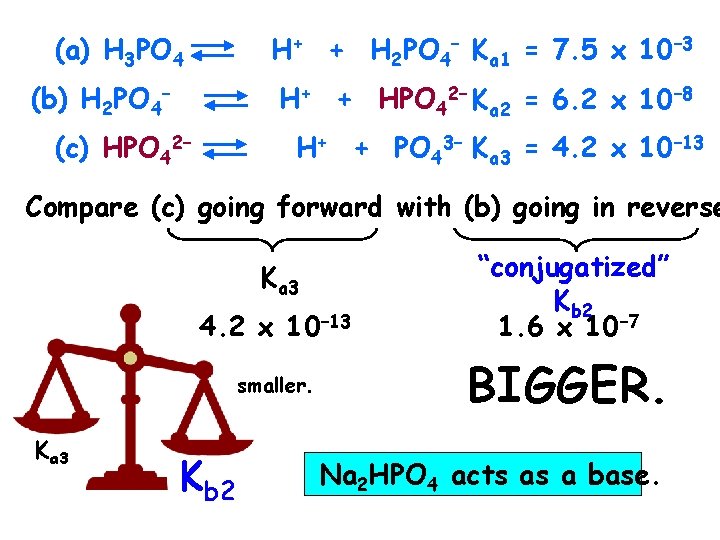
(a) H 3 PO 4 H+ + H 2 PO 4– Ka 1 = 7. 5 x 10– 3 (b) H 2 PO 4– H+ + HPO 42– Ka 2 = 6. 2 x 10– 8 (c) HPO 42– H+ + PO 43– Ka 3 = 4. 2 x 10– 13 Compare (c) going forward with (b) going in reverse Ka 3 4. 2 x 10– 13 smaller. Ka 3 Kb 2 “conjugatized” Kb 2 1. 6 x 10– 7 BIGGER. Na 2 HPO 4 acts as a base.

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is from a strong base, anion from a strong acid KCl, KNO 3 Na. Cl Na. NO 3 Both ions are neutral p. H of solution Neutral These salts simply dissociate in water: KCl(s) K+(aq) + Cl-(aq)

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is from a strong base, anion from a weak acid Na. C 2 H 3 O 2 KCN, Na. F Cation is neutral, Anion is basic p. H of solution Basic The basic anion can accept a proton from water: C 2 H 3 O 2 - + H 2 O HC 2 H 3 O 2 + OHbase acid base
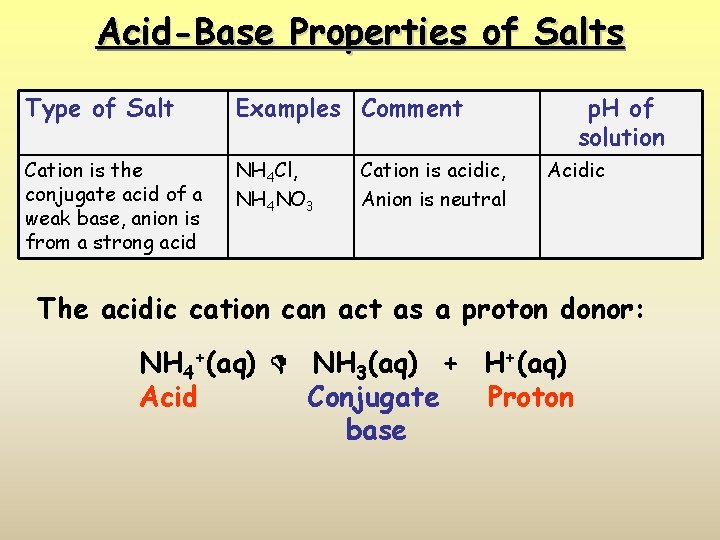
Acid-Base Properties of Salts Type of Salt Examples Comment Cation is the conjugate acid of a weak base, anion is from a strong acid NH 4 Cl, NH 4 NO 3 Cation is acidic, Anion is neutral p. H of solution Acidic The acidic cation can act as a proton donor: NH 4+(aq) NH 3(aq) + H+(aq) Acid Conjugate Proton base

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is the conjugate acid of a weak base, anion is conjugate base of a weak acid NH 4 C 2 H 3 O 2 Cation is acidic, NH 4 CN Anion is basic w w w p. H of solution See below IF Ka for the acidic ion is greater than Kb for the basic ion, the solution is acidic IF Kb for the basic ion is greater than Ka for the acidic ion, the solution is basic IF Kb for the basic ion is equal to Ka for the acidic ion, the solution is neutral

Acid-Base Properties of Salts Type of Salt Examples Comment Cation is a highly Al(NO 3)2 charged metal ion; Fe. Cl 3 Anion is from strong acid Hydrated cation acts as an acid; Anion is neutral p. H of solution Acidic Step #1: Al. Cl 3(s) + 6 H 2 O Al(H 2 O)63+(aq) + Cl-(aq) Salt water Complex ion anion Step #2: Al(H 2 O)63+(aq) Al(OH)(H 2 O)52+(aq) + H+(aq) Acid Conjugate base Proton
- Slides: 11