AcidBase Equilibria Some Definitions Arrhenius An acid is

Acid-Base Equilibria

Some Definitions • Arrhenius – An acid is a substance that, when dissolved in water, increases the concentration of hydrogen ions. – A base is a substance that, when dissolved in water, increases the concentration of hydroxide ions.

Some Definitions • Brønsted-Lowry – An acid is a proton donor. – A base is a proton acceptor. A Brønsted-Lowry acid… …must have a removable (acidic) proton. A Brønsted-Lowry base… …must have a pair of nonbonding electrons.

If it can be either… …it is amphiprotic. HCO 3 HSO 4 H 2 O

What Happens When an Acid Dissolves in Water? • Water acts as a Brønsted-Lowry base and abstracts a proton (H+) from the acid. • As a result, the conjugate base of the acid and a hydronium ion are formed.

Conjugate Acids and Bases • The term conjugate comes from the Latin word “conjugare, ” meaning “to join together. ” • Reactions between acids and bases always yield their conjugate bases and acids.

Acid and Base Strength • Strong acids are completely dissociated in water. – Their conjugate bases are quite weak. • Weak acids only dissociate partially in water. – Their conjugate bases are weak bases.

Acid and Base Strength • Substances with negligible acidity do not dissociate in water. – Their conjugate bases are exceedingly strong. Example: CH 4 contains hydrogen but does not demonstrate any acidic behavior in water. Its conjugate base (CH 3 -) is a strong base.

Acid and Base Strength • In any acid-base reaction, the equilibrium will favor the reaction that moves the proton to the stronger base. HCl (aq) + H 2 O (l) H 3 O+ (aq) + Cl- (aq) • H 2 O is a much stronger base than Cl-, so the equilibrium lies so far to the right that K is not measured (K>>1).

Acid and Base Strength • In any acid-base reaction, the equilibrium will favor the reaction that moves the proton to the stronger base. CH 3 CO 2 H (aq) + H 2 O (l) H 3 O+ (aq) + CH 3 CO 2 - (aq) • Acetate is a stronger base than H 2 O, so the equilibrium favors the left side (K<1).

Autoionization of Water • As we have seen, water is amphoteric. • In pure water, a few molecules act as bases and a few act as acids. H 2 O (l) + H 2 O (l) H 3 O+ (aq) + OH- (aq) • This is referred to as autoionization.

Ion-Product Constant • The equilibrium expression for this process is Kc = [H 3 O+] [OH-] • This special equilibrium constant is referred to as the ion-product constant for water, Kw. • At 25 C, Kw = 1. 0 10 -14

p. H is defined as the negative logarithm in base 10 of the concentration of hydronium ion. p. H = -log [H 3 O+] or p. H = -log [H+]
![p. H • In pure water, Kw = [H 3 O+] [OH-] = 1. p. H • In pure water, Kw = [H 3 O+] [OH-] = 1.](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-14.jpg)
p. H • In pure water, Kw = [H 3 O+] [OH-] = 1. 0 10 -14 • Since in pure water [H 3 O+] = [OH-], [H 3 O+] = 1. 0 10 -14 = 1. 0 10 -7

p. H • Therefore, in pure water, p. H = -log (1. 0 10 -7) = 7. 00 • An acid has a higher [H 3 O+] than pure water, so its p. H is <7. • A base has a lower [H 3 O+] than pure water, so its p. H is >7.
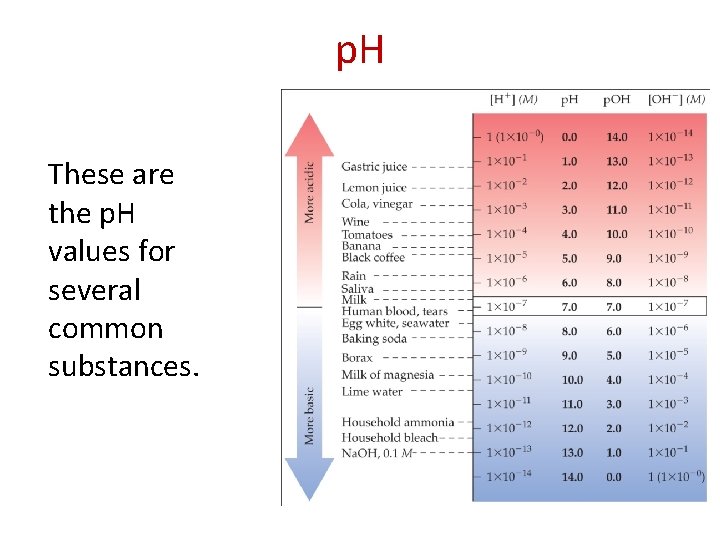
p. H These are the p. H values for several common substances.

