Acidbase equilibriums Buffer solutions Buffer solutions A solution

Acid-base equilibriums. Buffer solutions.

Buffer solutions • A solution that maintains a nearly constant p. H when a small amount of an acid or a base is added to it is called a buffer solution.

• A buffer is usually made by mixing a weak acid with its conjugate base, or by mixing a weak base with its conjugate acid. (For example CH 3 COOH/CH 3 COO-, + NH 3∙H 2 O/NH 4 …).

Buffer action • When an acid is added to a buffer it reacts with the base component of the buffer, when a base is added to the buffer it reacts with the acid component of the buffer. Thus the p. H of the buffer solution does not change.

• Acetate buffer solution and its buffer action CH 3 COOH ⇄ CH 3 COO- + H+ CH 3 COONa →CH 3 COO- + Na+ • when: H+ + CH 3 COO- → CH 3 COOH strong acid OH- + CH 3 COOH →CH 3 COO- + H 2 O strong base

Practical problem 1 •

Practical problem 2 •

Practical problem 3 •

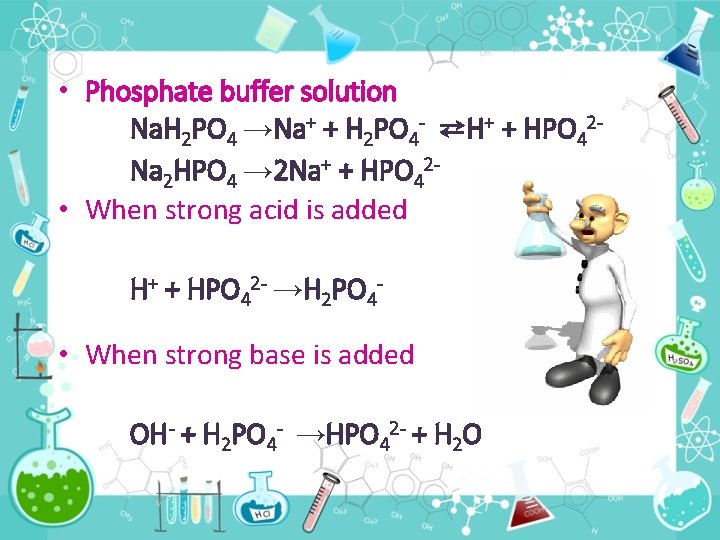
• Phosphate buffer solution Na. H 2 PO 4 →Na+ + H 2 PO 4 - ⇄H+ + HPO 42 Na 2 HPO 4 → 2 Na+ + HPO 42 • When strong acid is added H+ + HPO 42 - →H 2 PO 4 - • When strong base is added OH- + H 2 PO 4 - →HPO 42 - + H 2 O

Calculating the p. H of buffer solutions For acidic and salts buffers For basic buffers

Practical problem 4 •

Solution •

Practical problem 5 •


Practical problem 6 •

Solution •

Buffer systems in a blood Blood plasma Blood cells 1. Hydrocarbonate buffer(bicarbonate buffer). 1. Hemoglobin/Oxyhaemoglobin buffer. 2. Protein buffer. 2. Hydrocarbonate buffer. 3. Phosphate buffer
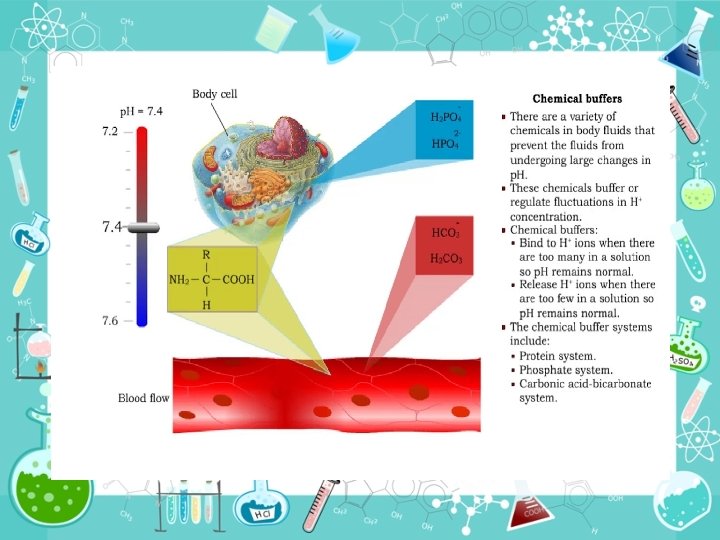

Buffer capacity •



A disturbance of acid-base status is rather dangerous for people’s health since p. H deviation may cause: Decrease in hormone and enzyme activity Change in osmotic pressure Alteration in rates of biochemical reactions catalyzed by protons

Even 0. 4 p. H units deviation from the normal p. H value in blood may cause coma or even death of a patient For babies even 0. 1 p. H deviations is also very dangerous

• The most dangerous types of acid-base disturbance in blood are: acidosis – increase in blood acidity and other body tissue alkalosis –increase in blood basicity Blood plasma p. H =7. 36
- Slides: 25