AcidBase Equilibrium Arrhenius Theory of Acids Bases Up

Acid/Base Equilibrium

Arrhenius’ Theory of Acids & Bases • Up until this point, you have learnt Arrhenius’ Theory: – – An acid dissociates in water to produce H+(aq) E. g. HCl(aq) H+(aq) + Cl-(aq) A base dissociates in water to produce OH-(aq) E. g. Na. OH(s) Na+(aq) + OH-(aq) • However, this theory could not explain exceptions like why NH 3 (ammonia) and Na. HCO 3 (baking soda) were considered bases
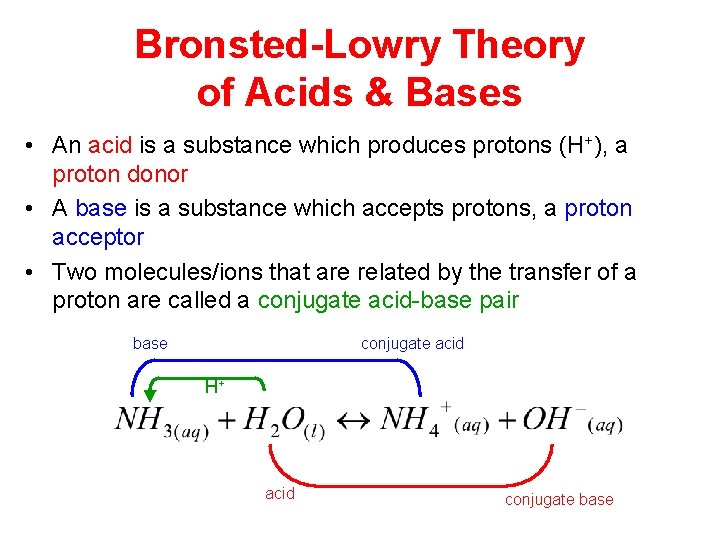
Bronsted-Lowry Theory of Acids & Bases • An acid is a substance which produces protons (H+), a proton donor • A base is a substance which accepts protons, a proton acceptor • Two molecules/ions that are related by the transfer of a proton are called a conjugate acid-base pair base conjugate acid H+ acid conjugate base

Conjugate Pairs Example • Identify the conjugate acid/base pairs and show the proton transfer acid conjugate base H+ base conjugate acid

Conjugate Pairs Example • Identify the conjugate acid/base pairs and show the proton transfer. • What is interesting about these systems? base conjugate acid H+ acid conjugate base H+ base conjugate acid Amphiprotic species can act as either an acid or a base. Has both an H-atom and a lone pair of electrons

Strong Acids and Bases • Dissociate completely • Quantitative reaction, no dynamic equilibrium Strong Acids Strong Bases All oxides & hydroxides of Group 1 & 2 metals except Be

Weak Acids and Bases • Do not dissociate completely • Dynamic equilibrium

Practice! • Worksheet • P. 532 #1, 2
- Slides: 8