Other “p” Scales • The “p” in p. H tells us to take the negative base-10 logarithm of the quantity (in this case, hydronium ions). • Some similar examples are – p. OH: -log [OH-] – p. Kw: -log Kw
![Watch This! Because [H 3 O+] [OH-] = Kw = 1. 0 10 -14, Watch This! Because [H 3 O+] [OH-] = Kw = 1. 0 10 -14,](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-18.jpg)
Watch This! Because [H 3 O+] [OH-] = Kw = 1. 0 10 -14, we know that -log [H 3 O+] + -log [OH-] = -log Kw = 14. 00 or, in other words, p. H + p. OH = p. Kw = 14. 00

How Do We Measure p. H? • For less accurate measurements, one can use – Litmus paper • “Red” paper turns blue above ~p. H = 8 • “Blue” paper turns red below ~p. H = 5 – Or an indicator.

How Do We Measure p. H? For more accurate measurements, one uses a p. H meter, which measures the voltage in the solution.

Strong Acids • The seven strong acids are HCl, HBr, HI, HNO 3, H 2 SO 4, HCl. O 3, and HCl. O 4. • These are, by definition, strong electrolytes and exist totally as ions in aqueous solution. • For the monoprotic strong acids, [H 3 O+] = [acid].

Strong Bases • Strong bases are the soluble hydroxides, which are the alkali metal and heavier alkaline earth metal hydroxides (Ca 2+, Sr 2+, and Ba 2+). • Again, these substances dissociate completely in aqueous solution.

Weak Acids Dissociation Constants • For a generalized acid dissociation, HA (aq) + H 2 O (l) A- (aq) + H 3 O+ (aq) the equilibrium expression would be Kc = [H 3 O+] [A-] [HA] • This equilibrium constant is called the aciddissociation constant, Ka.

Dissociation Constants The greater the value of Ka, the stronger is the acid.

Calculating Ka from the p. H The p. H of a 0. 10 M solution of formic acid, HCOOH, at 25 C is 2. 38. Calculate Ka formic acid at this temperature. We know that HCOOH + H 2 O H 3 O+ + HCOO- [H 3 O+] [HCOO-] Ka = [HCOOH]

Calculating Ka from the p. H To calculate Ka, we need the equilibrium concentrations of all three things. We can find [H 3 O+], which is the same as [HCOO-], from the p. H.
![p. H = -log [H 3 O+] 2. 38 = -log [H 3 O+] p. H = -log [H 3 O+] 2. 38 = -log [H 3 O+]](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-27.jpg)
p. H = -log [H 3 O+] 2. 38 = -log [H 3 O+] -2. 38 = log [H 3 O+] 4. 2 10 -3 = [H 3 O+] = [HCOO-] Ka = [4. 2 10 -3] [0. 10] = 1. 8 10 -4
![Calculating Percent Ionization [H 3 O+]eq • Percent Ionization = 100 [HA]initial • In Calculating Percent Ionization [H 3 O+]eq • Percent Ionization = 100 [HA]initial • In](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-28.jpg)
Calculating Percent Ionization [H 3 O+]eq • Percent Ionization = 100 [HA]initial • In this example [H 3 O+]eq = 4. 2 10 -3 M [HCOOH]initial = 0. 10 M 4. 2 10 -3 Percent Ionization = 0. 10 100 = 4. 2%

Calculating p. H from Ka Calculate the p. H of a 0. 30 M solution of acetic acid, HC 2 H 3 O 2, at 25 C. HC 2 H 3 O 2 (aq) + H 2 O (l) H 3 O+ (aq) + C 2 H 3 O 2 - (aq) Ka for acetic acid at 25 C is 1. 8 10 -5.
![The equilibrium constant expression is +] [C H O -] [H O 2 3 The equilibrium constant expression is +] [C H O -] [H O 2 3](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-30.jpg)
The equilibrium constant expression is +] [C H O -] [H O 2 3 2 Ka = 3 [HC 2 H 3 O 2] 2 (x) 1. 8 10 -5 = (0. 30) 2. 3 10 -3 = x p. H = -log [H 3 O+] p. H = -log (2. 3 10 -3) p. H = 2. 64
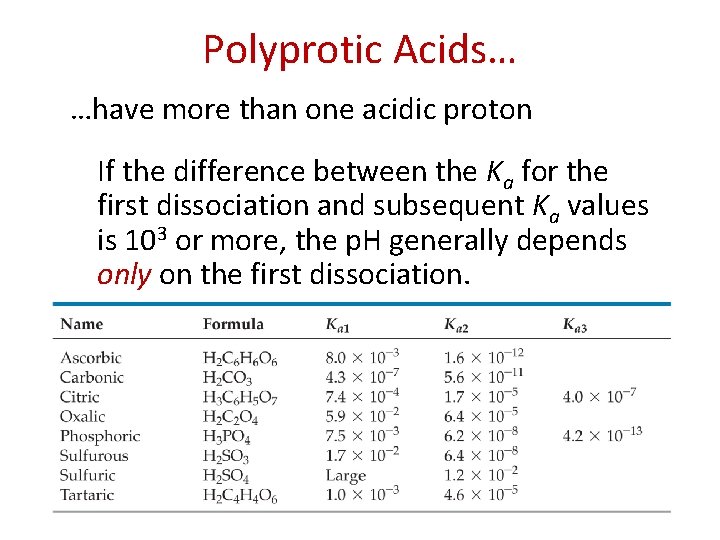
Polyprotic Acids… …have more than one acidic proton If the difference between the Ka for the first dissociation and subsequent Ka values is 103 or more, the p. H generally depends only on the first dissociation.
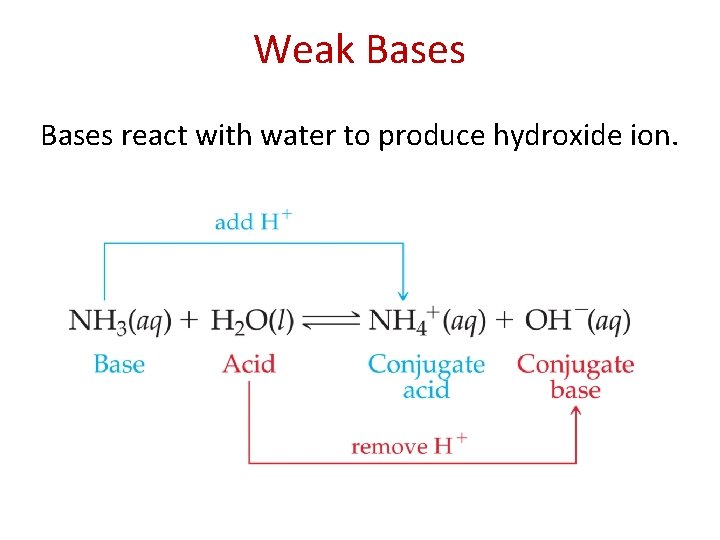
Weak Bases react with water to produce hydroxide ion.
![Weak Bases The equilibrium constant expression for this reaction is [HB] [OH-] Kb = Weak Bases The equilibrium constant expression for this reaction is [HB] [OH-] Kb =](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-33.jpg)
Weak Bases The equilibrium constant expression for this reaction is [HB] [OH-] Kb = [B-] where Kb is the base-dissociation constant.
![Weak Bases Kb can be used to find [OH-] and, through it, p. H. Weak Bases Kb can be used to find [OH-] and, through it, p. H.](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-34.jpg)
Weak Bases Kb can be used to find [OH-] and, through it, p. H.
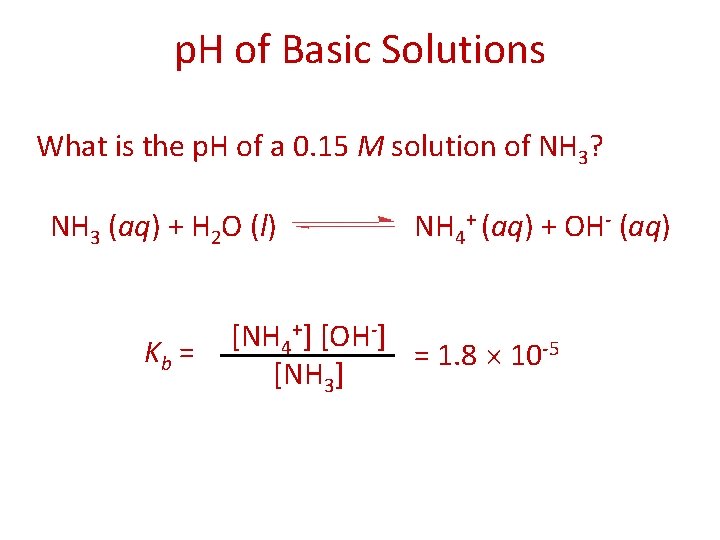
p. H of Basic Solutions What is the p. H of a 0. 15 M solution of NH 3? NH 3 (aq) + H 2 O (l) Kb = NH 4+ (aq) + OH- (aq) [NH 4+] [OH-] = 1. 8 10 -5 [NH 3]
![Kb = [NH 4+] [OH-] = 1. 8 10 -5 [NH 3] 1. 8 Kb = [NH 4+] [OH-] = 1. 8 10 -5 [NH 3] 1. 8](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-36.jpg)
Kb = [NH 4+] [OH-] = 1. 8 10 -5 [NH 3] 1. 8 10 -5 = (x)2 (0. 15) 1. 6 10 -3 = x 2 Therefore, [OH-] = 1. 6 10 -3 M p. OH = -log (1. 6 10 -3) p. OH = 2. 80 p. H = 14. 00 - 2. 80 p. H = 11. 20

Ka and Kb are related in this way: Ka Kb = Kw Therefore, if you know one of them, you can calculate the other.

Reactions of Anions with Water • Anions are bases. • As such, they can react with water in a hydrolysis reaction to form OH- and the conjugate acid: X- (aq) + H 2 O (l) HX (aq) + OH- (aq)

Reactions of Cations with Water • Cations with acidic protons (like NH 4+) will lower the p. H of a solution. • Most metal cations that are hydrated in solution also lower the p. H of the solution.

Reactions of Cations with Water • Attraction between nonbonding electrons on oxygen and the metal causes a shift of the electron density in water. • This makes the O-H bond more polar and the water more acidic. • Greater charge and smaller size make a cation more acidic.

The Common-Ion Effect Consider a solution of acetic acid: CH 3 COOH(aq) + H 2 O(l) H 3 O+(aq) + CH 3 COO−(aq) If acetate ion is added to the solution, Le Châtelier says the equilibrium will shift to the left.

The Common-Ion Effect Calculate the fluoride ion concentration and p. H of a solution that is 0. 20 M in HF and 0. 10 M in HCl. Ka for HF is 6. 8 10− 4. [H 3 O+] [F−] Ka = = 6. 8 10 -4 [HF]

The Common-Ion Effect HF(aq) + H 2 O(l) H 3 O+(aq) + F−(aq) Because HCl, a strong acid, is also present, the initial [H 3 O+] is not 0, but rather 0. 10 M. [HF], M [H 3 O+], M [F−], M Initially 0. 20 0. 10 0 Change −x +x +x 0. 20 − x 0. 20 0. 10 + x 0. 10 x At Equilibrium

The Common-Ion Effect 6. 8 10− 4 (0. 10) (x) = (0. 20) 1. 4 10− 3 = x Therefore, [F−] = x = 1. 4 10− 3 [H 3 O+] = 0. 10 + x = 0. 10 + 1. 4 10− 3 = 0. 10 M So, p. H = −log (0. 10) p. H = 1. 00

Buffers • Buffers are solutions of a weak conjugate acidbase pair. • They are particularly resistant to p. H changes, even when strong acid or base is added.
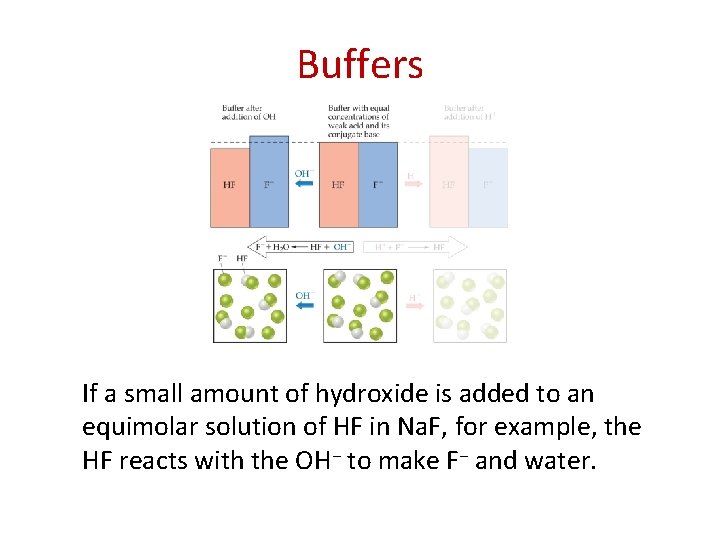
Buffers If a small amount of hydroxide is added to an equimolar solution of HF in Na. F, for example, the HF reacts with the OH− to make F− and water.
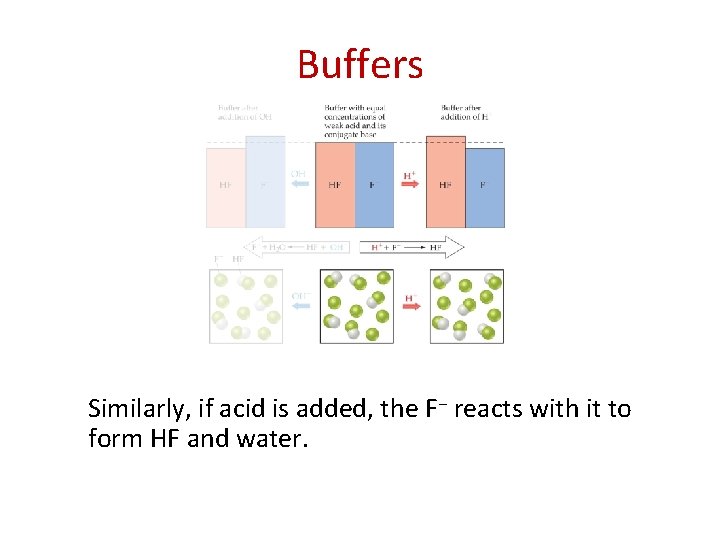
Buffers Similarly, if acid is added, the F− reacts with it to form HF and water.

Buffer Calculations Consider the equilibrium constant expression for the dissociation of a generic acid, HA: HA + H 2 O [H 3 O+] [A−] Ka = [HA] H 3 O + + A −
![Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA]](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-49.jpg)
Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Taking the negative log of both side, we get −] [A −log Ka = −log [H 3 O+] + −log [HA] p. Ka p. H base acid
![Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging, Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging,](http://slidetodoc.com/presentation_image/559e4c303315a1738ec7185f963c44e1/image-50.jpg)
Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging, this becomes [base] p. H = p. Ka + log [acid] • This is the Henderson–Hasselbalch equation.

What is the p. H of a buffer that is 0. 12 M in lactic acid, CH 3 CH(OH)COOH, and 0. 10 M in sodium lactate? Ka for lactic acid is 1. 4 10− 4. [base] p. H = p. Ka + log [acid] (0. 10) p. H = −log (1. 4 10− 4) + log (0. 12) p. H = 3. 85 + (− 0. 08) p. H = 3. 77

p. H Range • The p. H range is the range of p. H values over which a buffer system works effectively. • It is best to choose an acid with a p. Ka close to the desired p. H.

When Strong Acids or Bases Are Added to a Buffer… …it is safe to assume that all of the strong acid or base is consumed in the reaction.

Calculating p. H Changes in Buffers A buffer is made by adding 0. 300 mol HC 2 H 3 O 2 and 0. 300 mol Na. C 2 H 3 O 2 to enough water to make 1. 00 L of solution. The p. H of the buffer is 4. 74. Calculate the p. H of this solution after 0. 020 mol of Na. OH is added.
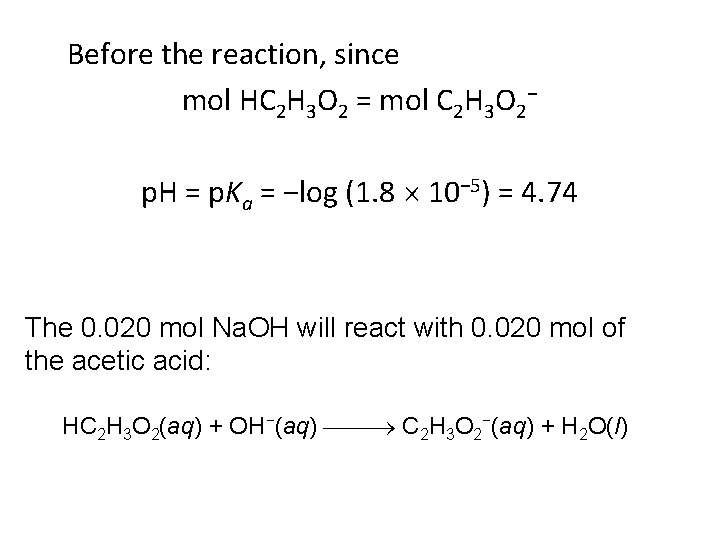
Before the reaction, since mol HC 2 H 3 O 2 = mol C 2 H 3 O 2− p. H = p. Ka = −log (1. 8 10− 5) = 4. 74 The 0. 020 mol Na. OH will react with 0. 020 mol of the acetic acid: HC 2 H 3 O 2(aq) + OH−(aq) C 2 H 3 O 2−(aq) + H 2 O(l)

HC 2 H 3 O 2 C 2 H 3 O 2 − OH− Before reaction 0. 300 mol 0. 020 mol After reaction 0. 280 mol 0. 320 mol 0. 000 mol Now use the Henderson–Hasselbalch equation to calculate the new p. H: p. H = 4. 74 + log p. H = 4. 74 + 0. 06 p. H = 4. 80 (0. 320) (0. 280)

Solubility Products Consider the equilibrium that exists in a saturated solution of Ba. SO 4 in water: Ba. SO 4(s) Ba 2+(aq) + SO 42−(aq) The equilibrium constant expression for this equilibrium is Ksp = [Ba 2+] [SO 42−] where the equilibrium constant, Ksp, is called the solubility product.
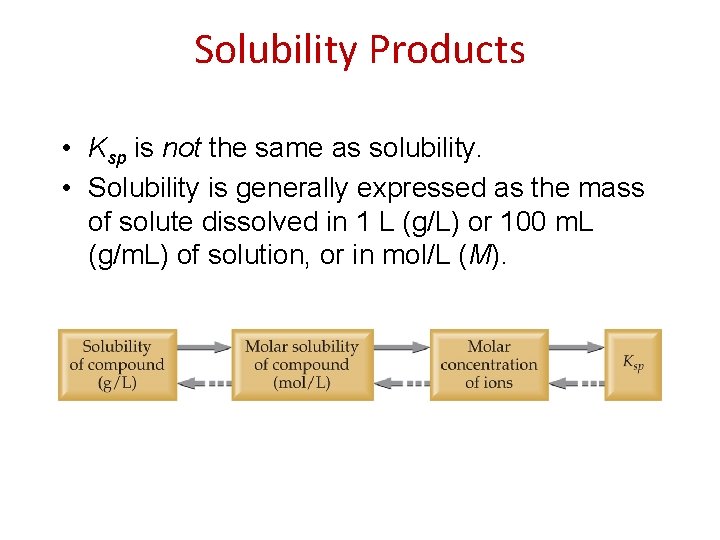
Solubility Products • Ksp is not the same as solubility. • Solubility is generally expressed as the mass of solute dissolved in 1 L (g/L) or 100 m. L (g/m. L) of solution, or in mol/L (M).
- Slides: 58